N. Iranpoor et al. / Tetrahedron Letters 46 (2005) 7963–7966
7965
References and notes
SiO2
O
+
O
)
PCl3-n
CR
EtO
n
1. For reviews, see: (a) Rana, S. S.; Barlow, J. J.; Matta, K.
L.. Tetrahedron Lett. 1982, 22, 5007–5010; (b) Green, T.
W.; Wuts, P. G. M. Protecting Groups in Organic
Synthesis, 3rd ed.; Wiley: New York, 1999; (c) Kocienski,
P. J. Protecting Groups; Thieme: Stuttgart, 1994.
2. (a) Kawabata, T.; Mizugaki, T.; Ebitani, K.; Kaneda, K.
Tetrahedron Lett. 2003, 44, 9205–9208; (b) Sharghi, H.;
Hosseini, S. M. Tetrahedron 2003, 3627–3633; (c) Zhu, H.
P.; Yang, F.; Tang, J.; He, M. Y. Green Chem. 2003, 5, 38–
39; (d) Shirini, F.; Zolfigol, M. A.; Mohammadi, K.
Phosphorus, Sulfur, Silicon Relat. Elem. 2003, 178, 1617–
1621; (e) Palaniappan, S.; Ram, M. S. Green Chem. 2002,
4, 53–55; (f) Ishihara, K.; Nakayama, M.; Ohara, S.;
Yamamoto, H. Tetrahedron 2002, 58, 8179–8188; (g)
Ishihara, K.; Nakayama, M.; Ohara, S.; Yamamoto, H.
Synlett 2001, 1117–1120; (h) Ramalinga, K.; Vijaya-
lakshmi, P.; Kaimal, T. N. B. Tetrahedron Lett. 2002,
43, 879–882; (i) Wakasugi, K.; Misaki, T.; Yamada, K.;
Tanabe, Y. Tetrahedron Lett. 2000, 41, 5249–5252; (j) Das,
B.; Venkataiah, B.; Madhusudhan, P. Synlett 2000, 59–60;
(k) Iranpoor, N.; Shekarriz, M. Bull. Chem. Soc. Jpn.
1999, 72, 455–458; (l) Saravanan, P.; Singh, V. K.
Tetrahedron Lett. 1999, 40, 2611–2614.
3. For a review see: (a) Grasa, G. A.; Singh, R.; Nolan, S. P.
Synthesis 2004, 971–985; (b) Bartoli, G.; Bosco, M.;
Dalpozzo, R.; Marcantoni, E.; Massaccesi, M.; Sambri, L.
Eur. J. Org. Chem. 2003, 4611–4617; (c) Habibi, M. H.;
Tangestaninejad, S.; Mirkhani, V.; Yadollahi, B. Monatsh.
Chem. 2002, 133, 323–327; (d) Habibi, M.; Tangestanine-
jad, S.; Mirkhani, V.; Yadollahi, B. Tetrahedron 2001, 57,
8333–8337; (e) Iranpoor, N.; Firouzabadi, H.; Zolfigol,
M. A. Synth. Commun. 1998, 28, 1923–1934.
4. Ishihara, K.; Kubota, M.; Yamamoto, H. Synlett 1996,
265–266.
5. Izumi, J.; Shiina, I.; Mukaiyama, T. Chem. Lett. 1995,
141–142.
EtCl
R'
XH
SiO2
O
O
C
R
O
P
Cl2-n
O
+
(SiO2)nPCl2-nOH R'XCR
R= -CH3, -H
R'=1o, 2o, benzylic, allylic
X= O, N
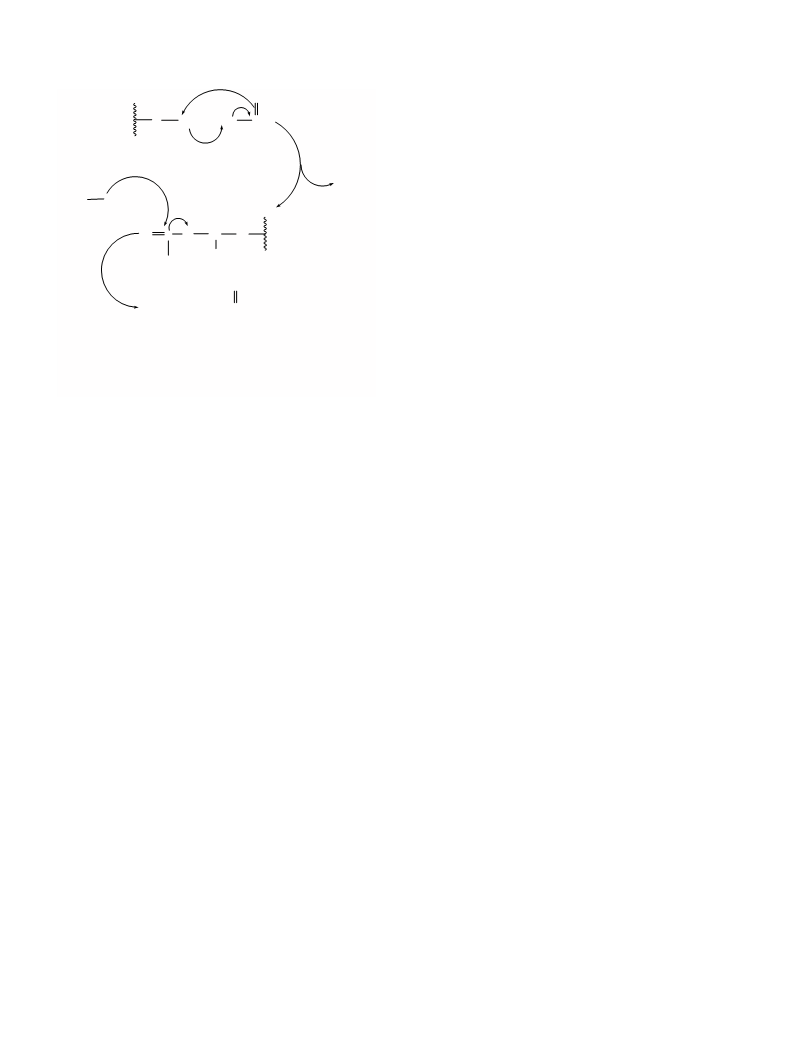
Figure 2. The proposed mechanism for formylation and acetylation of
alcohols and amines.
