Published on Web 10/22/2008
Synthesis and Degradation of Nucleic Acid Components by
Formamide and Iron Sulfur Minerals
Raffaele Saladino,*,† Veronica Neri,† Claudia Crestini,‡ Giovanna Costanzo,§
Michele Graciotti,| and Ernesto Di Mauro*,|
Dipartimento di Agrobiologia ed Agrochimica, UniVersita` della Tuscia, Viterbo, Italy,
Dipartimento di Scienze e Tecnologie Chimiche, UniVersita` Tor Vergata, Roma 00100, Italy,
Istituto di Biologia e Patologia Molecolari, CNR, Roma, Italy, and Dipartimento di Genetica e
Biologia Molecolari, UniVersita` “Sapienza”, Roma 00185, Italy
Received June 23, 2008; E-mail: saladino@unitus.it; Ernesto.dimauro@uniroma1.it
Abstract: We describe the one-pot synthesis of a large panel of nucleic bases and related compounds
from formamide in the presence of iron sulfur and iron-copper sulfur minerals as catalysts. The major
products observed are purine, 1H-pyrimidinone, isocytosine, adenine, 2-aminopurine, carbodiimide, urea,
and oxalic acid. Isocytosine and 2-aminopurine may recognize natural nucleobases by Watson-Crick and
reverse Watson-Crick interactions, thus suggesting novel scenarios for the origin of primordial nucleic
acids. Since the major problem in the origin of informational polymers is the instability of their precursors,
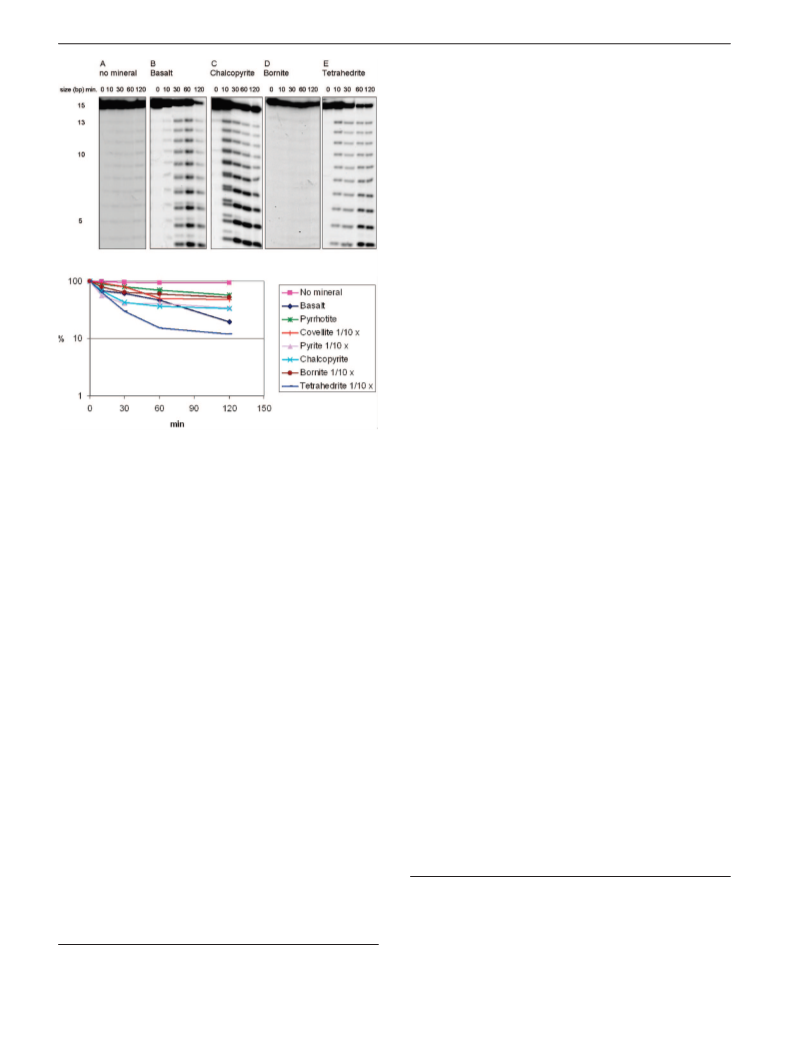
we also investigate the effects of iron sulfur and iron-copper sulfur minerals on the stability of
ribooligonucleotides in formamide and in water. All of the iron sulfur and iron-copper sulfur minerals
stimulated degradation of RNA. The relevance of these findings with respect to the origin of informational
polymers is discussed.
Introduction
presence of different metal oxides and minerals in the reaction
mixture,7 thus modeling the environment of the primitive Earth.
