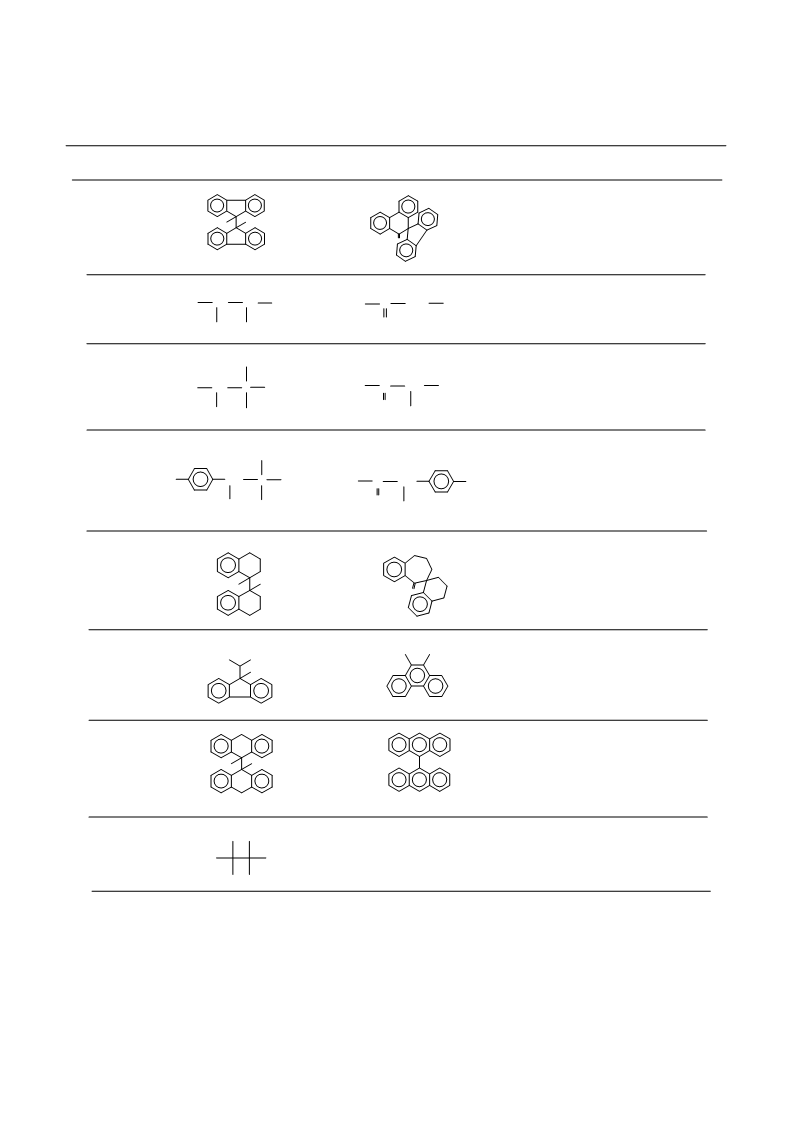
Molecules 2001, 6
446
1
Compound 4, H-NMR (500 MHz, CDCl ) δ: 3.75 (s, 3H), 5.97(s, 1H), 6.84 (m, 2H), 7.18-7.26(m,
5
3
1
3
H), 7.29-7.32(m, 2H), 7.37-7.40(m, 2H), 7.48-7.50(m, 1H), 7.99-7.99(m, 2H); C-NMR (125 MHz,
CDCl ) δ: 55.17, 58.55, 126.99, 128.53, 128.62, 128.89, 129.00, 130.12, 131.11, 132.90, 136.87,
3
1
39.45, 158.66, 198.41, HRMS calcd for C H O 302.14191, found 302.13247.
