Organometallics
Article
1
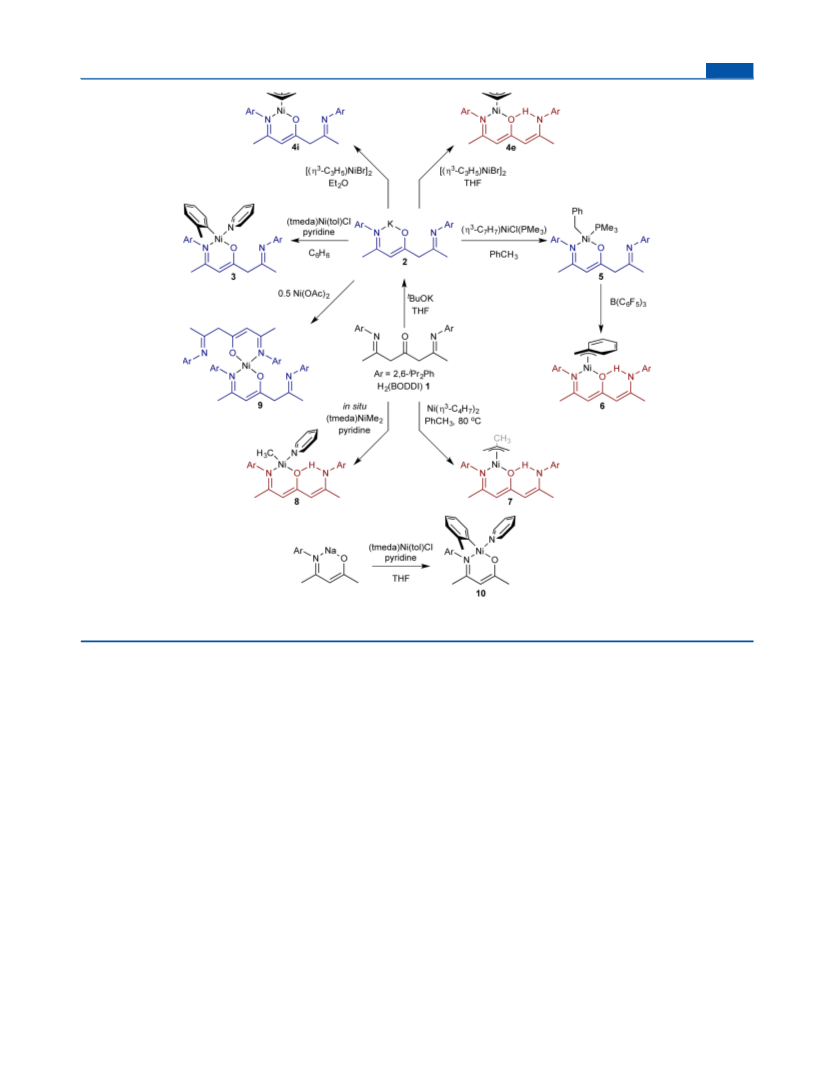
Synthesis of C H N OPNi, H(BODII)Ni(η -C H )PMe (5). A
was concentrated in vacuo to less than 1 mL of total volume. A 5 mL
amount of pentane was added to precipitate out an orange powder.
The mother liquor was decanted out, and the solid was washed by
another 5 mL of pentane (118.7 mg, 65% yield). Large X-ray quality
brown crystals were obtained by slow diffusion of pentane into a
41
59
2
7
7
3
3
solution of (PMe )Ni(η -C H )Cl (29.2 mg, 0.1 mmol) in toluene
2 mL) was added dropwise to a suspension of the deprotonated
3
7
7
(
ligand 2 (49.9 mg, 0.1 mmol) in toluene (2 mL) at ambient
temperature. The suspension became a deep brown solution, and
within 10 min KCl precipitation was evident. The mixture was stirred
at ambient temperature for 1.5 h. The mixture was then filtered, and all
the volatiles of the filtrate were removed in vacuo to yield a brown
solid. The solid was used without further purification. H NMR (C D ,
00 MHz): δ 7.90 (s, 2H), 7.23−6.98 (m, 9H), 5.36 (s, 1H), 4.09
sep, 2H, J = 6.5 Hz), 3.27 (s, 2H), 3.09 (sep, 2H, J = 6.5 Hz),
1
concentrated Et O solution of 8 at −30 °C. H NMR (C D , 500
2
6
6
3
MHz): δ 9.25 (s, 1H), 8.88 (d, 2H, J = 5.0 Hz), 7.13 (s, 3H), 7.02
(m, 1H), 6.88 (s, 1H), 6.87 (s, 1H), 6.25 (t, 1H, J = 8.0 Hz), 6.03
HH
3
HH
1
3
3
6
6
(t, 2H, J = 7.0 Hz), 5.23 (1, 1H), 4.95 (s, 1H), 4.17 (sep, 2H, J
H
H
H
H
3
3
5
(
1
1
= 7.0 Hz), 2.99 (sep, 2H, J = 6.5 Hz) 1.69 (d, 6H, J = 6.5 Hz),
HH HH
1.55 (s, 3H), 1.34 (s, 3H), 1.20 (d, 6H, J = 7.0 Hz), 0.99 (d, 6H,
JHH = 6.5 Hz), 0.75 (d, 6H, J = 6.5 Hz), −0.76 (s, 3H) ppm.
