2
04
K. Riener et al. / Journal of Catalysis 331 (2015) 203–209
2
.3.2. Reaction of [{IrCl(COD)}
In an NMR tube equipped with a J. Young valve, a mixture of
2.1 L Me SiHCl (27.6 mg, 292 mol, 10 equiv.) and 30.5
(33.6 mg, 292 mol, 10 equiv.) in 1.0 mL CD Cl is added
to a solution of 19.6 mg [{IrCl(COD)} ] (29.2 mol, 1.0 equiv.) in
is used as internal standard for NMR analysis.
2 2 2
] and a mixture of Me SiHCl/MeSiHCl
3
l
2
l
lL
MeSiHCl
2
l
2
2
2
l
1
2 2 3
.6 mL CD Cl . CHCl
Scheme 1. Ir- and Pt-catalyzed hydrosilylation of allyl compounds [1,2,4].
2
.3.3. Synthesis of [Ir(allyl)(Cl)
.11 g [{IrCl(COD)} ] (1.65 mmol, 1.0 equiv.) is dissolved in
20 mL CH Cl . 1.35 mL allyl chloride (1.26 g, 16.5 mmol, 10 equiv.)
is added, the reaction mixture is stirred for 15 min at room temper-
ature and all volatiles are removed in vacuum. [Ir(allyl)(Cl) (COD)]
is obtained in quantitative yield. H NMR (major isomer; 400 MHz,
CD Cl , 213 K): d = 1.66 (m , 1 H, CH -COD), 1.87 (m , 1 H, CH -COD),
.37–2.51 (m, 3 H, CH -COD), 2.62–2.77 (m, 1 H, CH -COD), 3.00
H,H = 8.2 Hz, 1 H, CH -COD), 3.22 (m , 1 H,
H,H = 8.2 Hz, 1 H, CH-COD), 3.49 (d,
2
(COD)]
1
2
1H and 13C chemical shifts are reported in parts per million (ppm)
2
2
relative to TMS, with the residual solvent peak serving as internal
reference [25]. Elemental analyses were performed in the microan-
alytical laboratory of TUM-CRC and FAB mass spectrometry was
carried out using a Finnigan MAT 90. A Jasco VT-550 photometer
was used to conduct the UV–Vis measurements in dichloro-
methane. TEM images were recorded at a nominal magnification
of 250,000 on a JEOL JEM 2011 electron microscope operated at
20 kV. The samples were prepared by adding a drop of the iridium
nanoparticles suspended in ethanol on copper grids covered with a
Quantifoil Multi A holey carbon film and a 2 nm carbon film on top.
2
1
2
2
c
2
c
2
2
2
2
2
3
(
dd,
J
H,H = 15.4 Hz,
J
2
c
3
CH
2
-COD), 3.42 (pseudo-q, J
3
3
J
H,H = 13.9 Hz, 1 H, anti CH
2
-allyl), 3.76 (d, JH,H = 10.1 Hz, 1 H, anti
H,H = 7.2 Hz, H, syn CH -allyl), 4.62
H,H = 8.2 Hz, H, CH-COD), 4.92 (pseudo-t,
H,H = 7.3 Hz, 1 H, CH-COD), 4.97 (d,
1
3
CH
2
-allyl), 4.32 (d,
J
1
2
3
(
pseudo-q,
J
J
1
3
3
J
H,H = 8.6 Hz, 1 H, syn
H,H = 7.3 Hz, 1 H, CH-COD), 5.67–5.77
m, 1 H, CH-allyl). C { H} NMR (major isomer; 101 MHz, CD Cl
-COD),
-allyl), 85.9 (CH-COD),
8.5 (CH-COD), 92.1 (CH-COD), 93.8 (CH-COD), 118.4 (CH-allyl).
3
2
CH -allyl), 5.07 (pseudo-t, J
(
2
3
1
3
1
2
2
,
2
2
.2. Catalytic reactions
13 K): d [ppm] = 25.5 (CH
6.8 (CH -COD), 43.6 (CH
2
-COD), 28.4 (CH
-allyl), 84.2 (CH
2 2
-COD), 35.3 (CH
2
2
2
.2.1. General protocol for hydrosilylation experiments
A mixture of [{IrCl(COD)} ], cyclooctadiene (COD) and the allyl
2
8
1
H NMR (minor isomer; 400 MHz, CD
2
Cl
2
, 213 K): d [ppm] = 1.26
compound (55.0 mmol, 1.0 equiv.) is stirred (500 rpm) at 40 °C
for 10 min in a reaction flask equipped with a reflux condenser.
