ELECTROCHEMICAL AMINATION: IX.
1179
the yield of the meta isomer (Table 2) can be ex-
plained in terms of decrease in the concentration of
B and Ti(IV) with increasing concentration of acetic
acid, then the effect of the concentration of the
organic solvent on the para/ortho ratio implies par-
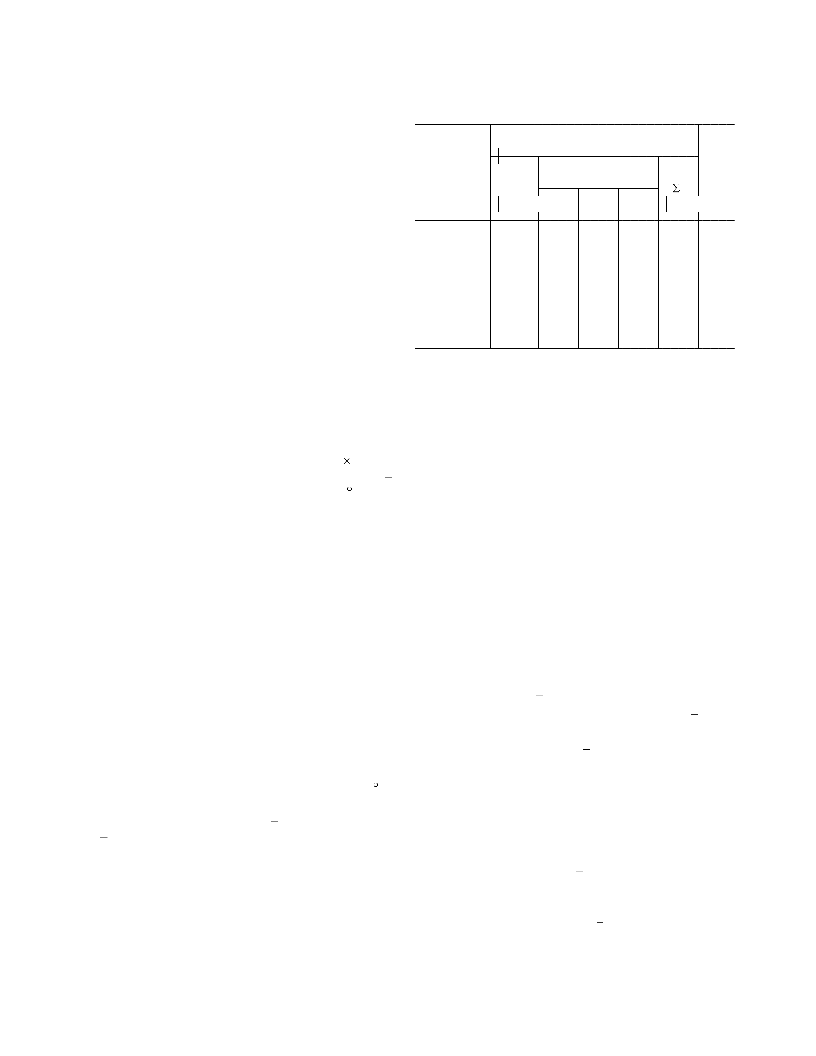
Table 2. Effect of the concentration of acetic acid on the
yield and composition of the products of benzene amina-
tion at a constant H O/H SO molar ratio of 2.7a
2
2
4
Bc, %
ticipation of CH COOH in complex formation. In
3
[
CH COOH],
para/
ortho
3
principle, solvent molecules can both react with the
monoamination product and enter the coordination
sphere of metal ions [1]. The alteration in the ligand
surrounding of Ti(IV) ions may, in particular, in-
fluence the stage of rearomatization of intermediate
III.
(H N) C H
2 2 6 4
M
b
PhNH2
ortho meta para
0
2
4
6
7
20.6
65.5
59.0
46.3
39.6
13.9
10.4
8.2
6.0
4.1
1.7
0.5
0.0
0.0
0.0 41.0 69.8 5.0
0.7 24.1 96.3 4.0
1.6 12.9 77.6 3.2
Hence, our present results show that acetic acid
affects the mechanism of electrochemical amination
and together with sulfuric acid it can serve as a tool
for contolling the relative rate and selectivity of
substitution.
