Molecular Interactions between Ammonium ILs and DMF
was added to a solution of tetramethylammonium bromide
J. Phys. Chem. B, Vol. 114, No. 42, 2010 13417
dielectric liquid. Experimental values of F and u at various
temperatures are collected in Table 1 for ILs, DMF, and their
mixtures over the whole composition range. It was found that
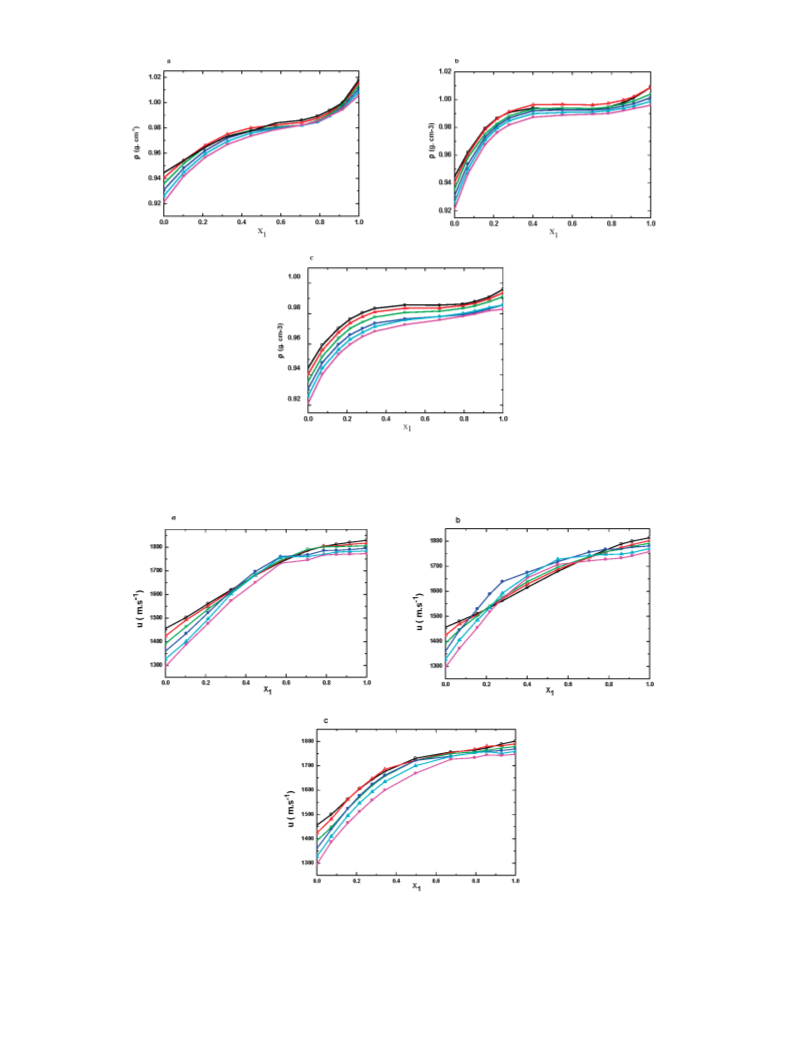
the F or u of the mixtures increases with increasing concentra-
tions of the IL in DMF, as shown in Figures 2 and 3,
respectively. The effect of the ILs on the F and u in the DMF
has been examined at various temperatures. As it can be
observed, the F and u sharply decrease as the temperature
increases in the three systems. For the sake of clarity and
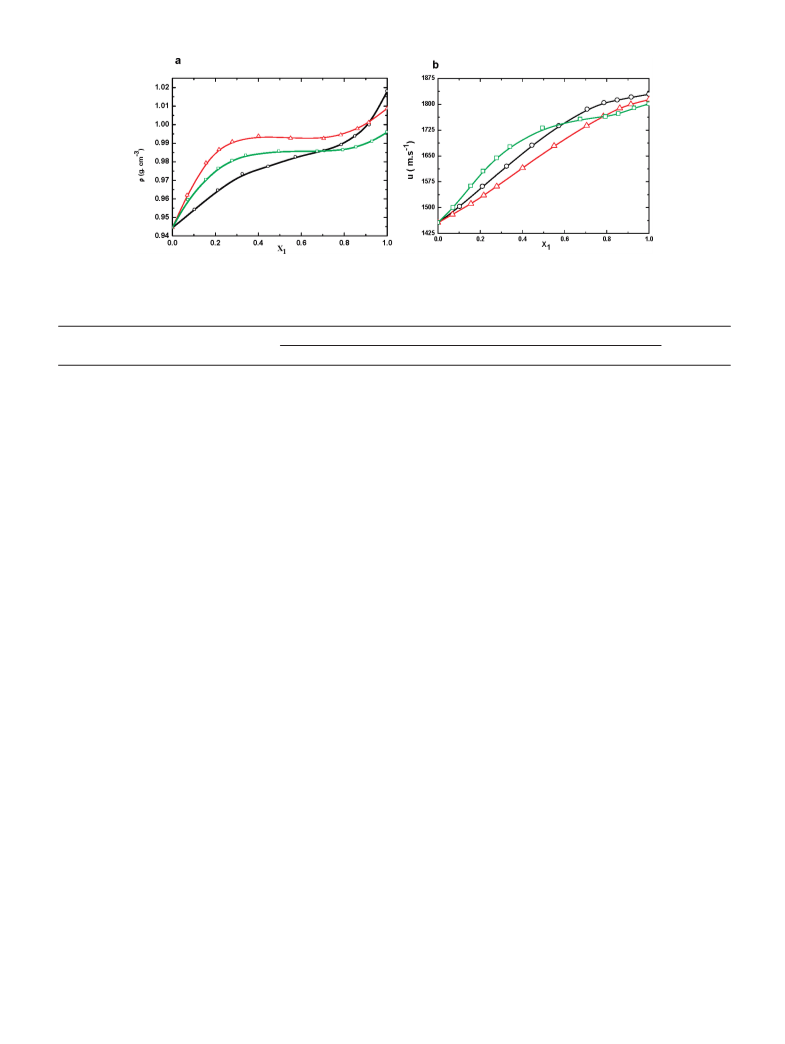
comparison between three ILs, the F and u values of IL + DMF
over the whole composition range at 25 °C have been presented
in Figure 4. The results in Figure 4a clearly show that the density
values of the TEAH + DMF mixtures are higher when
compared to the values for mixtures of the TMAH + DMF
and TPAH + DMF. However, the TMAH + DMF mixtures
have higher F values at very rich IL concentration ≈0.9600.
The density of all the ILs increases as the mole fraction of IL
increases up to 0.6000. A drastic change in F was found in the
case of TMAH + DMF at the mole fraction of TMAH
[
(CH
and the mixture was stirred vigorously at room temperature for
0 h. The precipitated KBr was filtered off, and the filtrate was
evaporated to leave the crude [(CH N][OH] as a viscous liquid
