RSC Advances
Paper
NCH), 2.66–2.51 (m, 1H, CCH2C), 2.35 (s, 1H, NH), 1.94 (t, 1H,
CCH2C), 1.09 (d, 3H, J ¼ 7.64 Hz, CH3). 13C{1H} NMR (100 MHz,
CDCl3): 147.8, 147.1, 128.4, 128.8, 127.1, 127.1, 126.1 (Ar–C),
57.9 (NCH2), 57.2 (CPh2), 53.2 (NCH), 47.1 (CH2), 22.5 (CH3).
N-Methyl-2-methyl-5,5-diphenylpyrrolidine (7g)
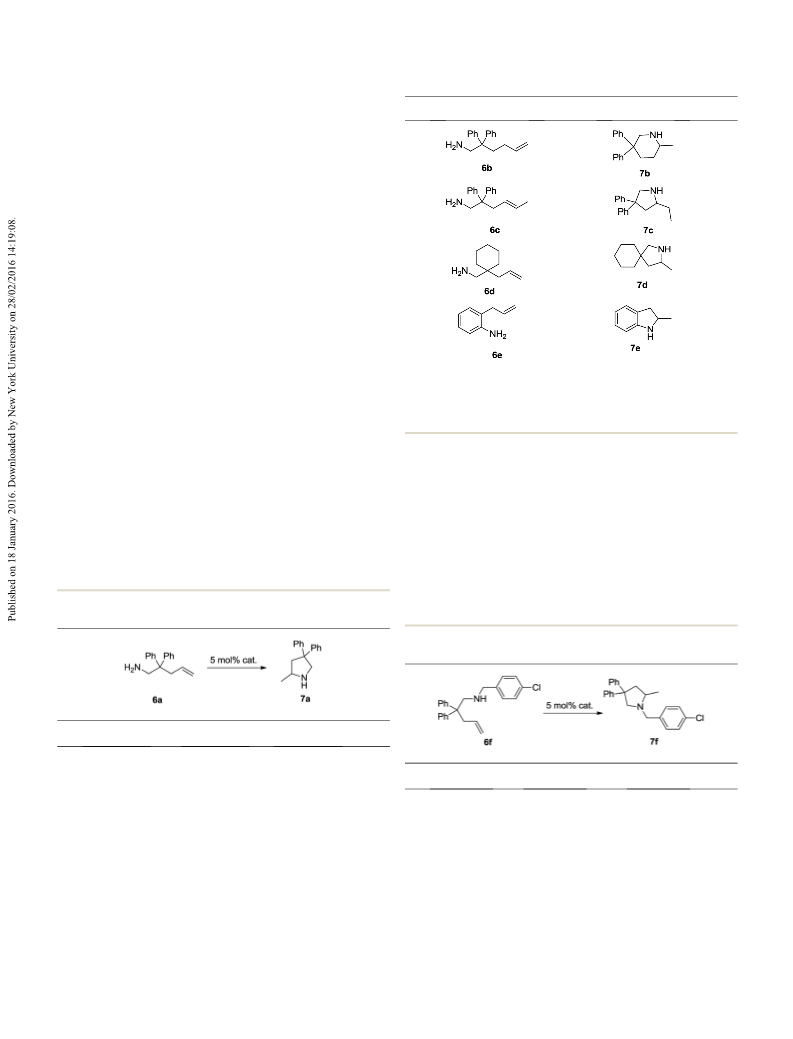
7g was prepared from 6g (126 mg, 0.5 mmol) as a colourless oil
1
in 95% yield (119 mg). H NMR (400 MHz, CDCl3): d 7.33–7.13
(m, 10H, ArH), 3.82 (d, 1H, J ¼ 8.96 Hz, NCHC), 2.92–2.87 (m,
2H, NCH2C), 2.56–2.48 (m, 1H, CCH2C), 2.38 (s, 1H, NCH3),
2.27–2.21 (m, 1H, CCH2C), 1.16 (d, 3H, J ¼ 6.06 Hz, CCH3). 13
C
2-Methyl-5,5-diphenylpiperidine (7b)
{1H} NMR (100 MHz, CDCl3): 128.4, 128.3, 127.6, 127.3, 126.0,
125.7 (Ar–C), 70.5 (NCH3), 62.1 (NCH2), 52.8 (CPh2), 48.7 (NCH),
40.7 (CH2), 19.1 (CCH3).
