1616
S. Syukri et al. / Tetrahedron Letters 48 (2007) 1613–1617
decreasing the catalytic activity during repeated runs. A
noteworthy finding of this work is that the somewhat lar-
ger size (steric bulk) of a salen ligand seems to lead to a
slightly better recyclability, beside its somewhat higher
selectivity.22–24 The lower degree of leaching compared
to mesoporous carrier materials is an important advan-
tage of the polymeric carrier materials applied in this
work.
2.3. General procedure for aldehyde olefination in THF
In an oven dried Schlenk tube 1 mol % of the polymer
grafted catalyst (based on the Ru-loading), aldehyde
(0.25 mmol), PPh3 (0.30 mmol) and 3.5 ml of THF were
placed and heated to 60 °C in an oil bath. EDA
(0.30 mmol), dissolved in 1.5 ml of THF, was added
and then the mixture was kept for the times given in
Table 1. The suspension was then cooled to room temper-
ature and the solution was separated from the catalyst
by filtration. Afterwards, the residue was concentrated
and chromatographed over a silica gel column with
n-hexane/ethyl acetate (20:1–8:1) affording the desired
olefins.
Two types of Ru(II) salen complexes are successfully
grafted on the poly(4-vinylpyridine). The obtained het-
erogeneous catalysts are found to be of lower activity
for the olefination of various aldehydes than the homo-
geneous compounds. The better recyclability of the
polymer supported material, when compared to catalyst
immobilized on mesoporous material is a noteworthy
advantage, however. The ruthenium(II) compound li-
gated by a larger salen ligand shows a better recyclabil-
ity and selectivity than that with the smaller ligand. An
aim for future work will be the generation of a covalent
surface-metal bond, possibly with an organometallic lin-
ker, which might further reduce leaching.
Acknowledgements
S.S. is grateful to the TPSDP (Technological Profes-
sional Skills and Development Project) supported by
Asian Development Bank Loan No. 1792-INO for
financial support. W.S. thanks the Alexander von Hum-
boldt foundation for financial support. The authors also
thank Dr. Gabriele Raudaschl-Sieber for experimental
support. The Fonds der Chemischen Industrie and the
Leonhard-Lorenz-Stiftung are acknowledged for finan-
cial support.
2. Experimental
2.1. Synthetic procedure
P4VP is used as provided by Aldrich without purifica-
tion for the synthesis of the grafted materials. All the
grafting procedures are carried out under an oxygen-
and water free argon atmosphere using standard
Schlenk techniques if not indicated otherwise. Dichloro-
methane (DCM) is dried over calcium hydride and kept
References and notes
1. Cavallo, L.; Jacobsen, H. J. Org. Chem. 2003, 68, 6202.
2. Venkataramanan, N. S.; Kuppuraj, G.; Rajagopal, S.
Coord. Chem. Rev. 2005, 294, 1250–1251.