At this temperature, the silane (55.0 mmol, 1.0 equiv.) is added
using a Landgraf LA-30 syringe pump, followed by the addition
of 1.28 mL mesitylene (1.10 g, 9.15 mmol, 6.0 equiv.) as internal
standard. The reaction mixture is further stirred for 10 min at
⁄
2
(
m
J
c
, 1 H, CH
2
-COD), 1.66 (1 H, CH
2
-COD), 2.18 (dd, JH,H = 14.7 Hz,
3
H,H = 8.9 Hz, 1 H, CH
2
-COD), 2.25–2.33 (m, 1 H, CH
2
-COD), 2.37–
⁄
⁄
2
.51 (1 H, CH
2
-COD), 2.62–2.77 (1 H, CH
H,H = 8.0 Hz, 1 H, CH
2
-COD), 2.88 (dd,
2
3
⁄
J
H,H = 15.6 Hz,
J
2
-COD), 2.97–3.03 (1 H,
3
⁄
CH
2
-COD), 4.24 (pseudo-q, JH,H = 8.0 Hz, 1 H, CH-COD), 4.31–4.34
3
(1 H, anti CH
2
-allyl), 4.49 (d,
.59–4.65 (1 H, CH-COD), 4.72 (m
JH,H = 4.5 Hz, 1 H, syn CH
2
-allyl),
4
0 °C and an aliquot is taken for NMR analysis.
⁄
⁄
4
c
, 1 H, CH-COD), 5.05–5.09
3
(2 H, CH-allyl, anti CH
2
-allyl), 5.17 (d,
JH,H = 4.5 Hz, 1 H, syn
3
3
13
CH
{
2
-allyl), 5.54 (dd,
J
H,H = 6.0 Hz,
J
H,H = 8.0 Hz, 1 H, CH-COD).
Cl , 213 K): d [ppm] = 25.9
-COD), 37.3 (CH -COD), 45.3
-allyl), 86.2 (CH-COD), 87.1 (CH-COD), 88.1 (CH-COD), 93.3
CH-COD), 94.8 (CH -allyl), 110.4 (CH-allyl). MS (FAB): m/z (%)
713 (51) [2 M – allyl – 2 Cl] , 377 (51) [M – Cl] , 335 (100)
C
2
.2.2. Hydrosilylation of allyl chloride-d
In an NMR tube equipped with a J. Young valve, 4.10 mg [{IrCl
COD)} ] (6.10 mol, 14.6 mol% Ir) is dissolved in 0.7 mL CD Cl
L allyl chloride-d (6.58 mg, 83.8 mol, 1.0 equiv.) is added
2 2
and Me SiHCl
1
H} NMR (minor isomer; 101 MHz, CD
-COD), 28.4 (CH -COD), 34.0 (CH
2
2
(
(
(
CH
CH
2
2
2
2
(
7
2
l
2
2
.
2
.00
l
2
l
2
to the reaction mixture and the NMR tube is shaken for 15 min, fol-
lowed by addition of 9.20 L Me SiHCl (7.91 mg, 83.6 mol,
.0 equiv.). Mesitylene is used as internal standard for NMR
analysis.
+
+
=
l
2
l
+
[M – allyl – Cl – H] . Elemental analysis (%): calc.: C, 32.04; H, 4.16.
1
⁄
found: C, 31.82; H, 4.25. Overlapping signals with major isomer.
