Full Papers
if the catalyst was recalcined at 573 K and reused for the sev-
enth run.
with an aqueous solution containing two metal precursors, namely,
Ni(NO ) ·6H O (98%, Alfa Aesar) and Fe(NO ) ·9H O (98%, Sigma–
3 2
2
3 3
2
Aldrich). After impregnation and solvent removal by evaporation,
the produced solids were dried overnight at 383 K. All the dried
samples were calcined at 573 K in air for 4 h. Before the reaction,
the samples were reduced by 5% H /95% N at 673 K for 4 h. The
The Ni content in the aqueous phase was checked after
each cycle by using inductively coupled plasma atomic emis-
sion spectrometry. The results indicate that the amount of Ni
and Fe species leached is lower than the detectable level
2
2
bimetallic catalyst was labeled as Ni ÀFe /CNTs, in which x and y
x
y
(
Table S4). The XRD patterns of the as-reduced Ni ÀFe /CNTs
2
1
were the atomic ratios of Ni and Fe, respectively.
and the used catalyst (Figure S6) demonstrate that the intensi-
ty of the metallic Ni diffraction peaks of the catalysts obtained
after the reaction is only slightly higher than that of the peaks
of the as-reduced catalysts. Therefore, the aggregation of Ni
nanoparticles during the reaction is minimal. The TEM image
gave an intuitive Ni particle size of 7.2 nm after the reaction,
which was slightly larger than that of the fresh catalyst. The
thermogravimetric profiles provide an important information
on the reactivity (Figure S7). Compared to the fresh Ni ÀFe /
Characterization
The N adsorption–desorption isotherms were measured at 77.3 K
2
with a Micromeritics TriStar II 3020 porosimetry analyzer. Before ad-
sorption, the sample was degasified at 473 K for 2 h. The specific
surface area was obtained by using the BET method at P/P =0.05–
0
0.3. By using the Barret–Joyner–Halenda method, the average pore
diameter and pore size distribution were evaluated from the pore
size distribution curve, which was obtained by analyzing the de-
sorption isotherm branch. The total pore volume of the samples
depended on the amount of N vapor adsorbed at P/P ꢀ0.99.
The Ni and Fe contents were determined with a Bruker S8 TIGER X-
ray fluorescence spectrometer. The samples were prepared by
mixing the catalyst (0.20 g) and boric acid (0.80 g) and compress-
ing into tablets (diameter=36 mm; thickness=2 mm).
2
1
CNT catalyst, the sudden weight loss of the used catalyst from
00 to 800 K is mainly due to the combustion of the organic
6
compound, for example, the small amount of humin derived
from the polymerization of HMF. Therefore, deactivity may be
due to the slow formation of the polymer, which covered the
activity center of the catalyst.
2
0
Conclusions
H -TPR was performed with a Micromeritics AutoChem II 2920
2
chemisorption analyzer. Samples (0.10 g) were weighed and flush-
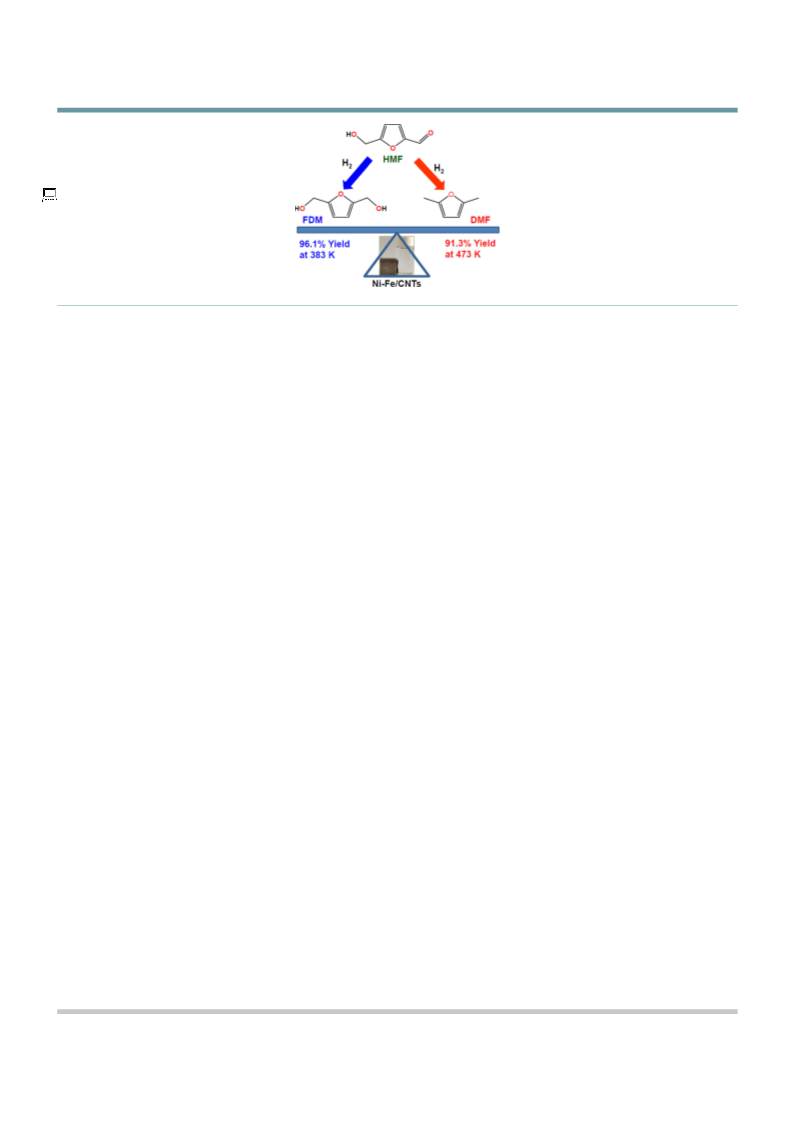
We developed an inexpensive, effective, nonprecious carbon
nanotube-supported bimetallic NiÀFe (NiÀFe/CNT) catalyst for
the selective hydrogenation and hydrogenolysis of 5-hydroxy-
methylfurfural with molecular hydrogen as a hydrogen donor.