3
3
3
HH
HH
HH
3
3
3
.71 (s, 3H), 1.57 (s, 3H), 1.41 (d, 6H, J = 6.5 Hz), 1.29 (m, 12H),
.22 (d, 6H, J = 6.5 Hz), 0.81 (s, 2H), 0.64 (d, 9H, JPH = 8.5 Hz)
HH
HH
3
2
13
1
HH
C{ H} NMR (C D , 125 MHz): δ 176.03(O−C), 163.08 (NC),
6
6
13
1
ppm. C{ H} NMR (C D , 125 MHz): δ 173.86 (O−C), 167.47
6
6
151.67 (N−C), 150.90, 148.88, 147.27, 141.50, 135.25, 134.82, 127.20,
(
NC), 159.91 (NC), 146.83, 141.33, 136.23, 129.31, 128.20,
1
2
24.86, 123.29, 123.14, 122.81, 97.85 (βC), 96.19 (βC), 28.43,
1
2
2
25.46, 123.45, 123.37, 123.04, 122.94, 98.03 (βC), 53.53 (−βC),
7.86, 27.69, 25.53, 24.39, 24.03, 24.00, 23.97, 22.98, 22.33, 19.64,
1
8.06, 28.01, 25.11, 24.42, 24.19, 23.32, 22.85, 19.63, 11.78 (d, J
=
PC
−4.77 ppm. Anal. Calcd (%) for C H N ONi: C, 72.55; H, 8.39; N,
37
51
3
2
31
1
5.1 Hz), 9.35 (d, J = 31.3 Hz) ppm. P{ H} NMR (C D , 161
PC
6
6
6.86. Found: C, 72.14; H, 8.26; N, 6.59.
Synthesis of C H N O Ni, [H(BODII)] Ni (9). A solution of
MHz): δ −13.60 ppm.
3
62 86
4
2
2
Synthesis of C H N ONi, H(BODEI)Ni(η -C H ) (6). Without
purification, crude product 5 and B(C F ) (51.3 mg, 0.1 mmol)
were dissolved in toluene (2 mL) separately and cooled at −30 °C.
B(C F ) solution was transferred to the solution of complex 5
dropwise. After being stirred at ambient temperature for 30 min, the
volatile of the filtrate was removed in vacuo to get a brown solid.
Pentane (2 mL) was added to the crude product, and the oil-like
brown precipitate stayed insoluble. After being stirred in pentane for 1
h, the precipitate was filtered out and the volatile of the filtrate was
removed in vacuo to yield 6 as a deep red solid. The product was
dissolved in minimal diethyl ether and cooled to −30 °C for several
days to yield X-ray quality dark brown crystals (29.7 mg, 51% overall
3
8
50
2
7 7
deprotonated ligand 2 (49.9 mg, 0.1 mmol) in THF (1 mL) was
6
5 3
added to a suspension of the Ni(OAc) (17.6 mg, 0.1 mmol) in THF
2
(2 mL) at ambient temperature. The suspension became a brown
6
5 3
solution, and within 10 min KCl precipitation was evident. The
mixture was stirred at ambient temperature for 1 h. All the volatiles
were removed in vacuo, and n-pentane (5 mL) was added to the crude
mixture. After stirring in n-pentane for 1 h, the mixture was filtered and
the filtrate was concentrated and cooled to −30 °C overnight to yield
1
green plate crystals (66.5 mg, 68% yield). H NMR (C D , 500 MHz):
6
6
3
3
δ 7.14−7.08 (m, 6H), 7.04 (d, 2H, J = 10.0 Hz), 9.69 (d, 4H, J
HH
HH
3
=
9.5 Hz), 5.43 (s, 2H), 4.01 (sep, 4H, J = 8.5 Hz), 2.91 (sep, 4H,
HH
3
3
JHH = 8.5 Hz), 2.62 (s, 4H), 1.70 (d, 12H, J = 8.5 Hz), 1.43 (s,
H), 1.36 (s, 6H), 1.21 (d, 12H, J = 8.5 Hz), 1.17 (d, 24H, J
8.5 Hz) ppm. C{ H} NMR (C D , 125 MHz): δ 173.16 (O−C),
1
3
HH
yield). H NMR (C D , 500 MHz): δ 9.55 (s, 1H), 7.12 (t, 1H, J
.3 Hz) 7.11−7.07 (m, 5H), 6.95−6.93 (m, 3H), 6.80−6.78 (m, 3H),
.95 (s, 1H), 4.84 (s, 1H), 3.92 (sep, 2H, J = 7.0 Hz), 3.25 (sep,
=
6
6
HH
3
3
6
=
HH
HH
7
4
2
3
6
13
1
3
6 6
HH
3
3
166.97 (NC), 166.25 (NC), 146.58, 144.33, 141.94, 136.18,
125.22, 123.62, 123.24, 123.06, 98.47 (βC), 49.73 (−βC), 34.15,
28.55, 27.99, 24.30, 23.64, 23.34, 22.78, 22.43, 20.35, 13.98 ppm. Anal.
