A. Palav, B. Misal, P. Ganwir et al.
Tetrahedron Letters 73 (2021) 153094
aromatics without any activation. Due to its high electrophilicity, it
may satisfy the criteria of an ideal oxidizing agent. Although the
reagent is also used as a starting material for preparing N-trifluoro
methyl thiolating reagent [34] however, its potential as an oxidiz-
ing agent is being explored for the first time.
According to the physicochemical parameters of N-chloro
reagents and NCBSI in-silico by density-functional theory (DFT)
theory, NCBSI exhibited the highest electrophilic nature and reac-
tivity due to a longer N-Cl bond length, lower bond dissociation
energy (BDE), and lower absolute charge density on chlorine
a longer time (Table 3, entries 3, 4, 6, and 7). In the case of nitro
alcohols, 4-nitrobenzyl alcohol, and 1-(4-nitrophenyl)ethanol
(Table 3, entries 9 and 13) were oxidized smoothly to 4-nitroben-
zaldehyde and 4-nitroacetophenone. For 1,4-phenylene dimetha-
nol selective mono oxidation was not observed using one mole
equivalent of the reagent. With 2 mole equivalents of reagent, both
the alcohols were successfully oxidized to obtain terephthalalde-
hyde (Table 3, entry 10). The reagent was selective towards 1,2-
diphenylethane-1,2-diol, resulting in mono keto product (Table 3,
entry 17). Aliphatic alcohols oxidized to ketones and aldehyde in
good yields under solvent-free condition (Table 3, entries 18, 19).
Alicyclic alcohols (±) cyclohexanol, (±) menthol, and (±) borneol
were also oxidized smoothly to cyclohexanone, menthone, and
camphor (Table 3, entries 20–21).
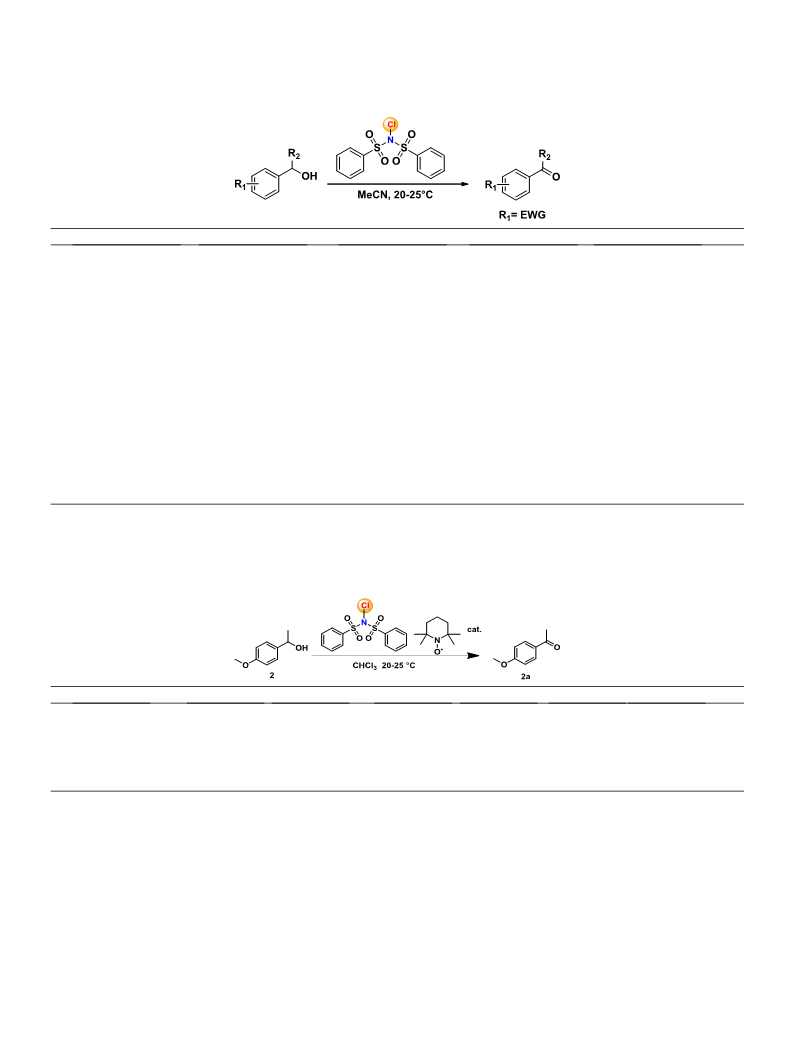
When the same protocol was applied for the oxidation electron-
rich alcohol, viz. 1-(4-methoxyphenyl)ethanol, the ring chlorina-
tion dominated over oxidation due to the high electrophilic nature