7b was prepared from 6b (126 mg, 0.5 mmol) as a colourless oil
in 97% yield (122 mg). 1H NMR (400 MHz, CDCl3): d 7.40 (d, 2H,
J ¼ 7.28 Hz, ArH), 7.33 (t, 2H, ArH), 7.08–7.23 (m, 6H, ArH), 3.89
(dd, J ¼ 3.19 Hz, J ¼ 3.15 Hz, 1H), 3.09 (d, 1H, J ¼ 13.53 Hz,
NCH2C), 2.80–2.72 (m, 1H, NCH), 2.67–2.73 (m, 1H, CCH2C),
2.24–2.16 (m, 1H, CCH2C), 1.65–1.61 (m, 1H, CCH2C), 1.48 (s,
1H, NH), 1.20–1.10 (m, 1H, CCH2C) 0.99 (d, 3H, J ¼ 6.33 Hz,
CH3). 13C{1H} NMR (100 MHz, CDCl3): 149.0, 144.9, 128.4,
128.8, 128.4, 126.6, 126.0 (Ar–C), 56.0 (NCH2), 52.5 (CPh2), 45.4
(NCH), 35.6 (CH2), 31.6 (CH2), 22.6 (CH3).
N-Methyl-2-methyl-5,5-diphenylpiperidine (7h)
7h was prepared from 6h (132 mg, 0.5 mmol) as a colourless oil
in 97% yield (129 mg). 1H NMR (400 MHz, CDCl3): d 7.41 (d, 2H,
J ¼ 8.28 Hz, ArH), 7.27–7.11 (m, 8H, ArH), 3.47 (d, 1H, J ¼ 11.8
Hz, NCHC), 2.40–2.44 (m, 1H, NCH2C), 2.28 (s, 1H, NCH3), 2.19–
2.11 (m, 1H, NCH2C), 1.58–1.51 (m, 1H, CCH2C), 1.31–1.21 (m,
1H, CCH2C), 1.02 (d, 3H, J ¼ 6.06 Hz, CCH3). 13C{1H} NMR (100
MHz, CDCl3): 149.1, 146.9, 128.9, 128.1, 127.8, 127.2, 126.8,
125.8, 125.3 (Ar–C), 66.0 (NCH3), 59.0 (NCH2), 46.8 (CPh2), 43.2
(NCH), 35.2 (CH2), 31.0 (CH2), 19.7 (CH3).
2-Ethyl-4,4-diphenylpyrrolidine (7c)
7c was prepared from 6c (132 mg, 0.5 mmol) as a colourless oil
1
in 88% yield (117 mg). H NMR (400 MHz, CDCl3): d 7.28–7.10
1-Benzyl-2-methyl-4,4-diphenylpyrrolidine (7i)
(m, 10H, ArH), 3.64 (d, 1H, J ¼ 13.39 Hz, NCH2C), 3.35 (d, 1H, J
¼ 10.08 Hz, NCH2C), 3.13–3.08 (m, 1H, NCH), 2.74–2.70 (m, 1H,
CCH2C), 1.98–1.96 (m, 1H, CCH2C), 1.46–1.21 (m, 2H, CCH2C),
1.20 (s, 1H, NH), 0.89 (t, 3H, CH3). 13C{1H} NMR (100 MHz,
CDCl3): 147.9, 147.1, 130.0, 128.5, 128.4, 127.2, 127.1, 126.2 (Ar–
C), 59.6 (NCH2), 57.8 (NCH), 56.9 (CPh2), 45.1 (CH2), 30.5 (CH2),
11.8 (CH3).
7i was prepared from 6i (164 mg, 0.5 mmol) as a colourless oil in
85% yield (139 mg). 1H NMR (400 MHz, CDCl3): d 7.37–7.08 (m,
15H, ArH), 4.06 (d, 1H, J ¼ 14.84 Hz, NCH2C), 3.61 (d, 1H, J ¼
9.86 Hz, NCH2C), 3.22 (d, 1H, J ¼ 12.75 Hz, NCH2C), 2.93–2.88
(m, 1H, NCHC), 2.84–2.81 (m, 2H, CCH2C), 2.76 (d, 1H, J ¼ 9.66
Hz, NCH2C), 2.22–2.17 (m, 1H, CCH2C), 1.15 (d, 3H, J ¼ 6.08 Hz,
CCH3). 13C{1H} NMR (100 MHz, CDCl3): 150.8, 146.9, 148.9,
140.3, 128.8, 128.4, 128.3, 128.0, 127.6, 127.5, 127.0, 126.0, 125.6
(Ar–C), 66.7 (NCH2Ph), 59.9 (NCH2), 58.2 (NCH), 52.7 (CPh2),
48.2 (CH2), 19.7 (CH3).