2
1
18
2
1
Compound 5, H-NMR (500 MHz, CDCl ) δ: 1.70-2.08(m, 6H), 2.15-2.21(m, 1H), 2.27-2.33(m, 1H),
3
1
3
2.73-2.93(m, 4H), 6.97-7.01(m, 1H), 7.04-7.18(m, 4H), 7.30-7.37(m, 1H), 7.37-7.44(m, 2H); C-
NMR (125 MHz, CDCl ) δ: 19.17, 22.33, 29.44, 29.85, 32.31, 36.25, 52.82, 125.91, 126.16, 126.94,
3
-1
1
27.70, 127.78, 128.45, 129.51, 131.09, 136.15, 137.17, 141.93, 142.47, 214.86, FTIR (1680 cm ,
CO), HRMS calcd for C H O 276.15142, found 276.15170.
2
0
20
1
Compound 6, H-NMR (500 MHz, CDCl ) δ: 5.45(s, OH), 7.24-7.44(m, 2H), 7.46-7.55(m, 4H), 7.59-
3
7
.72(m, 4H), 8.38(ddd, J=8.2, 1.5 and 0.5Hz, 1H), 8.66(ddd, J=8.7, 1.2 and 0.5Hz, 1H), 8.71(ddd,
13
J=8.7, 1.2 and 0.5Hz, 1H); C-NMR (125 MHz, CDCl ) δ: 117.25, 122.51, 122.57, 123.07, 123.99,
3
1
1
24.98, 125.36, 126.41, 126.63, 126.81, 127.22, 127.85, 128.63, 129.80, 131.03, 131.47, 132.47,
34.49, HRMS calcd for C H O 270.10447, found 270.10628.
2
0
14
1
Compound 7, H-NMR (500 MHz, CDCl ) δ: 7.06-7.14(m, 8H), 7.41(dd, J=6.3 and 1.2Hz, 2H),
3
1
3
7
1
3
.43(dd, 6.3 and 1.2Hz, 2H), 8.13(d, 8.5Hz, 4H), 8.66(s, 2H); C-NMR (125 MHz, CDCl ) δ: 125.24,
25.75, 126.78, 127.16, 128.47, 131.492, 131.570, 133.024, HRMS calcd for C H 354.14085, found
54.13923.
3
2
8
18
References:
1
2
3
4
5
6
. Toda, F. and Imai, N. Ylide reactions in the solid state: a simple procedure for the synthesis of
cyclopropanes. oxiranes and aziridines J. Chem. Soc. Perkin Trans. 1, 1994, 2673-2674.
. Toda, F.; Tanaka, K. and Hamai, K. Aldol condensation in the absence of solvent: Acceleration of
the reaction and enhancement of the stereoselectivity J. Chem. Soc. Perkin Trans 1, 1990, 3207
. Toda, F. Hiroshi, A. Enantioselective Wittig-Horner reaction in the solid state. J. Org. Chem.,1990,
5
5, 3446-3447.
. Toda, F.; Kiyoshige, K.; Ygi, M. NaBH reduction of ketones in the solid state. Angew.Chem.Int.
4
Ed. Engl. 1989, 28, 320-321.
. Tanaka, K.; Kishigami, S.; Toda, F. Reformatsky and Luche reaction in the absence of solvent J.
Org. Chem. 1991, 56, 4333.
. a) Toda, F. and Shigemasa, T. Pinacol rearrangement in the solid state. J. Chem. Soc. Perkin
Trans.1, 1989, 209-211. b) March, J. Advanced Organic Chemistry, 4th Ed., John Wiley & Sons,
New York, 1992, p. 1073.
7
. Ghiaci, M. and Asghari, Friedel Crafts alkylation and acylation in the absence of solvent. Synth.
Commun. 1998, 28, 2213-2220.
8
. Ghiaci, M. and Imanzadeh, H. A facile Beckman rearrangement of oximes with AlCl in the solid
3
state. ibid. 1998, 28, 2275-2280.

 Rashidi-Ranjbar, Parviz
Rashidi-Ranjbar, Parviz
 Kianmehr, Ebrahim
Kianmehr, Ebrahim