2
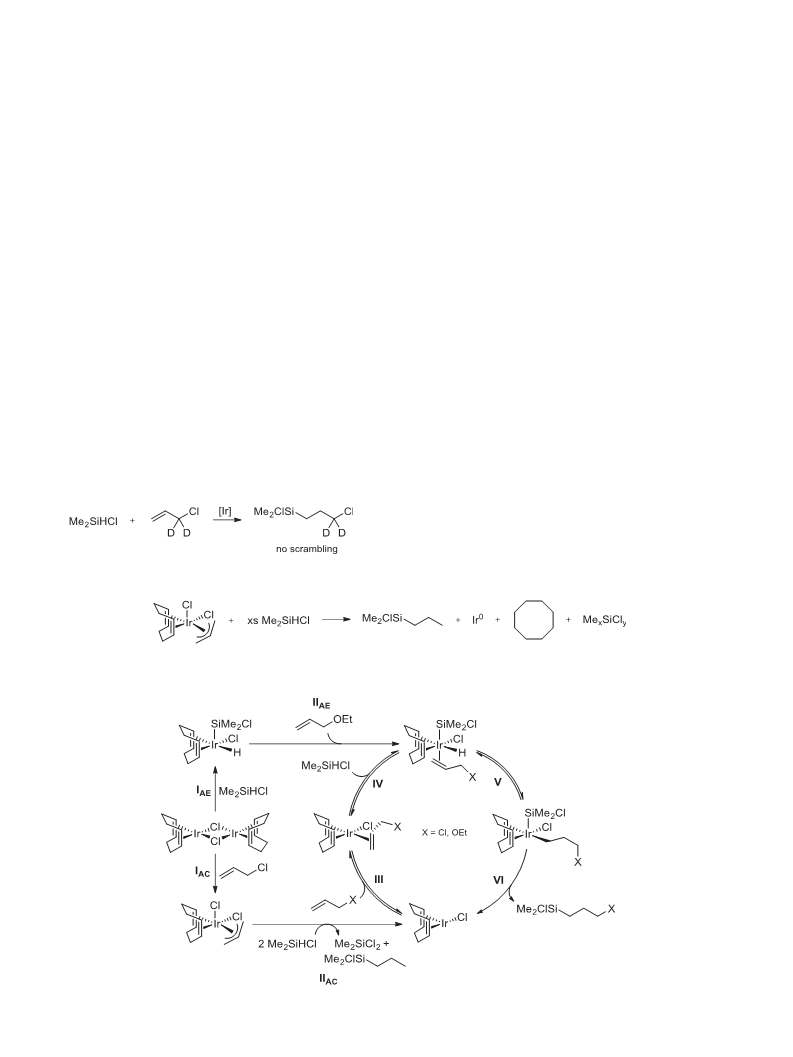
.3.4. Reaction of [Ir(allyl)(Cl)
In an NMR tube equipped with a J. Young valve, 22.2 mg
Ir(allyl)(Cl) (COD)] (53.8 mol, 1.0 equiv.) is dissolved in 1.6 mL
and 47.4 L Me SiHCl (40.8 mg, 431 mol, 8.0 equiv.) in
Cl is added to the reaction mixture. CHCl is used as
2 2
(COD)] and Me SiHCl
2
2
.3. Reactivity studies
[
CD
1
2
l
2 2
.3.1. Reaction of [{IrCl(COD)} ] and Me SiHCl
2
Cl
2
l
2
l
NMR-scale: In an NMR tube equipped with a J. Young valve,
.10 mg [{IrCl(COD)} (12.1 mol, 1.0 equiv.) is dissolved in
.5 mL CD Cl and 21.4 Me SiHCl (18.3 mg, 193 mol,
6 equiv.) is added to the reaction mixture. CHCl is used as inter-
.0 mL CD
2
2
3
8
0
1
2
]
l
L
internal standard for NMR analysis.
2
2
l
2
l
3
nal standard for NMR analysis.
