RSC Advances
Page 4 of 6
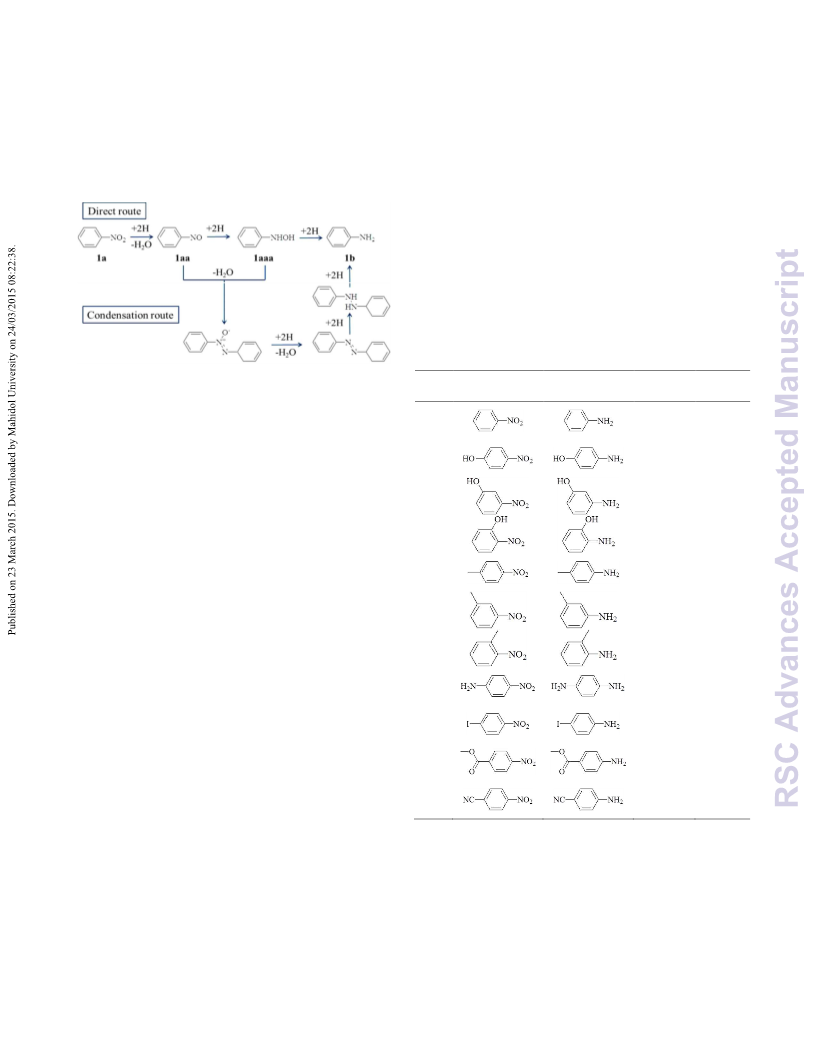
COMMUNICATION
DOI: 10.1039/C5RA01649E
ethanol and water. Then np-Ag was reused without further
purification. From scheme 2, it can be seen that the catalyst could be
reused even seven times without reducing the catalytic activity.
Fig.1b is the SEM image of the recovered catalyst after seven uses.
Compared to the fresh one (Fig. 1a), little change in porous
morphology and feature dimension was observed, but the ligament
size coarsened from 30 nm before the reaction to around 40 nm.
After the reaction the XPS spectrum of np-Ag was also detected, Fig.
2b. The binding energies of Ag 3d5/2 and Ag 3d3/2 were appeared at
368.5 and 374.5 eV, which implied that the surface status did not
change after the reaction. To clarify whether the np-Ag catalyst had
been leached into the reaction mixture or not, the reaction solution
was also tested by inductivity coupled plasma (ICP-AES) analysis
when the conversion of 4-NP was about 50%. The rate was lower
than the detection limits (<0.02 ppm). No leaching of silver was
detected. These evidences proved that np-Ag was a green and
sustainable catalyst for the reduction of aromatic nitro compounds.
Electronic Supplementary Information (ESI) available: [details of any
supplementary information available should be included here]. See
DOI: 10.1039/c000000x/
1
2
3
4
Y. Ding, M. Chen, MRS Bull. 2009, 34, 569-576.
X. Zhang, Y. Ding, Catal. Sci. Technol. 2013, , 2862-2868.
3
Y. Yamamoto, Tetrahedron 2014, 70, 2305-2317.
C. Xu, J. Su, X. Xu, P. Liu, H. Zhao, F. Tian, Y. Ding, J. Am. Chem.
Soc. 2007, 129, 42-43.
5
6
7
A. Wittstock, V. Zielasek, J. Biener, C. M. Friend, M. Bäumer,
Science 2010, 327, 319-322.
H. Yin, C. Zhou, C. Xu, P. Liu, X. Xu, Y. Ding, J. Phys. Chem. C
2008, 112, 9673-9678.
N. Asao, Y. Ishikawa, N. Hatakeyama, Y. Yamamoto
Menggenbateer, M. Chen, W. Zhang, A. Inoue, Angew. Chem. Int.
Ed. 2010, 49, 10093-10095.