Plausible scenarios for the origin of life entail the robust
prebiotic synthesis of informational polymers by condensation
of simple chemical precursors.1 Among the chemical precursors
taken into consideration, the two related compounds hydrogen
cyanide (HCN) and formamide (NH2COH, 1) have been the
subject of thorough analyses.1,2 These compounds are easily
converted by hydrolysis or dehydration. The attention given
these two compounds is mainly due to their ability to synthesize
nucleic bases under relatively mild experimental conditions
coherent with those existing on the primitive Earth.3 It is
noteworthy that formamide is the only chemical precursor able
to synthesize at the same time, in addition to some amino acid
derivatives, both purine and pyrimidine nucleic bases.4 In
agreement with the hypotheses by Bernal5 and Cairns-Smith,6
the prebiotic chemistry of formamide is finely tuned by the
These compounds can act as catalysts for condensation pro-
cesses, enhancing the concentration of the reactant and preserv-
ing newly formed biomolecules from chemical and photochemi-
cal degradation.8 Moreover, minerals can catalyze the in situ
decomposition of formamide to other chemicals that are
potentially useful for the construction of both purine and
pyrimidine scaffolds, such as ammonia and HCN.4 The effect
of metal oxides and minerals on the chemistry of formamide
has been studied in detail to better correlate the selectivity of
the prebiotic syntheses to the chemical properties and elemental
composition of the catalyst.2 Metal oxides characterized by
photoreactivity, such as titanium dioxide (TiO2), are efficient
catalysts for the synthesis of different nucleobases from for-
mamide, accompanied by release of formaldehyde.9 This
aldehyde, which is the main precursor of sugars through a
formose-like condensation,10 provides, in addition to formamide,
the optimal substrates for the first reported one-pot prebiotic
synthesis of nucleoside derivatives. The possibility of synthesiz-
ing the sugar moiety directly on the formylated nucleobases by
successive addition of one-carbon fragments opens novel
† Universita` della Tuscia.
‡ Universita` Tor Vergata.
§ Istituto di Biologia e Patologia Molecolari.
| Universita` “Sapienza”.
(1) Prebiotic Chemistry; Walde, P., Ed.; Topics in Current Chemistry,
Vol. 259; Springer-Verlag: Berlin Heidelberg, 2005.
(2) For recent reviews, see: (a) Saladino, R.; Crestini, C.; Ciciriello, F.;
Costanzo, G.; Negri, R.; Di Mauro, E. In Astrobiology: Future
PerspectiVes; Ehrenfreund, P., Ed.; Kluwer: Dordrecht, The Nether-
lands, 2004; pp 393-413. (b) Saladino, R.; Crestini, C.; Ciciriello,
F.; Costanzo, G.; Di Mauro, E. Origins Life EVol. Biosphere 2006,
36, 523–531.
(6) Cairns-Smith, G. In Possibile Role for Minerals in Early Organisms;
Tran Tharh Van, J., Mounolou, J. C., Schneider, J., McKay, C., Eds.;
Editions Frontie`res: Gif-sur-Yvette, France, 1992; pp 119-132.
(7) Saladino, R.; Crestini, C.; Costanzo, G.; Negri, R.; Di Mauro, E.
Bioorg. Med. Chem. 2001, 9, 1249–1253.
(3) Saladino, R.; Crestini, C.; Costanzo, G.; Di Mauro E. In Prebiotic
Chemistry; Walde, P., Ed.; Topics in Current Chemistry, Vol. 259;
Springer-Verlag: Berlin Heidelberg, 2005; pp 29-68.
(4) Saladino, R.; Crestini, C.; Ciambecchini, U.; Ciciriello, F.; Costanzo,
G.; Di Mauro, E. ChemBioChem 2004, 5, 1471–1478.
(5) Bernal, J. D. The Physical Basis of Life; Routledge and Kegen Paul:
London, 1951.
(8) Saladino, R.; Crestini, C.; Costanzo, G.; Di Mauro, E. Curr Org. Chem.
2004, 8, 1425–1443.
(9) Saladino, R.; Ciambecchini, U.; Crestini, C.; Costanzo, G.; Negri, R.;
Di Mauro, E. ChemBioChem 2003, 4, 514–521.
(10) (a) For a basic reference, see: Butlerow, A. Ann. Phys. 1861, 120,
296. (b) For a more recent reference, see: Decker, P.; Schweer, H.;
Pohlman, R. J. Chromatogr. 1982, 244, 281–291.
9
15512 J. AM. CHEM. SOC. 2008, 130, 15512–15518
10.1021/ja804782e CCC: $40.75
2008 American Chemical Society

 Saladino, Raffaele
Saladino, Raffaele
 Neri, Veronica
Neri, Veronica
 Crestini, Claudia
Crestini, Claudia
 Costanzo, Giovanna
Costanzo, Giovanna
 Graciotti, Michele
Graciotti, Michele
 Di Mauro, Ernesto
Di Mauro, Ernesto