TEM: 153 mg [{IrCl(COD)} ] (228
in 10 mL CH Cl and 401 Me
6 equiv.) are added to the reaction mixture. After stirring for
0 min at room temperature, the volatiles are removed at reduced
3. Results and discussion
2
l
mol, 1.0 equiv.) is dissolved
2
2
l
L
2
SiHCl (345 mg, 3.65 mmol,
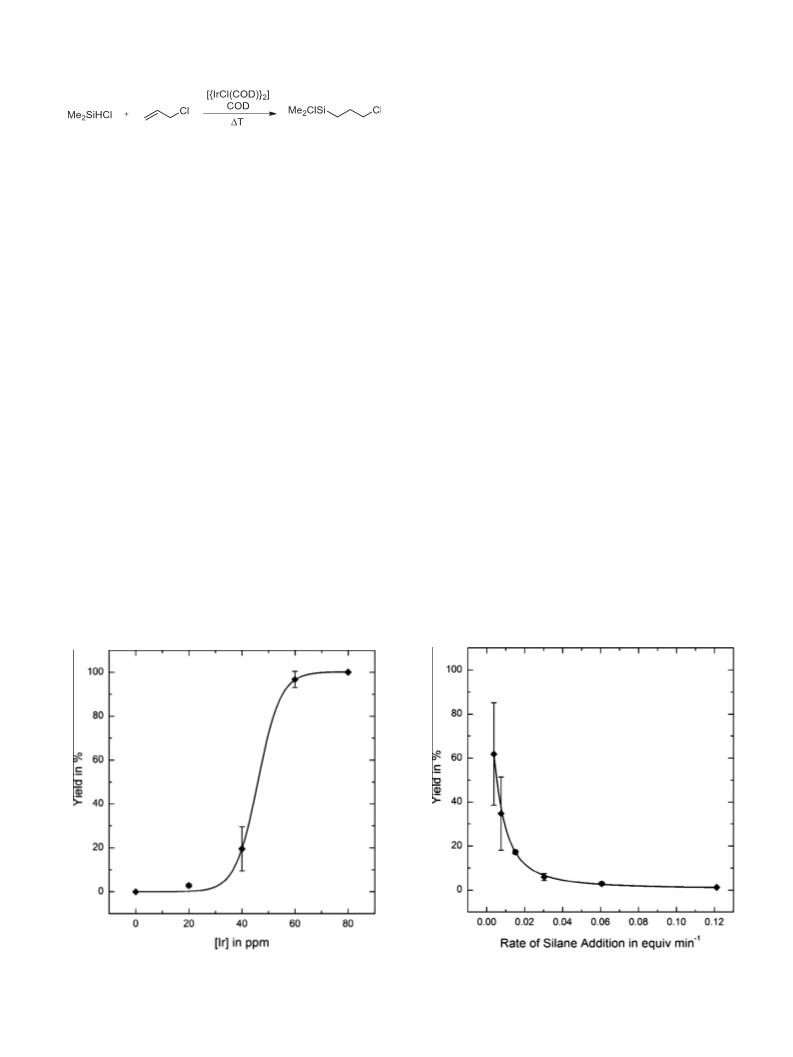
3.1. Influence of catalyst/co-catalyst concentration and the rate of
silane addition on catalytic performance
1
3
pressure and the solid is dried at 200 °C for 5 h. For TEM analysis,
the particles are suspended in ethanol.
The commonly used model system in iridium-catalyzed
hydrosilylation of allyl compounds is the reaction of allyl chloride
UV–Vis: 2.45 mg [{IrCl(COD)}
solved in 23 mL CH Cl and 1.5 mL of the solution is transferred
to a 1 cm UV–Vis cuvette for UV–Vis analysis. The solutions are
combined and 6.42 L Me SiHCl (5.52 mg, 58.3 mol, 16 equiv.)
is added. Again, 1.5 mL of the solution is transferred to a 1 cm
UV–Vis cuvette for UV–Vis analysis.
2
] (3.65
l
mol, 1.0 equiv.) is dis-
2 2
and Me SiHCl, with [{IrCl(COD)} ] and cyclooctadiene (COD) as
catalyst/co-catalyst combination, to yield chloro(3-chloropropyl)
dimethylsilane (Scheme 2) [11–22].
Typically, the silane (1.0 equiv.) is slowly added to a mixture of
the allyl compound (1.0 equiv.), the catalyst and the co-catalyst at
40 °C under neat conditions. Interestingly, three parameters differ
2
2
l
2
l

 Riener, Korbinian
Riener, Korbinian
 Meister, Teresa K.
Meister, Teresa K.
 Gigler, Peter
Gigler, Peter
 Herrmann, Wolfgang A.
Herrmann, Wolfgang A.
 Kühn, Fritz E.
Kühn, Fritz E.