3. Canali, L.; Sherrington, D. C. Chem. Soc. Rev. 1999, 28,
85–93.
˚
over 4 A molecular sieves. Tetrahydrofuran (THF) is
˚
dried over sodium and also kept over 4 A molecular
4. Smith, K.; Liu, C. H. Chem. Commun. 2002, 886–887.
sieves.
5. Sun, W.; Yu, B.; Kuhn, F. E. Tetrahedron Lett. 2006, 47,
¨
1993.
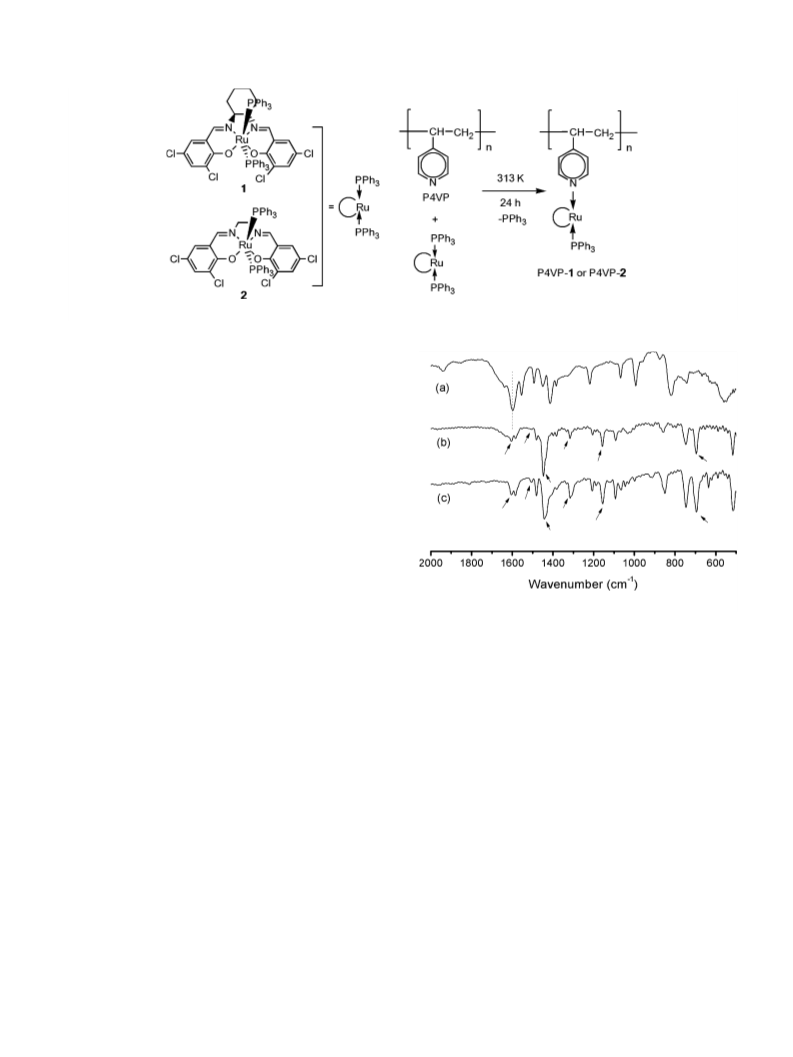
Complex 1 and complex 2 were synthesized as described
earlier.13,14 Grafting experiments (Scheme 3) were car-
ried out using standard Schlenk techniques under an
argon atmosphere with the following procedure: 1 g of
polymer [P4VP, 2% cross-linker] is dried under vacuum
at room temperature and then 0.0814 g (0.075 mmol) of
complex 1 or 0.0775 g (0.075 mmol) of complex 2 are
introduced in the presence of 30 ml acetonitrile as sol-
vent. The obtained mixture is stirred at 323 K for 24 h
the resulting materials are washed several times with
CH2Cl2 and dried at room temperature under vacuum.
The grafted polymer materials are designated as P4VP-
1 and P4VP-2.
6. Lloret, F.; Julve, M.; Mollar, M.; Castro, I.; Latorre, J.;
Faus, J.; Solans, X.; Morgenstern-Badaran, I. J. Chem.
Soc., Dalton Trans. 1989, 729.
7. Vos, D. E.; Knops-Gerits, P.; Parton, R. F.; Weckhuysen,
B. M.; Jakobs, P. A.; Schoonheydt, R. A. J. Ind. Phenom.
Mol. Recognit. Chem. 1995, 21, 185.
8. Paul, P. P. Prog. Inorg. Chem. 1999, 48, 457.
9. Balkus, K., Jr.; Eissa, M.; Levedo, R. J. Am. Chem. Soc.
1995, 117, 10753.
10. Syukri, S.; Sakthivel, A.; Sun, W.; Kuhn, F. E. Catal.
¨
Lett., in press.
11. Kim, J. H.; Kim, J. W.; Shokouhimehr, M.; Lee, Y. S.
J. Org. Chem. 2005, 70, 6715.
12. Schwarz, J.; Bo¨hm, V. P. W.; Gardiner, M. G.; Grosche,
M.; Herrmann, W. A.; Hieringer, W.; Raudaschl-Sieber,
G. Chem.-Eur. J. 2000, 6, 1773.
2.2. Characterization methods
13. Liang, J. L.; Yu, X. Q.; Che, C. M. Chem. Commun. 2002,
124.
14. Murray, K. S.; Bergen, A. M.; West, B. O. Aust. J. Chem.
1978, 31, 203.
15. Santana, A. L.; Noda, L. K.; Pires, A. T. N.; Bertolino, J.
R. P4VPm. Test 2004, 23, 839.
16. Belfiore, L. A.; McCurdie, M. P.; Das, P. K. P4Vpmer
2001, 21, 9995.
IR spectra are measured with a Unican Mattson Mod
7000 FTIR spectrometer using KBr pellets. Microanaly-
ses are performed at the Mikroanalytisches Labor of the
Technische Universita¨t Munchen (U. Ammari and co-
¨
workers). Thermogravimetry, combined with mass spec-
troscopy (TG–MS) is conducted with a Netzsch TG209
system; typically about 10 mg of a sample were heated
from 323 to 1273 K at a heating rate of 10 K minꢀ1
under an argon atmosphere.
17. Giacomelli, C.; Giacomelli, F. C.; Santana, A. L.;
Schmidt, V.; Pires, A. T. N.; Bertolino, J. R.; Spinelli,
A. J. Braz. Chem. Soc. 2004, 15, 823.

 Syukri, Syukri
Syukri, Syukri
 Sun, Wei
Sun, Wei
 Kühn, Fritz E.
Kühn, Fritz E.