that was washed with ether (2 × 20 mL) and dried at 70 °C for
h to obtain the pure IL. The sample was analyzed by Karl
Fisher titration which revealed very low levels of water (below
3 4
) N][Br] (40 mmol) in dry methylene chloride (20 mL),
1
3 4
)
5
1
7
0 ppm). The yield of TMAH was 68%. H NMR (DMSO-d
6
):
δ (ppm) 2.38 (s, 12H), 4.79 (s, OH). HRMS calculated for
+
C
4
H
13NO (M - OH) 91.15, found 91.09.
Synthesis of Tetraethylammonium Hydroxide (TEAH). A
similar procedure as delineated above for [C N][OH] was
followed with the exception of the use of [(C N][Br]
N][Br]. The yield of TEAH was
): δ (ppm) 1.01 (t, 12H), 3.06 (q,
2
5 4
H )
2
5 4
H )
(
[cation]) instead of [(CH
8%. H NMR (DMSO-d
H), 4.43 (s, OH). HRMS calculated for C
47.26, found 147.20.
3
6
)
4
1
7
8
1
+
8
H21NO (M - OH)
Synthesis of Tetrapropylammonium Hydroxide (TPAH). A
procedure similar to that above for [(CH N][OH] was followed
with the exception of the use of [(C N][Br] ([cation]) instead
3 4
)
0
.6000-0.9000, which may be due to the increased intermo-
3
7 4
H )
lecular interaction between the TMAH and DMF. However, in
the case of TPAH or TEAH, there is a sharp decrease in F. In
other words, we can state that the density does not increase
sharply at mole fraction 0.6000-0.9000, which may be due to
a decrease in the ion-pair interaction between these ILs and
DMF. The density generally decreases with increasing length
1
of [(CH
3
)
4
N][Br]. The yield of TPAH was 82%. H NMR
(
4
2
DMSO-d
6
): δ (ppm) 0.8 (t, 12H), 1.46 (m, 8H), 2.92 (t, 8H),
.56 (s, OH). HRMS calculated for C12
03.36, found 203.25.