4.5
7.8
0.0
0.0
5.0 57.5 2.9
1.4 49.3 2.7
0.0 13.9
c
9
c
10
0.0 10.4
a
c
The concentration of Ti(IV) ions in the absence of acetic acid
b
EXPERIMENTAL
is 0.3 M.
Total current yield of the amination products.
Electrolysis occurs at the potentials of electrochemical forma-
tion of hydrogen.
Quantitative analysis for aniline and isomeric
phenylenediamines was carried out by GLC on a
Chrom-4 chromatograph equipped with a flame-
ionization detector and a glass column [2500 3 mm,
hydrocarbonate. The amination products were extrac-
ted with chloroform.
5
% XE-60 on Chromaton N-AW-DMCS (0.160
.200 mm). The column temperature was 150 C, and
We used a 15% solution of titanium(IV) sulfate in
2 M sulfuric acid (analytical grade), chemical grade
sulfuric acid. Hydroxyalmine sulfate was twice re-
crystallized. Isomeric phenylenediamines were
purified by sublimation. Aniline was distilled in a
vacuum over KOH. Chemical grade benzene, analy-
tical grade chloroform, and chemical grade acetic acid
were distilled. All solutions were prepared with twice
distilled water.
0
the carrier gas was helium at a flow rate of 30 ml/min.
Cathode-initiated amination of benzene was carried
out in the three-electrode glass electrochemical cell
described in [4, 9]. The cell was equipped with a
reflux condenser and a ceramic diaphragm separating
the cathode and anode spaces. The volume of the
catholyte, aqueous sulfuric acid containing 0.2 M
hydroxylamine and required concentrations of Ti(IV)
ions and acetic acid, was 25 ml, and the volume of
benzene was 10 ml. Before electrolysis the two-phase
system was deoxygenated with argon. A highly
dispersed emultion of benzene in the electrolyte was
maintained by means of magnetic stirring, in the
course of amination argon was passed over the
emulsion. The anode was a mercury electrode with a
REFERENCES
1. Lisitsyn, Yu.A., Belyaeva, L.V., Sinyashin, O.G.,
and Kargin, Yu.M., Zh. Obshch. Khim., 1999, vol. 69,
no. 7, pp. 1196 1199.
2
3
. Minisci, F., Synthesis, 1973, no. 1, pp. 1 24.
. Tomat, R. and Rigo, A., J. Electroanal. Chem., 1977,
2
vol. 75, no. 2, pp. 629 635.
surface area of 12.3 cm . Electrolysis was carried out
in the galvanostatic mode using a P-5848 potentiostat,
cathodic current density 6 mA/cm , temperature 40 C.
4
. Lisitsyn, Yu.A. and Kargin, Yu.M., Zh. Obshch. Khim.,
2
1993, vol. 63, no. 6, pp. 1312 1317.
The working electrode potential was controlled by
means of a Sch 4313 voltmeter ammeter against a
silver silver chloride reference electrode. The anode
and anolyte were a platinum wire and a sulfuric acid
solution with a concentration equal to the acid con-
centration in the catholyte.
5. Lisitsyn, Yu.A., Kononchuk, A.M., and Kargin, Yu.M.,
Zh. Obshch. Khim., 1996, vol. 66, no. 12, pp. 2034
2038.
6. Tomat, R. and Rigo, A., J. Electroanal. Chem., 1972,
vol. 35, no. 1, pp. 21 26.
7
. Citterio, A., Gentile, A., Minisci, F., Navarrini, V.,
Sarravalle, M., and Ventura, S., J. Org. Chem., 1984,
vol. 49, no. 23, pp. 4479 4482.
When electrolysis was complete, the catholyte was
cooled, made weakly acidic with saturated aqueous
sodium hydroxide, and neutralized with sodium
8. Lisitsyn, Yu.A. and Kargin, Yu.M., Zh. Obshch. Khim.,
RUSSIAN JOURNAL OF GENERAL CHEMISTRY Vol. 71 No. 8 2001

 Lisitsyn
Lisitsyn
 Makarova
Makarova
 Kargin
Kargin