Calcd (%) for C H N O Ni: C, 76.14; H, 8.86; N, 5.73. Found: C,
H, J = 7.0 Hz), 1.48 (s, 3H), 1.41 (d, 6H, J = 7.0 Hz), 1.35 (s,
HH
HH
3
3
H), 1.15 (d, 6H, J = 7.0 Hz), 1.08 (d, 6H, J = 6.5 Hz), 1.01 (d,
HH
HH
3
13
1
H, J = 7.0 Hz), 0.67 (s, 2H) ppm. C{ H} NMR (C D , 125
HH
6
6
62
86
4
2
MHz): δ 176.34 (OC), 162.51 (NC), 152.76 (N−C), 151.94,
7
5.83; H, 8.81; N, 5.72.
Synthesis of C H N ONi, (BKI)Ni(o-tolyl)(py) (10). To a
1
1
2
47.64, 140.10, 135.03, 132.88, 126.33, 124.69, 123.38, 123.26, 115.79,
07.69, 96.29 (βC), 96.19 (βC), 27.89, 27.77, 27.12, 24.01, 23.99,
3.94, 23.39, 19.87 ppm. Anal. Calcd (%) for C H N ONi: C, 74.88;
2
9
36
2
suspension of NaH (35.0 mg, 1.4 mmol) in THF (2 mL) was
added dropwise a solution of BKI ligand (259 mg, 1.0 mmol) in THF
(3 mL) at ambient temperature. After the gas evolution ceased, the
solution was stirred for another 30 min and filtered. A mixture of
3
8
50
2
H, 8.27; N, 4.60. Found: C, 70.49; H, 7.75; N, 4.35.
3
Synthesis of C H N ONi, H(BODEI)Ni(η -C H ) (7). 1 (92.1 mg,
3
5
51
2
4 8
3
0
.2 mmol) and Ni(η -methallyl) (37.0 mg, 0.22 mmol) were dissolved
in toluene (2 mL), heated to 80 °C, and stirred overnight. Afterward,
some Ni black precipitate was filtered out, and all the volatiles were
2
(tmeda)Ni(o-tolyl)Cl (306 mg, 1.0 mmol) and pyridine (0.1 mL) in
0
THF (1 mL) was transferred to the solution at ambient temperature.
