ACS Combinatorial Science
Research Article
that for both the 400 and 100 mg resin quantities, increases in
flow rate beyond 5 mL min appear to reduce reaction yields.
Multiple Coupling Cycles Using a Single Resin Batch.
In theory with the utilization of 10-mM equivalents of resin-4,
one should be able to prepare at least five discrete batches of
amide on a 1 mmol scale or ten discrete amides on a 0.5 mmol
scale and so forth. To explore the viability of such a strategy,
the resin column was initially loaded with 800 mg of resin-4
methylbenzoic acid. As detailed in Tables 3 and 4, the
sequential injection protocol proved to be a viable strategy for
analog library synthesis. With regards to substrate amenability,
it appears that carboxylate electron density perturbation
imparts no adverse impacts on coupling efficiency as similar
conversion observed with the employment of 4-nitro- or 4-
methylbenzoic acid. Concerning amine amenability, for
primary amine-based analogs, the desired amide products
were afforded in >95% conversion. Furthermore, coupling with
aniline-based amines afforded a series of aromatic derivatives in
>90% conversion. However, while the protocol proved to be
both rapid and convenient, it does not appear to circumvent
the inherent coupling limitations of solution-phase carbodii-
mide couplings. For example, the strategy proved ineffectual
for the coupling of both hindered and deactivated amines (e.g.,
entries 2 and 3, Table 3 and entries 2 and 3, Table 4).
Thus, in summary, using the injection-based flow protocol,
two discrete series of amides were synthesized from a relatively
diverse pool of achiral amines. However, given the propensity
of additive-free carbodiimide based couplings to induce
epimerization, it was assumed that the practicability of the
flow protocol would not extend beyond achiral substrates.
Nonetheless, to the best of our knowledge, no studies
examining the effects that PS-carbodiimide-mediated imparts
on epimerization have been reported. Consequently, to gain
insight into the propensity for immobilized carbodiimide
reagents to induce epimerization and to ascertain whether the
inclusion of additives or solvent choice could influence
diastereomeric ratios, our focus turned to amino acid
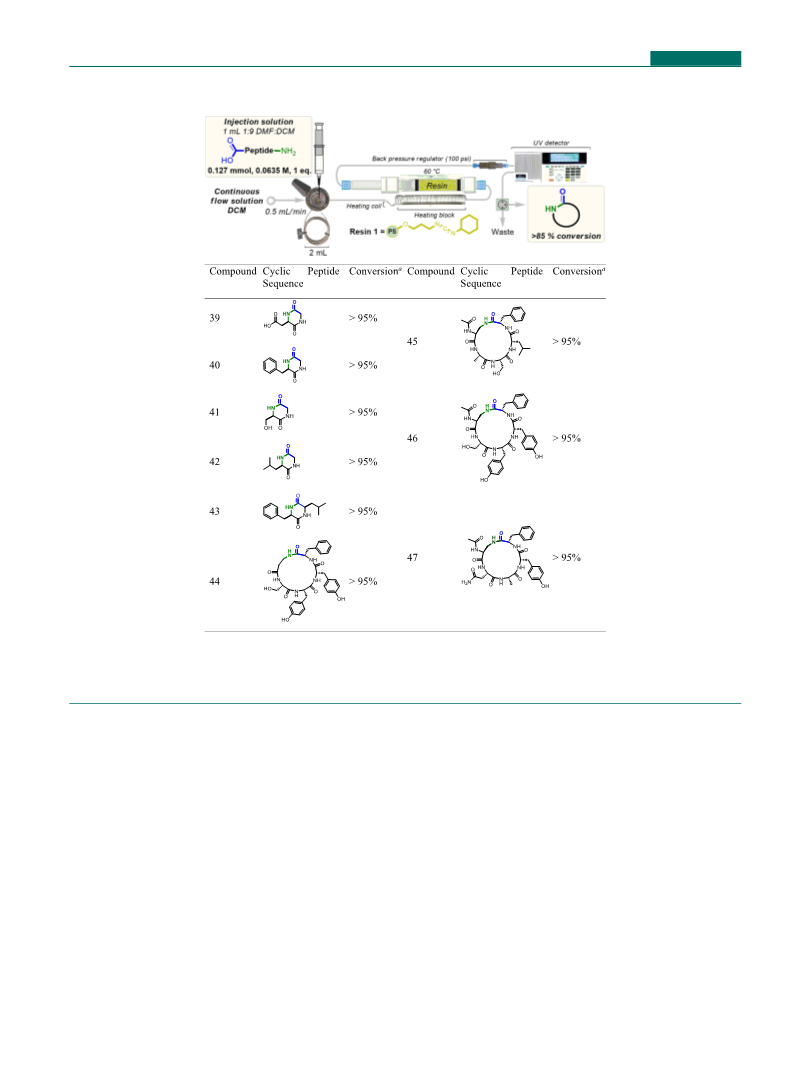
couplings.