of the reagent, and traces of oxidized product was obtained (see
supplementary information Fig. 3). The foresaid optimized proto-
col was not suitable for the oxidation of electron-rich aromatic
alcohols in a polar solvent. To achieve selective oxidation, there
was a need to alter the mechanism by modifying the ionic charac-
ter of the reagent by trapping-transfer of chlorine to dominate the
oxidation over chlorination. Since TEMPO is widely used for oxida-
tion of alcohol with a variety of secondary oxidant [35,36] and lit-
erature [22] encouraged us to use it in a catalytic amount. The
study began with a pilot reaction with a catalytic amount of
TEMPO (1.49% to 5.35 mol%) in acetonitrile, chlorinated product
dominated over oxidation (Table 4, entry 8). The loading of TEMPO
was optimized to 4.38 mol% for the oxidation of 1-(4-methoxyphe-
nyl)ethanol (Table 4, entry 4,). The oxidation was slower in sol-
[
33]. Overwhelmed with these findings, we eagerly performed oxi-
dation of benzyl alcohol using N-chloro reagents including NCBSI,
in the absence of an additive in specially dried acetonitrile under
an inert atmosphere at 20–25 °C (Table 1). No oxidation product
was observed in NCS, NCPI, and TCCA. This revealed that shorter
N-Cl bond length and higher BDE affect the oxidation. NCSAC and
DCDMH showed marginal oxidation products for over 12 h.
Although the values of these N-Cl reagents are close to NCBSI, both
had comparatively shorter bond lengths. Hence, practically NCBSI
is a rapid oxidant that resulted in the complete conversion of ben-
zyl alcohol to benzaldehyde in acetonitrile (MeCN) in 5 min with
9
8% yield and 99.8% purity (Table 1, entry 6).
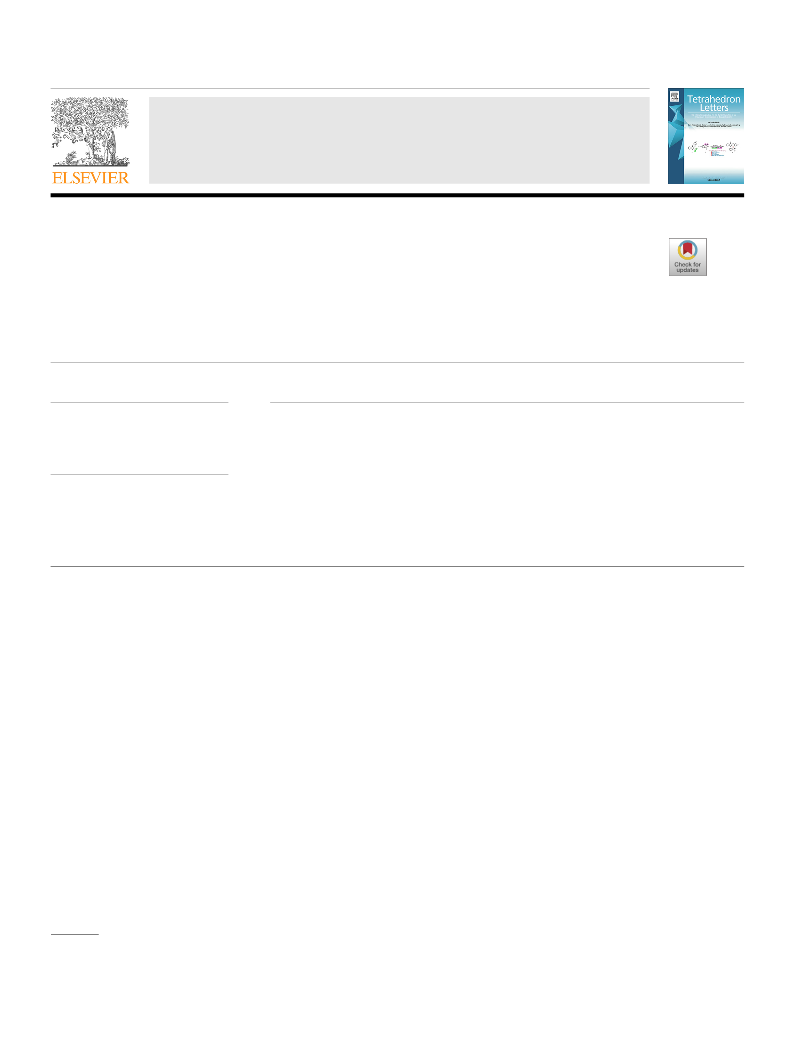
A solvent optimization study was also performed (Table 2). In
chlorinated solvents, the time for the reaction was increased from
