
Tetrahedron p. 5699 - 5702 (2008)
Update date:2022-08-11
Topics:
 Kamitanaka, Takashi
Kamitanaka, Takashi
 Yamamoto, Kenji
Yamamoto, Kenji
 Matsuda, Tomoko
Matsuda, Tomoko
 Harada, Tadao
Harada, Tadao
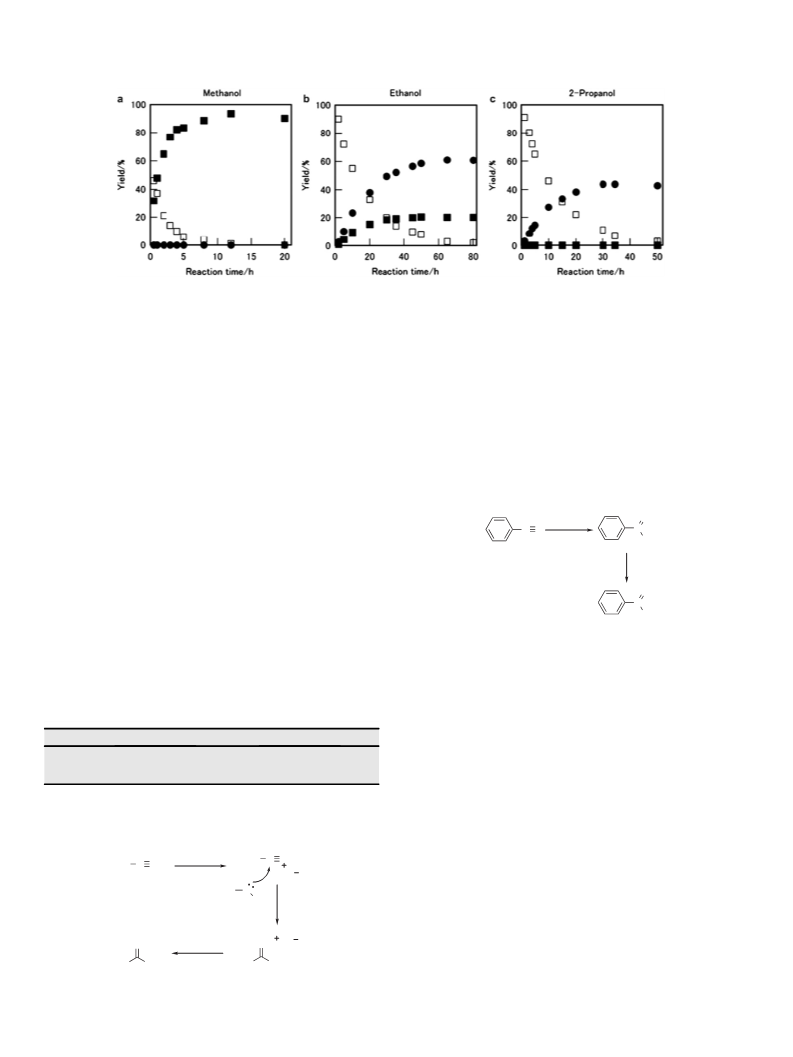
The reactions of benzonitrile in supercritical methanol, ethanol, and 2-propanol were investigated under non-catalytic conditions. In supercritical methanol, benzonitrile was converted to methyl benzoate in high yield. The esterification reaction also occurred in supercritical ethanol to afford ethyl benzoate in moderate yield. The esterification could occur via a route analogous to the Pinner reaction. On the other hand, benzonitrile in supercritical 2-propanol yielded no ester. Benzyl alcohol was the major product in supercritical 2-propanol. We investigated the reaction of the C{double bond, long}N bond in supercritical 2-propanol. In supercritical 2-propanol, N-benzylideneaniline was transferred to the reduction product (N-benzylaniline) and hydrolysis products (benzyl alcohol and aniline). The hydrolysis reaction was restricted when the reaction was carried out in supercritical 2-propanol with a low water content. This indicates that the water in the 2-propanol acts as a reagent for the hydrolysis of the C{double bond, long}N bond. These results suggested the following reaction process: C6H5C{triple bond, long}N→C6H5CH{double bond, long}NH→C6H5CHO→C6H5CH2OH.
View More

Buffett (China) Holding Co.,Ltd
Contact:4006570891
Address:
Contact:+86 0310 3166373
Address:shanghai
Shanghai PotentPharm Science and Technology Co.,Ltd
Contact:86-021-51969655
Address:Unit B, Building 18, No.300, Chuantu Rd,Pudong District, Shanghai 201202, China
Penglai Qianwei Chemical Co., Ltd.
Contact:86-535-3357802
Address:Shahelu (north), Penglai, Shandong, China
Wuhan Kemi-Works Chemical Co., Ltd
website:http://www.kemiworks.com
Contact:86-27-85736489
Address:Rm. 1503, No. 164, Jianghan North Rd., Wuhan, 430022 China
Doi:10.1055/s-1998-1972
(1998)Doi:10.1021/jo030252t
(2003)Doi:10.1021/ol048847d
(2004)Doi:10.1016/S0040-4039(00)01569-0
(2000)Doi:10.1039/c7ra06181a
(2017)Doi:10.1039/d0ob01790f
(2020)