2 of 13
SALAHSHOURNIA AND GHIACI
and metal oxide type supports can lead to the desirable
catalytic performance.[21] Also, one of the best supports,
especially for palladium or platinum, has been activated
carbon. Therefore, a combination of these can make it
possible to apply graphene oxide as a support, which
might be beneficial. In recent years, much research has
been done on graphene and graphene oxide due to their
unique structures.[22,23] Especially, in catalytic applica-
tions, in addition to effective catalytic components, these
materials usually exhibit synergistic contributions to the
catalytic reactions.[24,25]
TEM images obtained in the Philips CM‐120 transmission
electron microscope with the accelerator voltage of 100
KV (Germany). BET specific surface area and pore size
distribution were measured on a PHS‐1020 (PHS, China)
system from the nitrogen adsorption–desorption iso-
therms at 77 K. FT‐IR spectra were collected from a
potassium bromide (KBr) pellet in a Jasco‐680 plus spec-
trophotometer (Japan).
In this work, in the continuation of our previous
research interests, we designed a new catalyst, Pd‐Pt/m‐
GO, in which the three‐dimensional m‐GO acted as the
support for immobilizing palladium and platinum nano-
particles. The prepared composite was characterized by
FT‐IR, Raman, X‐ray diffraction, BET, TEM, FESEM
and X‐ray photoelectron spectroscopy (XPS). The cata-
lytic performance of the Pd‐Pt/m‐GO for the liquid‐phase
reduction of various nitroaromatic compounds was inves-
tigated using a hydrogen source, with focus on stability of
the catalyst.
2.2 | Preparation of GO
Graphene oxide was prepared from graphite by the Hum-
mers method.[23]
2.3 | Preparation of the modified
graphene oxide with diisocyanate (m‐GO)
In a typical procedure, a 100 ml round bottom flask was
loaded with 500 mg graphene oxide and 50 ml of anhy-
drous dimethyl formaldehyde (DMF); then, the mixture
was stirred for 15 min under nitrogen gas. Subsequently,
20 mmol toluene diisocyanate (TDI) was added and
stirred under nitrogen gas for 24 hrs. Then, the reaction
mixture was poured into 200 ml of methylene chloride,
filtered and thoroughly washed with 50 ml of methylene
chloride; and finally, the solid was dried under vacuum.
2 | EXPERIMENTAL SECTION
2.1 | Materials and techniques
The reactor used for the hydrogenation reactions was a
stainless steel Bochiglasuster‐type hydrogenation reactor,
model cyclone 075, with the capacity of 300 ml, the max-
imum pressure of 60 bar and the temperature of 250 °C.
Toluene diisocyanate (TDI) was a mixture of the 2,4‐
and 2,6‐ isomers (80:20) with the purity of at least
99.5%, as purchased from Karun Petrochemical Company
(KRNPC). Other chemicals including PdCl2 and K2PtCl6,
graphite, and all of the solvents, such as dichloromethane
(DCM), dimethyl formaldehyde (DMF) and methanol
(MeOH), were purchased from Sigma‐Aldrich. DMF pre-
liminary was dried over MgSO4; then, it was distilled
under vacuum over NaH. Graphene oxide was prepared
from purified graphite by the Hummers method.[23]
A VARIAN gas chromatograph, model CP‐3800,
equipped with an FID detector was used for monitoring
the reactions. The column was a Varian capillary column
(CP‐Sil 5 CB, 30 m 0.32 mm 0.25 μm). The initial temper-
ature (100 to 280 °C with the rate 7.5 °C/min) was held at
280 °C for 15 min and the total time was 40.33 min. The
products of the reduction reactions were identified by the
GC/MS instrument (Fisons instrument 8060, USA).
FESEM micrographs were obtained from the Scanning
Electron Microscope (Mira 3‐XMU FESEM, Germany).
