
Angewandte Chemie - International Edition p. 13254 - 13258 (2019)
Update date:2022-08-12
Topics:
 Watthaisong, Pratchaya
Watthaisong, Pratchaya
 Pongpamorn, Pornkanok
Pongpamorn, Pornkanok
 Pimviriyakul, Panu
Pimviriyakul, Panu
 Maenpuen, Somchart
Maenpuen, Somchart
 Ohmiya, Yoshihiro
Ohmiya, Yoshihiro
 Chaiyen, Pimchai
Chaiyen, Pimchai
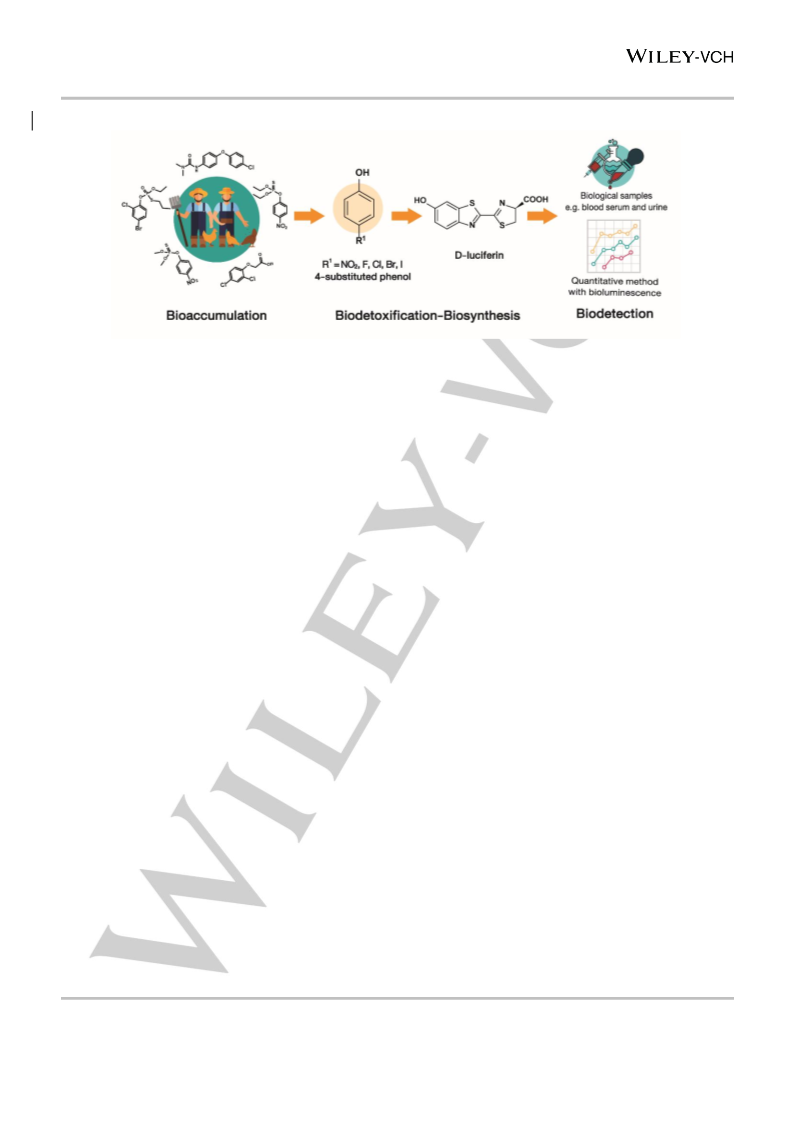
The flavin-dependent monooxygenase, HadA, catalyzes the dehalogenation and denitration of the toxicants, nitro- and halogenated phenols, to benzoquinone. The HadA reaction can be applied in one-pot reactions towards the de novo synthesis of d-luciferin by coupling with d-Cys condensation. d-luciferin, a valuable chemical widely used in biomedical applications, can be used as a substrate for the reaction of firefly luciferase to generate bioluminescence. As nitro- and halogenated phenols are key indicators of human overexposure to pesticides and pesticide contamination, the technology provides a sensitive and convenient tool for improved biomedical and environmental detection at ppb sensitivity in biological samples without the requirement for any pre-treatment. This dual-pronged method combines the advantages of waste biodetoxification to produce a valuable chemical as well as a smart detection tool for environmental and biomedical detection.
View More


website:http://www.orchid-chem.com
Contact:+86-571-85395792
Address:607, North Zhongshan Road, Hangzhou 310000 China
Zhejiang Bolaite Technology Co.,Ltd
Contact:+86-0570-6523168 +86-0570-6523150
Address:No.5 YinKeng Road Kaihua Industrial Park(Chengguan Town) Zhejiang Prov.
Contact:021-61840961 021-61840962
Address:No.157,Zhuguang Rd, Qingpu, Shanghai, China
ShenZhen InnoSyn Biotech Co.,Ltd
website:http://www.innosyns.com
Contact:+86-755-28351685
Address:Floor 5 & 6, Building A1, HAIKEXING Strategic Innovative Industrial Park, 16 BaoShan Road, PingShan District
Shenzhen Sunrising Industry Co., ltd.
Contact:+86 755 86571158 / 86571159 / 86571160
Address:2108 ZHENYE INT. BUSINESS CENTER,NO.3101-90 QIANHAI RD, NANSHAN,SHENZHEN, CHINA
Doi:10.1021/acs.orglett.5b00320
(2015)Doi:10.1016/j.tetlet.2021.153140
(2021)Doi:10.1016/S0040-4020(01)00769-4
(2001)Doi:10.1016/S0040-4020(01)85104-8
(1988)Doi:10.1021/acs.macromol.5b00470
(2015)Doi:10.1021/ja00373a030
(1982)