Table 1. Composition of Reaction Crudes in the Cyclization Step at 3 ha
temp
(˚C)
Pro-NCA
Pro N-carbamoyl
chloride (%)
Pro-Pro DKP
(%)
Et3Nc
(%)
entry
amino acid
base
equiv
(%)
b
1
2
3
4
5
6
7
8
9
H-Glu(OBzl)-OH
H-Pro-OH
-
-
Et3N
Et3N
Et3N
TBD-PS
DMAP-PS
PIP-PS
DIEA-PS
MPH-PS
DEAM-PS
-
-
1
1
1
1
1
1
1
1
1
50
50
-25
0
25
25
25
25
25
25
25
100
3
0
97
<1
38
54
86
86
27
25
30
29
0
0
-
-
<1
2, 7
6, 6
-
-
-
-
-
b
H-Pro-OH
H-Pro-OH
H-Pro-OH
H-Pro-OH
H-Pro-OH
H-Pro-OH
H-Pro-OH
H-Pro-OH
H-Pro-OH
59
27
41
13
13
70
72
68
70
40, 0
35
5
<1
<1
3
3
2
<1
1
1
0
1
-
a
1
b
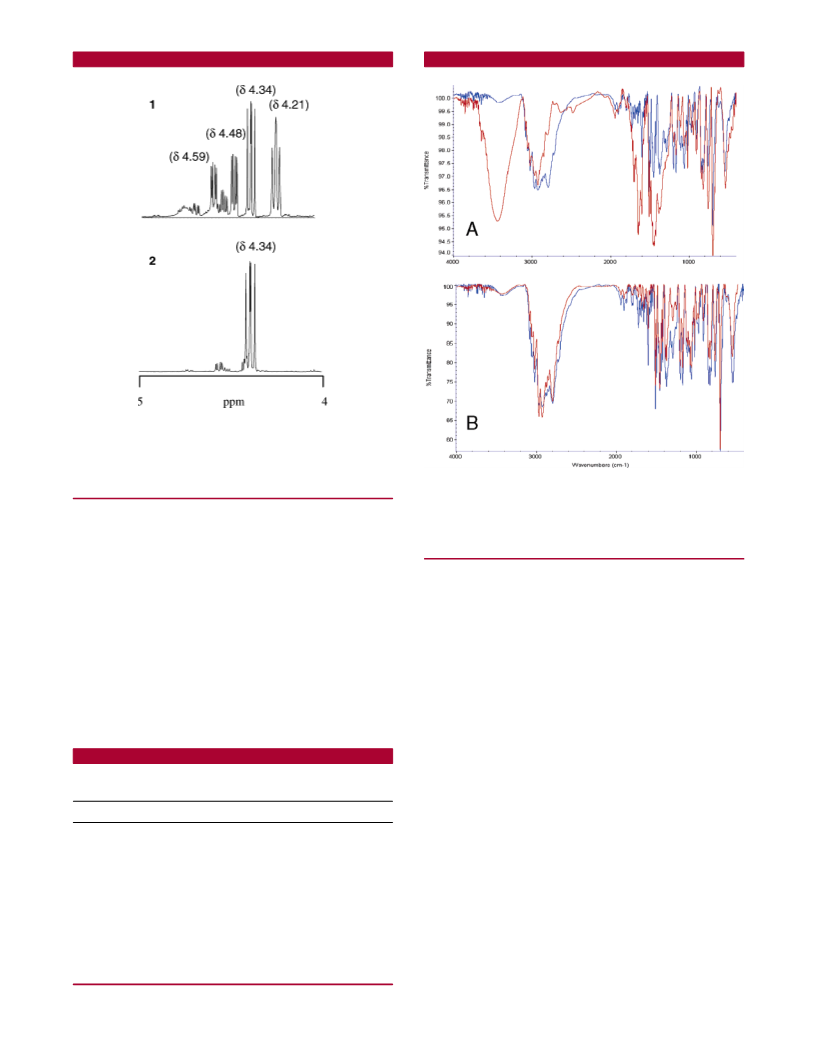
Data have been calculated (average of triplicates) by integration of proton signals in H NMR spectra (see Figure 2). Typical synthesis only with
c
triphosgene. Calculated with respect to the initial amount of Et3N.
side chain yielding a cyclic secondary amine (pirrolidine)
and showing some conformational restrictions. These features
probably underlie the poor synthetic yields obtained using
currently available methods for R-amino acid N-carboxy-
anhydride formation.7 Generally speaking, N-carboxy-
anhydrides (NCAs) are obtained by treatment of the corre-
sponding amino acid with phosgene, the so-called Fuchs
crystallized proline NCA product. In the present article, we
describe the preparation of Pro-NCA in high yields and
1
0
11
purities using solid triphosgene and polymer-bound
1
2
bases (Figure 1).