dichloromethane (DCM) to carbon tetrachloride (CCl ), due to
4
decreased solubility of the reagent (Table 2, entries 2–5). In DMF,
the oxidation was highly exothermic, resulted in low yield due to
work-up issues (Table 2, entry 6). To study, the generality and
scope, the optimized reaction condition were applied to primary
and secondary alcohols with electron-withdrawing and electron-
donating substitution on aromatic rings (Table 3)
We applied optimized condition to electron-deficient alcohols,
in which unsubstituted and 4-halo substituted benzyl alcohols
were oxidized readily with excellent yield (Table 3, entries 1, 2,
3
vents other than chloroform (CHCl ) (Table 4, entries 6, 7).
Keeping eco-friendliness and recovery on priority, the use of
polymer-supported TEMPO was necessary. Besides the availability
of a wide variety of supported TEMPO reagents [37,38], we chose a
5
, 8, and 12). On the other hand, 2 and 3 halo benzyl alcohols took
Table 1
a
Comparison of commercial N-chloro reagents with (NCBSI).
b
c
Entry
Reagent
SM (%)
Time
Yield (%)
e
1
2
3
4
5
6
NCS
NCSAC
TCCA
NCPI
DCDMH
NCBSI
100
98
100
100
94
12 h
12 h
12 h
12 h
12 h
5 min.
NR
2
e
NR
e
NR
6
98
d
0
a
b
c
d
e
Reaction condition: benzyl alcohol (3.6 mmol), oxidant (3.6 mmol) in 5 mL MeCN at 20–25 °C. SM recovery by HPLC. Yield by HPLC. Isolated yield NR-No reaction.
Table 2
Solvent optimization study.
a
b
c
Entry
Solvent
Time (min.)
Yield (%)
Purity (%)
1
2
3
4
5
6
MeCN
DCM
EDC
5
98
95
96
95
94
70
99.8
99
99.2
99
98
99
10
10
15
20
2
CHCl
CCl
DMF
3
4
a
b
c
Reaction condition: benzyl alcohol (3.6 mmol), NCBSI (3.6 mmol), 5 mL solvent, 20–25 °C. Isolated Yield. Purity by HPLC. EDC = Ethylene dichloride.
2

 Badani, Purav
Badani, Purav
 Chaturbhuj, Ganesh
Chaturbhuj, Ganesh
 Ganwir, Prerna
Ganwir, Prerna
 Misal, Balu
Misal, Balu
 Palav, Amey
Palav, Amey