3-Methyl-2-azaspiro[4.5]decane (7d)
1
7d was prepared from 6d (46 mg, 0.3 mmol) in 95% yield. H
NMR (400 MHz, PhBr-d5): d 2.85–2.76 (m, 1H, NCH), 2.48 (d, 1H,
J ¼ 10.32 Hz, NCH2), 2.28 (d, 1H, J ¼ 11.16 Hz, NCH2), 1.42–1.35
(m, 1H, CCH2C), 1.12–1.06 (m, 1H, CCH2C), 1.28 (s, 1H, NH),
1.12–1.06 (m, 10H, CCH2CH2CH2CH2CH2C), 0.83 (d, 3H, J ¼
7.64 Hz, CH3), 0.69–0.64 (m, 1H, CCH2C).
2-Methyl-1-(4-nitrobenzyl)-4,4-diphenylpyrrolidine (7j)
7j was prepared from 6j (186 mg, 0.5 mmol) as a colourless oil in
97% yield (181 mg). 1H NMR (400 MHz, CDCl3): d 8.06 (d, 2H, J
¼ 12.00 Hz, ArH), 7.42 (d, 2H, J ¼ 7.63 Hz, ArH), 7.15–7.02 (m,
10H, ArH), 3.99 (d, 1H, J ¼ 16.38 Hz, NCH2C), 3.49 (d, 1H, J ¼
11.62 Hz, NCH2C), 3.30 (d, 1H, J ¼ 13.73 Hz, NCH2C), 2.85–2.78
(m, 2H, CCH2C), 2.73 (d, 1H, J ¼ 9.79 Hz, NCH2C), 2.20–2.14 (m,
1H, NCHC), 1.07 (d, 3H, J ¼ 5.10 Hz, CCH3). 13C{1H} NMR (100
MHz, CDCl3): 150.2, 148.5, 148.4, 147.2, 129.2, 128.4, 128.1,
127.5, 127.2, 126.1, 125.8, 123.7 (Ar–C), 66.6 (NCH2Ph), 59.8
(NCH2), 57.5 (NCH), 52.8 (CPh2), 47.8 (CH2), 19.9 (CH3).
2-Methylindoline (7e)
1
7e was prepared from 6e (40 mg, 0.3 mmol) in 94% yield. H
NMR (400 MHz, PhBr-d5): d 7.19–6.44 (m, 4H, ArH), 3.39 (s, 2H,
CH2), 1.95 (s, 1H, NH), 1.65 (s, 1H, CH), 0.81 (s, 3H, CH3).
1-(4-Chlorobenzyl)-2-methyl-4,4-diphenylpyrrolidine (7f)
1-(4-Methoxybenzyl)-2-methyl-4,4-diphenylpyrrolidine (7k)
7f was prepared from 6f (181 mg, 0.5 mmol) as a colourless oil in
87% yield (157 mg). 1H NMR (400 MHz, CDCl3): d 7.21–7.05 (m, 7k was prepared from 6k (179 mg, 0.5 mmol) as a colourless oil
1
14H, ArH), 3.96 (d, 1H, J ¼ 13.69 Hz, NCH2C), 3.54 (d, 1H, J ¼ in 88% yield (157 mg). H NMR (400 MHz, CDCl3): d 7.30–7.09
9.44 Hz, NCH2C), 3.16 (d, 1H, J ¼ 14.63 Hz, NCH2C), 289–2.84 (m, 12H, ArH), 6.86 (d, 2H, J ¼ 8.18 Hz, ArH), 4.01 (d, 1H, J ¼
(m, 1H, NCH2C), 2.81–2.75 (m, 1H, CCH2C), 2.71 (d, 1H, J ¼ 9.67 14.37 Hz, NCH2C), 3.80 (s, 3H OCH3), 3.62 (d, 1H, J ¼ 10.09 Hz,
Hz, NCH2C), 2.20–2.14 (m, 1H, CCH2C), 1.10 (d, 3H, J ¼ 6.00 Hz, NCH2C), 3.18 (d, 1H, J ¼ 18.95 Hz, NCH2C), 2.95–2.89 (m, 1H,
CCH3). 13C{1H} NMR (100 MHz, CDCl3): 150.6, 148.8, 138.9, NCHC), 2.83–2.80 (m, 1H, CCH2C), 2.75 (d, 1H, J ¼ 10.20 Hz,
132.6, 130.1, 128.5, 128.1, 128.0, 127.6, 126.1, 128.0, 127.6, NCH2C), 2.23–2.18 (m, 1H, CCH2C), 1.17 (d, 3H, J ¼ 5.36 Hz,
125.7, 127.0, (Ar–C), 66.6 (NCH2Ph), 59.8 (NCH2), 57.5 (NCH), CCH3). 13C{1H} NMR (100 MHz, CDCl3): 158.7, 150.87, 149.0,
52.7 (CPh2), 48.1 (CH2), 19.7 (CH3).
132.3, 129.9, 128.3, 128.0, 127.6, 127.5, 126.0, 125.6, 113.8 (Ar–
10546 | RSC Adv., 2016, 6, 10541–10548
This journal is © The Royal Society of Chemistry 2016

 Zhang, Yu
Zhang, Yu
 Sun, Qiu
Sun, Qiu
 Wang, Yaorong
Wang, Yaorong
 Yuan, Dan
Yuan, Dan
 Yao, Yingming
Yao, Yingming
 Shen, Qi
Shen, Qi