8
method. In the case of proline, the N-carbamoyl intermediate
does not cyclize spontaneously as it takes place with other
amino acids, and the use of a nonnucleophilic base, typically
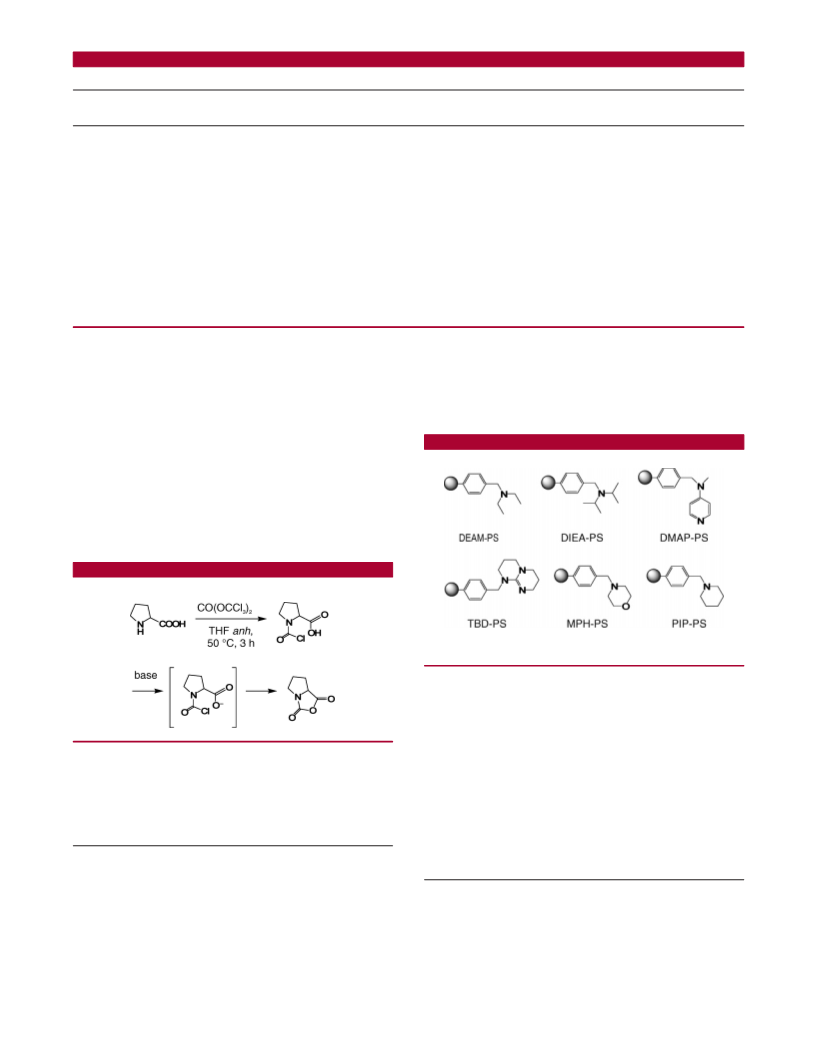
a tertiary amine, is required for the cyclization to the
N-carboxyanhydride (Scheme 1).
Scheme 1. Synthesis of Proline NCA
Figure 1. Polymer-bound tertiary amines.
Six polymer-supported bases were chosen to catalyze NCA
cyclization (Figure 1). The percentages of different species
present in their action crude after 3 h, namely, Pro-NCA,
N-carbamoyl chloride, and the Pro-Pro diketopiperazine
byproduct (Pro-Pro DKP), are summarized in Table 1. For
comparison purposes, the yield of formation of the N-
carboxyanhydride of the benzyl-protected glutamic acid,
Glu(OBzl)-NCA, and the yield of formation of Pro-NCA,
both in the absence of a base, are shown in entries 1 and 2
of Table 1. As expected, the yield of Glu(OBzl)-NCA is
quantitative whereas the yield of Pro-NCA is practically nil
Procedures described in the literature require slow addition
of solutions of phosgene at low temperature and the use of
tertiary amines, such as triethylamine,7,9 that are difficult to
eliminate and appear in variable amounts in the final
(
6) (a) Woodward, R. B.; Scramm, C. H. J. Am. Chem. Soc. 1947, 69,
1
8
8
5
551. (b) Brown, C. J.; Coleman, D.; Farthing, A. C. Nature 1949, 163,
34. (c) Fasman, G. D.; Idelson, M.; Blout, E. R. J. Am. Chem. Soc. 1961,
3, 709. (d) Deming, T. J.; Curtin, S. A. J. Am. Chem. Soc. 2000, 122,
710. (e) Dimitrov, I.; Schlaad, H. Chem. Commun. 2003, 23, 2944.
(10) (a) Petri, E. M.; Staverman, A. J. Recl. TraV. Chim. 1952, 71, 385.
(b) Blout, E. R.; Karlson, R. H.; Doty, P.; Hargitay, B. J. Am. Chem. Soc.
1954, 76, 4492.
(11) Daly, W. H.; Poche, D. Tetrahedron Lett. 1988, 29, 5859
(12) (a) Booth, R. J.; Hodges, J. C. J. Am. Chem. Soc. 1997, 119, 4882.
(b) Xu, W.; Mohan, R.; Morrisey, M. M. Tetrahedron Lett. 1997, 42, 7337.
(c) Hulme, C.; Ma, L.; Romano, J.; Morrisette, M. Tetrahedron Lett. 1999,
40, 7925. (d) Yadav-Bhatnagar, N.; Dejonqu e` res, N.; Mauger, J. J. Comb.
Chem. 2002, 4, 48.
(
7) Iizuka, Y.; Uchida, C.; Wakamatsu, K.; Oya, M. Bull. Chem. Soc.
Jpn. 1993, 66, 1269.
8) (a) Fuchs, F. Berichte 1922, 55, 189. (b) Farthing, A. C.; Reynolds,
R. J. W. Nature 1950, 22, 647.
9) (a) Wider, R.; Mobashery, S. J. Org. Chem. 1992, 57, 2755. (b)
(
(
Xavier, L. C.; Mohan, J. J.; Mathre, D. J.; Thompson, A. S.; Carroll, J. D.;
Corley, E. G.; Desmond, R. Org. Synth. 1997, 74, 50.
5386
Org. Lett., Vol. 8, No. 23, 2006

 Gulin, Oscar P.
Gulin, Oscar P.
 Rabanal, Francesc
Rabanal, Francesc
 Giralt, Ernest
Giralt, Ernest