H
29NO (M+ - OH)
Methods. The density measurements were performed with
an Anton-Paar DMA 4500 M vibrating-tube densimeter, equipped
with a built-in solid-state thermostat and a resident program
with accuracy of temperature of (0.03 °C. Typically, density
of alkyl chain in a cation or anion as documented earlier for
28,29
ILs.
Interestingly, this conclusion is not consistent with the
observations of the present study. The experimental results
described that the densities of investigated systems increase with
increasing the length of alkyl chain in IL. We observed the F
values to be higher in the TEAH mixture, lower in the TMAH,
and moderate in the TPAH at equimolar mixture. These
discrepancies vary from IL to IL and solvent to solvent and
also depend on the nature as well as structural arrangement of
IL and solvent.
-3
precisions are 0.000 05 g cm . Proper calibration at each
temperature was achieved with doubly distilled, deionized water
E
and with air as standards. The excess molar volumes (V )
3
-1
(
(0.003 cm mol ) were deduced from the densities of the
pure compounds and mixture (F ) using the standard equations.
m
Ultrasonic sound velocities were measured by a single-crystal
ultrasonic interferometer (model F-05) from Mittal Enterprises,
New Delhi, India, at 2 MHz frequency at various temperatures.
A thermostatically controlled, well-stirred circulated water bath
with temperature controlled to (0.01 °C was used for all the
ultrasonic sound velocity measurements. The uncertainty in
Ultrasonic sound velocities are also another important source
of information about the properties of different solvents and
their mixture. The values of u were found to decrease with an
increase in temperature while u values increase with increasing
in mole fraction of IL. As noted from Figure 3, there is a sharp
increase of u in all ILs, except in the mixture of TEAH or TPAH
with DMF at the mole fraction range from 0.4000 to 0.9900 of
IL. At this range the u values increase slightly for the mixtures
of TEAH or TPAH with DMF at all investigated temperatures.
By addition of TEAH to DMF, u values increase rapidly up to
≈0.4000 of IL and finally slightly increase to the corresponding
value in TEAH through a maximum at XIL ) 0.9000 (Figure
1
sound velocity is 0.02%. H (400 MHz) spectra were recorded
6
on a JEOL 400 NMR spectrometer in DMSO-d . The reactions
were monitored by thin layer chromatography (TLC) using
aluminum sheets with silica gel 60 F254 (Merck). Clear
solutions were prepared gravimetrically using a Mettler Toledo
balance with a precision of (0.0001 g. The uncertainty in
solution composition expressed in mole fraction was found to
-4
be less than 5 × 10 . Mixing of the two components was
promoted by the movement of a small glass sphere (inserted in
the vial prior to the addition of the ILs) as the flask was slowly
and repeatedly inverted. After sample was mixed, the bubble-
free homogeneous sample was transferred into the U-tube of
the densimeter or the sample cell of ultrasonic interferometer
through a syringe.
4
b). This u value is significantly increased in IL-solvent
interactions when the mole fraction of IL increases. Fascinating
results are seen in the case of TPAH for which its u value at 30
C is higher than that at 25 °C, while a decrease is noted when
°
higher mole fraction is reached. This may be due to the self-
interaction occurring between the TPAH molecules at 30 °C.
As seen from Figure 4b, the sound velocity of TMAH or TEAH
with DMF at 25 °C increases with increasing composition of
IL. On the other hand, in the case of TPAH with DMF, there is
a rapid increase in u values up to the mole fraction ≈0.6000 of
TPAH, with the values sharply increasing up to mole fraction
0.9900 of TPAH. As seen from the results in Figure 4b, it is
clear that there is no regular order in the values of u for the ILs
with DMF and this may be due to the large steric hindrance in
the bulky groups, which tends to resist crowding more than
smaller groups. The TPAH IL exhibits a higher u values up to
Results and Discussion
To understand the molecular interactions of DMF with the
alkyl series of the hydroxide family of ammonium ILs, we have
measured the thermophysical properties such as F and u over
the entire composition range at temperature ranging from 25 to
5
0 °C under atmospheric pressure. In reality, ILs are miscible
with medium to high dielectric liquids and immiscible with low
dielectric liquids.27 In the present study, all ILs are completely
2
3
miscible in DMF (ε ) 36.71 at 25 °C), since DMF is a high

 Attri, Pankaj
Attri, Pankaj
 Venkatesu, Pannuru
Venkatesu, Pannuru
 Kumar, Anil
Kumar, Anil