Particle size and morphology were determined from
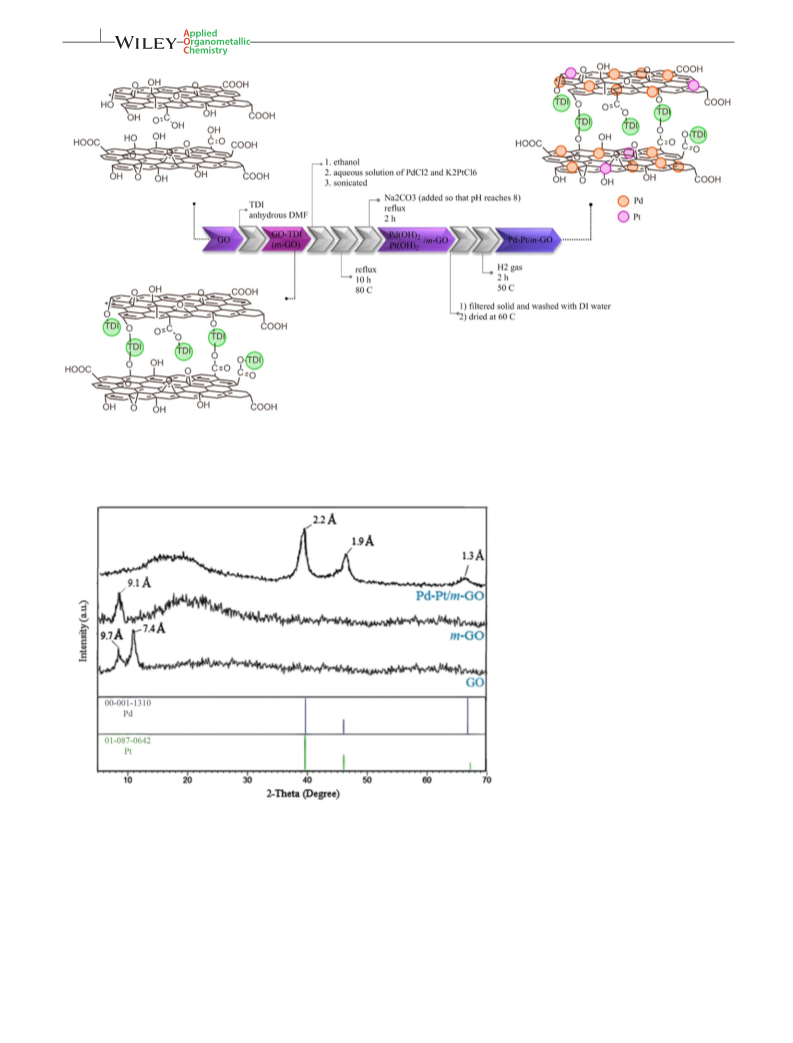
2.4 | Synthesis of the Pd‐Pt/m‐GO
catalysts
In a typical procedure, 37.2 mg of PdCl2 and 7.9 mg of
K2PtCl6 were dissolved in 5 ml of deionized water and
stirred for 30 min. 500 mg of the modified graphene oxide
with toluene diisocyanate (m‐GO) was dispersed in 10 ml
of ethanol; then the aqueous solution containing palla-
dium and platinum salts was added to the mixture and
sonicated (2 × 15 min). The mixture was refluxed for
10 hr at 80 °C. Then sodium carbonate (100 mg) was
added and the mixture was refluxed for 2 hr (pH reached
to 8). At this step, palladium hydroxide and platinum
hydroxide were immobilized on the m‐GO. The solid
was filtered and thoroughly washed with deionized water
and dried at 60 °C. Finally, the palladium and platinum
hydroxides immobilized on m‐GO were reduced by hydro-
gen at 50 °C for 2 hr. The prepared catalyst contained
5 wt% palladium and platinum, with a ratio of 7 to 1
(calculations are shown in the supplementary data).

 Salahshournia, Hossein
Salahshournia, Hossein
 Ghiaci, Mehran
Ghiaci, Mehran