(
1.072 mmol, 4.0 equiv), through which was successively pass
six 1 mL injections, each containing benzoic acid (10) (0.27
mmol, 0.27 M, 1.0 equiv) and benzylamine (8) (0.27 mmol,
To initially ensure the flow protocol was amenable to
coupling protected amino acid residues, a preliminary trial was
performed, which employed Fmoc-Leu-OH (32) and the ethyl
ester protected lysine HCl derivative (33). Here the desired
dipeptide 34 was afforded in quantitative yield with no
epimerization observed (Figure 9). Further, this result
demonstrated HCl-amine salts could be coupled without the
inclusion of base. While this is currently under further
examination, at this point, we suspect that the excess
carbodiimide may sequester the HCl.
Figure 8. (a) Condensation reaction between benzylamine 8 and
benzoic acid 10 using PS-DCC to afford amide 11. (b) HPLC
chromatographs of column eluents obtained from flowing successive
injections of 1 mL solutions containing benzylamine and benzoic acid
through a single batch of resin-4.
equated with 0.25 mol equiv relative to the resin-4, it was
proposed that quantitative conversion to the amide 11 should
be afforded from the first two injections and that this would
incrementally decrease across subsequent injections. Indeed, as
detailed in Figure 8, it was apparent that there was a certain
amount of credence to the supposition mentioned above. For
example, analysis of the column eluent emerging from
injections one and two indicated that 11 was afforded in
quantitative conversion (tR = 8.5 min). With regards to
injections three and four, conversion to 11 reduced to 80% and
With this result, our attention then turned to the coupling of
residues that are vulnerable to direct epimerization, such as
48−50
serine.
Hence, as detailed in Table 5, Fmoc-(t-Bu)Ser-OH
(35) and the ethyl ester protected lysine derivative (33) served
as coupling partners for the initial epimerization assessments.
To assess the epimerization vulnerability of this coupling, two
solution-phase batch couplings were initially performed. Both
of these couplings were conducted in a 10% DMF DCM using
2 equiv of solution-phase DCC under ambient conditions. For
the initial additive-free experiment, the coupling of 33 and 35
was effected with an overall conversion of ∼80% and a
diastereomeric ratio (d.r.) of 19:1 (Figure 10). For the second
reaction, which incorporated a catalytic quantity of DMAP,
near-complete chiral inversion (i.e., d.r. 1:15.7, Figure 10) was
observed.
Upon establishing vulnerability to stereomutation, focused
switched to assessing the coupling of 33 to 35 under flow
conditions. As detailed in Table 5, across the 14 trials, which
examined additive and solvents variation, no epimerization was
observed (Table 5). The additive-free approach also afforded
the histidine-based dipeptide 37 and the cysteine-based
dipeptide 38 with >80% conversions, with no evidence of
stereomutation (Table 6). Hence, it appears the utilization of
6
0% respectively, whereas, for the fifth and six doses, trace and
no observable quantities of 11 were observed, respectively.
Hence, to remain viable, the above trials suggest that at least
twice the molar equivalences of resin-4 are required to effect
quantitative amide formation. While theory dictates that 1-
equivalent should be sufficient, it is suspected that the
relatively high resin loading (1.27 mmol/g) may impose steric
hindrance. Nonetheless, under the proviso that carbodiimide
excess is maintained, successive couplings could be cleanly
effected using a single batch of resin.
Assessing Analogue Library Synthesis and Substrate
Amenability. With an eye to compound library synthesis, the
subsequent reaction trials explored synthesizing a small library
of amide derivatives based upon either 4-nitro- or 4-
G
ACS Comb. Sci. XXXX, XXX, XXX−XXX

 Aldrich-Wright, Janice R.
Aldrich-Wright, Janice R.
 Dankers, Christian
Dankers, Christian
 Gordon, Christopher P.
Gordon, Christopher P.
 Harman, David G.
Harman, David G.
 Nguyen, Thanh V.
Nguyen, Thanh V.
 Tadros, Joseph
Tadros, Joseph