After being stirred for 1 h, all the volatiles of the mixture were removed
in vacuo. Hexanes (5 mL) was added to the crude mixture, and the
mixture was stirred for another 30 min. The mixture was filtered, and
the filtrate was concentrated and cooled to −30 °C overnight to yield
removed in vacuo to yield 7 as an orange-yellow solid (92.9 mg, 81%
yield). X-ray quality, needlelike, yellow crystals were obtained by
1
cooling of a concentrated Et O solution of 7. H NMR (C D , 500
2
6
6
MHz): δ 10.31 (s, 1H), 7.13−7.01 (m, 6H), 5.19 (s, 1H), 5.04 (s, 1H),
1
3
3
orange needlelike crystals (40.6 mg, 42% yield). H NMR (C D , 500
6
6
4
3
1
4
.10 (sep, 1H, J = 6.5 Hz), 3.45 (sep, 1H, J = 7.0 Hz), 3.38−
HH
HH
3
3
4
MHz): δ 8.58 (d, 2H, J = 5.5 Hz), 7.27 (d, 1H, J = 7.5 Hz),
.30 (m, 2H) 2.58 (d, 1H, J = 2.0 Hz), 2.52 (s, 1H), 2.18 (s, 3H),
HH HH
HH
3
3
3
7.09 (t, 1H, J = 7.5 Hz), 6.73 (d, 1H, J = 7.5 Hz), 6.62−6.54
3 3
.53 (s, 3H), 1.43 (s, 3H), 1.32 (d, 3H, J = 6.5 Hz), 1.28−1.27 (m,
HH
HH
HH
3
13
1
(m, 4H), 6.41 (t, 1H, J = 7.5 Hz), 6.07 (t, 2H, J = 7.5 Hz), 5.20
H), 1.13−1.10 (m, 16H), 1.00 (d, 3H, J = 6.5 Hz) ppm. C{ H}
HH HH
HH
3
(
s, 1H), 4.61 (sep, 1H, J = 6.5 Hz), 3.75 (s, 3H), 2.88 (sep, 1H,
JHH = 7.0 Hz), 2.32 (d, 3H, J = 6.5 Hz) 1.95 (s, 3H), 1.51 (s, 3H),
.27 (d, 3H, J = 7.0 Hz), 1.04 (d, 3H, J = 7.0 Hz), 0.49 (d, 3H,
JHH = 6.5 Hz) ppm. C{ H} NMR (C D , 125 MHz): δ 176.13 (O−
NMR (C D , 125 MHz): δ 176.93 (O−C), 163.35 (NC),
HH
6
6
3
3
1
1
(
2
1
4
53.19(N−C), 151.79, 147.74, 147.55, 140.79, 139.47, 135.34,
24.65, 123.80, 123.49, 123.26, 123.17, 123.02, 96.56 (βC), 95.88
HH
3
3
1
HH HH
3
13
1
βC), 65.54, 57.49, 49.08, 28.29, 28.27, 27.93, 27.56, 24.90, 24.79,
6
6
4.39, 24.15, 24.02, 23.91, 23.56, 23.20, 22.40, 22.13, 22.00, 19.49,
5.23 ppm. Anal. Calcd (%) for C H N ONi: C, 73.30; H, 8.79; N,
C), 166.71 (NC), 151.83, 150.91, 148.09, 143.43, 141.41, 141.18,
135.81, 135.18, 125.28, 125.19, 123.25, 123.21, 122.84, 122.25, 121.77,
98.61 (βC), 28.66, 27.69, 25.79, 25.34, 24.76, 24.62, 24.36, 24.13,
35
51
2
.88. Found: C, 73.46; H, 8.75; N, 4.89.
Synthesis of C H N ONi, H(BODEI)Ni(Me)(py) (8). MeLi (0.625
22.85 ppm. Anal. Calcd (%) for C29H N ONi: C, 71.48; H, 7.45; N,
36 2
3
7 51 3
mL, 1.6 M in diethyl ether, 1 mmol) was added dropwise to a stirring
suspension of (tmeda)Ni(OAc)2 (146.5 mg, 0.5 mmol) in a 9:1
toluene/THF mixture (5 mL) at −45 °C. The temperature was kept
below −25 °C. Once the green suspension turned into a brown-yellow
solution, a solution of 1 (138.2 mg, 0.3 mmol) in toluene (1 mL) was
added to the mixture at −25 °C followed by an addition of pyridine
5.75. Found: C, 71.34; H, 7.34; N, 5.76.
Synthesis of C43 OLiNi, Li(BODDI)Ni(o-tolyl)py (3Li). To a
solution of 3 (68.9 mg, 0.1 mmol) in Et O (1 mL) was added
dropwise a solution of LiHMDS·Et O (24.1 mg, 0.1 mmol) in Et O (2
mL) under stirring at ambient temperature for an hour. Volatiles were
removed in vacuo to yield a red solid. The crude product was
H N
54 3
2
2
2
(
0.2 mL, 2.4 mmol). The solution was then heated to 45 °C and
precipitated from a mixture of Et O (0.1 mL) and pentane (1 mL)
2
stirred for 2 days. Afterward, the mixture was filtered, and the filtrate
cooled to −30 °C overnight, yielding 3Li as a red precipitate. The
C
Organometallics XXXX, XXX, XXX−XXX

 Chiu, Hsin-Chun
Chiu, Hsin-Chun
 Pearce, Adam J.
Pearce, Adam J.
 Dunn, Peter L.
Dunn, Peter L.
 Cramer, Christopher J.
Cramer, Christopher J.
 Tonks, Ian A.
Tonks, Ian A.