Table 4. Reusability studies of np-Ag catalyst.
8
9
M. Yan, T. Jin, Y. Ishikawa, T. Minato, T. Fujita, L. Chen, M. Bao,
N. Asao, M. Chen, Y. Yamamoto, J. Am. Chem. Soc. 2012, 134
,
17536-17542.
R. Wang, C. Wang, W. Cai, Y. Ding, Adv. Mater. 2010, 22, 1845-
1848.
Reaction Cycle a)
Yield/%
1
2
3
4
5
6
7
10 R. Wang, C. Xu, X. Bi, Y. Ding, Energ. Environ. Sci. 2012, 5, 5281-
5286.
99 99 98 99 97 98 97
120 120 120 120 120 135 135
11 S. Tanaka, T. Kaneko, N. Asao, Y. Yamamoto, M. Chen, W. Zhang,
A. Inoue, Chem. Commun. 2011, 47, 5985–5987.
Time/min
a)
Reactions were performed using catalyst 0.1 mmol, 4-NP (1 mmol) and
12 T. Kaneko, S. Tanaka, N. Asao, Y. Yamamoto, M. Chen, W. Zhang,
A. Inoue, Adv. Synth. Catal. 2011, 353, 2927–2932.
NaBH4 (25 mmol) in 10 mL aqueous solution at room temperature.
13 Z. Li, S. Lin, L. Ji, Z. Zhang, X. Zhang, Y. Ding, Catal. Sci. Technol.
2014, 4, 1734-1737.
Conclusions
14 Z. Li, C. Zhang, J. Tian, Z. Zhang, X. Zhang, Y. Ding, Catal.
Commun. 2014, 53, 53-56.
Briefly conclusion, we realized the reduction of nitroarenes
with lesser amount of NaBH4 reductant under the mild reaction
condition by using nanoporous silver as a high active and
selective catalyst. The aromatic nitro compounds containing
both electro-withdrawing and electro-donating substituents
could be reduced with high yields. The catalyst can be reused
several times without evident loss of its catalytic activity. As a
cost-effective nanoporous material, the well reduction property
exhibited by nanoporous silver here will further arouse the
investigated interest of nanoporous silver in wider green
organic synthesis field.
15 S. Lawrence, Amines: Synthesis, properties and applications,
Cambridge University Press, Cambridge, 2004.
16 E. Burkhardt, K. Matos, Chem. Rev. 2006, 106, 2617-2650.
17 I. Nakamula, Y. Yamanoi, T. Imaoka, K. Yamamoto, H. Nishihara,
Angew. Chem. Int. Ed. 2011, 50, 5830-5833.
18 N. Sakai, K. Fujii, S. Nabeshima, R. Ikeda, T. Konakahara, Chem.
Commun. 2010, 46, 3173-3175.
19 R. V. Jagadeesh, G. Wienhöfer, F. A. Westerhaus, A.-E. Surkus, H.
Junge, K. Junge, M. Beller, Chem. Eur. J. 2011, 17, 14375-14379.
20 H. Wei and Y. Lu, Chem. Asian J. 2012, 7, 680-683.
21 K. Kurodaa, T. Ishidaa, M. Haruta, J. Mol. Catal. A-Chem. 2009,
298, 7-11.
Acknowledgements
22 K. Layek, M. Lakshmi-Kantam, M. Shirai, D. Nishio-Hamane, T.
Sasaki, M. Maheswaran, Green Chem. 2012, 14, 3164-3174.
23 S. Saha, A. Pal, S. Kundu, S. Basu, T. Pal, Langmuir 2010, 26, 2885–
2893.
Financial support from the National Science Foundation of China
(21472117 and 21176144) was acknowledged.
Notes and references
a
School of Chemistry and Chemical Engineering, Shandong University,
24 Z. Zhang, C. Shao, Y. Sun, J. Mu, M. Zhang, P. Zhang, Z. Guo, P.
Liang, C. Wang, Y. Liu, J. Mater. Chem. 2012, 22, 1387-1395.
25 T. Kou, D. Li, C. Zhang, Z. Zhang, H. Yang, J. Mol. Catal. A-Chem.
2014, 382, 55-63.
Jinan 250100, China.
b School of Pharmacy, Shihezi University, Shihezi 832000, China.
c
Department of Petroleum Engineering, University of Stavanger,
26 F. Kelly, J. Johnston, ACS Appl. Mater. Interfaces, 2011, 3, 1083-
Stavanger 4036, Norway.
1092.
†
Footnotes should appear here. These might include comments
27 S. Saha, A. Pal, S. Kundu, S. Basu, T. Pal, Langmuir 2010, 26, 2885-
relevant to but not central to the matter under discussion, limited
experimental and spectral data, and crystallographic data.
2893.
4 | J. Name., 2012, 00, 1-3
This journal is © The Royal Society of Chemistry 2012

 Li, Zhiwen
Li, Zhiwen
 Xu, Xiaohong
Xu, Xiaohong
 Jiang, Xiaojian
Jiang, Xiaojian
 Li, Yingchun
Li, Yingchun
 Yu, Zhixin
Yu, Zhixin
 Zhang, Xiaomei
Zhang, Xiaomei