200 °C, lit.23 bp 201 °C. The product was found to be
highly pure by GC analysis.
Typical procedure for acetylation of 2-phenylethanol: 2-
Phenylethanol (1.0 mmol, 0.12 g) was added to 1.5 g of
Silphos [PCl3Àn(SiO2)n] in refluxing ethyl acetate
(5 mL) and the mixture was stirred. The progress of
the reaction was monitored by TLC. After completion
of the reaction (6 h), the reaction mixture was filtered
in order to separate the oxidized Silphos. The solvent
was evaporated giving pure 2-phenylethyl acetate
(97%, 0.16 g); bp 229 °C, lit.24 bp 231–233 °C. The prod-
uct was found to be highly pure by GC analysis.
6. Kumareswaran, R.; Gupta, A.; Vankar, Y. D. Synth.
Commun. 1997, 27, 227–282.
7. Okano, T.; Miyamoto, K.; Kiji, J. Chem. Lett. 1995,
246.
8. Iqbal, J.; Srivastava, R. R. J. Org. Chem. 1992, 57, 2001–
2007.
9. Mukaiyama, T.; Shiina, I.; Miyashita, M. Chem. Lett.
1992, 625–628.
10. Miyashita, M.; Shiina, I.; Mukaiyama, T. Bull. Chem. Soc.
Jpn. 1993, 66, 1516–1520.
11. Firouzabadi, H.; Iranpoor, N.; Nowrouzi, F.; Amani, K.
Chem. Commun. 2003, 764–765.
12. (a) Chakraborti, A. K.; Basak, A.; Grover, V. J. Org.
Chem. 1999, 64, 8014–8017; (b) Merz, A. Angew. Chem.,
Int. Ed. Engl. 1973, 12, 846–847.
13. For examples, see: (a) Johnstone, R. A. W.; Rose, M. E.
Tetrahedron 1979, 35, 2169–2173; (b) Chevallet, P.;
Garrouste, P.; Malawska, B.; Martinez, J. Tetrahedron
Lett. 1993, 34, 7409–7412; (c) Wang, T.; Lui, A. S.;
Cloudsdale, I. S. Org. Lett. 1999, 1, 1835.
Typical procedure for N-formylation of aniline: To a mix-
ture of 1.0 g of Silphos and 5.0 mL of ethyl formate was
added 1.0 mmol of aniline (0.09 g) at room temperature.
The reaction mixture was stirred and monitored by
TLC. After completion of the reaction (75 min), the oxi-
dized Silphos was separated by filtration. The solvent
was evaporated giving pure formanilide in 92% yield
(0.11 g); mp 45–47 °C, lit.25 mp 48–50 °C. The product
was found to be highly pure by GC analysis.
Typical procedure for N-acetylation of benzylamine:
1.0 mmol of benzylamine (0.1 g) was added to 1.5 g of
Silphos [PCl3Àn(SiO2)n] in refluxing ethyl acetate
(5 mL) and stirred. The reaction was monitored by
TLC. After 0.2 h, the reaction mixture was filtered in
order to separate the oxidized Silphos. The solvent was
evaporated giving pure N-benzylacetamide (0.145 g,
97%); mp 59–60 °C, lit.26 mp 61 °C. The product was
found to be highly pure by GC analysis.
14. Shieh, W.-C.; Dell, S.; Repicˇ, O. J. Org. Chem. 2002, 67,
2188–2191.
15. (a) Gooßen, L. J.; Do¨hring, A. Synlett 2004, 263–266; (b)
Ho¨efle, G.; Steglich, W.; Vorbrueggen, H. Angew.
¨
Chem., Int. Ed. Engl. 1978, 90, 569–583; (c) Scriven,
E. F. V. Chem. Soc. Rev. 1983, 12, 129–161; (d) Hassner,
A.; Alaxanian, V. Tetrahedron Lett. 1978, 19, 4475–
4478.
Acknowledgements
We gratefully acknowledge the support of this study by
the Management and Programming Organization of
Iran and Shiraz University Research Council.
16. Staab, H. A. Angew. Chem. 1962, 74, 407–423.
17. Neises, B.; Steglich, W. Angew. Chem., Int. Ed. Engl. 1978,
17, 522–524.

 Iranpoor, Nasser
Iranpoor, Nasser
 Firouzabadi, Habib
Firouzabadi, Habib
 Jamalian, Arezu
Jamalian, Arezu