The catalyst with an appropriate Ni/Fe atomic ratio showed an
excellent selectivity to 2,5-furandimethanol or 2,5-dimethylfur-
an at 383 and 473 K, respectively. Contrary to the monometal-
lic Ni/CNT catalyst, a negligible amount of the overhydrogena-
tion product of the furan ring (2,5-dimethyltetrahydrofuran)
was observed. The XRD and TEM results indicated that NiÀFe
alloy species were formed on the surface of CNTs. Detailed re-
search shows that NiÀFe alloy species are beneficial to the
cleavage of the CÀO bond at a higher reaction temperature.
The catalyst can be easily separated with a magnet and reused
several times. The high activity and stability of the Ni ÀFe /CNT
3
À1
ed with high purity He (flow rate: 30 cm min ) at 473 K for 1 h
and then cooled to 323 K. Subsequently, 5% H /95% Ar was
2
flowed through the samples while the temperature was increased
À1
from 323 to 1173 K (heating rate: 10 Kmin ). The thermal conduc-
tivity detector signal automatically recorded the H consumption.
2
The XRD pattern was recorded with a Philips PANalytical X’pert
PRO diffractometer equipped with a graphite monochromator and
a CuK radiation source (40 kV and 30 mA), scanning at 2q=20 to
a
908. The results were compared with the standard powder XRD
data (in the JCPDS database).
Static CO chemisorption was measured with a Micromeritics ASAP
2
020 (M+C) apparatus. The catalysts were treated by a flow of 5%
H /95% N2 with the increase in temperature from 303 to 673 K
2
À1
(heating rate: 2 Kmin ) and then cooled to RT. The as-prepared
catalysts were then soaked in H at 673 K for 15 min, evacuated for
2
2
1
6
0 min, and cooled to 308 K. After pretreatment, the catalysts were
catalyst under the given reaction conditions suggest that the
catalyst is feasible and a promising material for 2,5-dimethyl-
furan production through the selective hydrogenation and hy-
drogenolysis of 5-hydroxymethylfurfural.
analyzed by pumping CO gas at 308 K. The initial isotherm was
then measured. A 30 min evacuation at 308 K was then performed
to remove the reversibly adsorbed H2 on the catalyst surface
before performing the measurement for a second isotherm under
conditions similar to those of the first isotherm. The difference be-
tween the first and second isotherms reflected the quantity of irre-
Experimental Section
versibly chemisorbed H on the catalyst sample.
2
Catalyst preparation
The TEM images were obtained with a Tecnai F20 electron micro-
scope operated at an accelerating voltage of 200 kV. The powder
was dispersed in ethanol by ultrasonication for 10 min, and then
drops of suspensions were deposited on a copper grid coated with
carbon. The statistical mean diameter of the nanoparticles was
measured by counting at least 200 particles for each catalyst.
All the reagents were used as received without further purification.
CNTs (diameter=10–20 nm) with a purity of 95% were purchased
from Shenzhen Nanotech Port Co., Ltd. First, pristine CNTs were
purified and functionalized in concd HNO (68 wt%) at 353 K for
3
1
6 h and heated to reflux to remove amorphous carbon and the
remaining catalyst residues. Then, the treated CNTs were filtered,
extensively washed with deionized water until the pH of the rins-
ing water became neutral, and dried at 373 K overnight. Supported
bimetallic NiÀFe catalysts were prepared through coimpregnation
XPS was performed with a PHI Quantum 2000 Scanning ESCA Mi-
croprobe with an AlK radiation source (hn=1486.6 eV). The bind-
a
ing energy was calibrated from that of C1s (284.6 eV). The reduced
samples were attached to the sample holder with conductive
&
ChemCatChem 0000, 00, 0 – 0
6
ꢀ 0000 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
ÝÝ These are not the final page numbers!

 Yu, Lili
Yu, Lili
 He, Le
He, Le
 Chen, Jin
Chen, Jin
 Zheng, Jianwei
Zheng, Jianwei
 Ye, Linmin
Ye, Linmin
 Lin, Haiqiang
Lin, Haiqiang
 Yuan, Youzhu
Yuan, Youzhu