R. Khusnutdinov, A. Bayguzina, R. Aminov, and U. Dzhemilev
Vol 000
carrier gas, temperature programming from 40 to 300°C at 8°C/min,
evaporation temperature 280°C, temperature of the ion source
Acknowledgements. This work was financially supported by the
Russian Foundation for Basic Research (grant no. 12-03-00183)
and a Grant of the RF President (Sci. Sch. À 2136.2014.3).
2
00°C, and ionization energy 70 eV). Chromatographic analysis
was carried out on a Shimadzu GC-9A, GC-2014 instrument
2 m × 3 mm column, silicone SE-30 (5%) on Chromaton
[
REFERENCES
N-AW-hexamethyldisiloxane as the stationary phase, tempera-
ture programming from 50 to 270°С at 8°C/min, helium as the
carrier gas (47 mL/min)].
[
[
1] Tsuji, Y.; Huh, K. T.; Watanabe Y. J. Org Chem 1987, 52,
1673.
Commercially, available substituted anilines, 1,2-ethanediol,
2] Zheng, J. C.; Liu, J.; Tan, W.; Shi, L.; Sun, Q. Chin J Catal
1
,2-propanediol, 1,2-butanediol (Acros), carbon tetrachloride (re-
2008, 29, 1199.
[3] Hu, Y.; Lü, W.; Liu, D.; Liu, J.; Shi, L.; Sun, Q. J Natural Gas
Chem 2009, 18, 445.
agent grade, component-reactive), were used as the starting
agents with distillation before use.
Iron containing catalysts FeCl
FeCl •4H O, Fe(C , Fe(acac) , Fe(OAc)
were recrystallized and dried in a vacuum desiccator before use.
[4] Campanati, M.; Franceschini, S.; Piccolo, O.; Vaccari, A.
3
•6H
2
O (Sigma-Aldrich), FeCl
3
,
J Catal 2005, 232, 1.
2
2
5
H
5
)
2
3
2
, and Fe (CO) (Acros)
2
9
[5] (a) Tsuji, Y.; Huh, K. T.; Watanabe Y. Terahedron Lett 1986,
27, 377; (b) Tursky, M.; Lorentz-Petersen, L. L. R.; Olsen, L. B.; Madsen, R.
Org Biomol Chem 2010, 8, 5576.
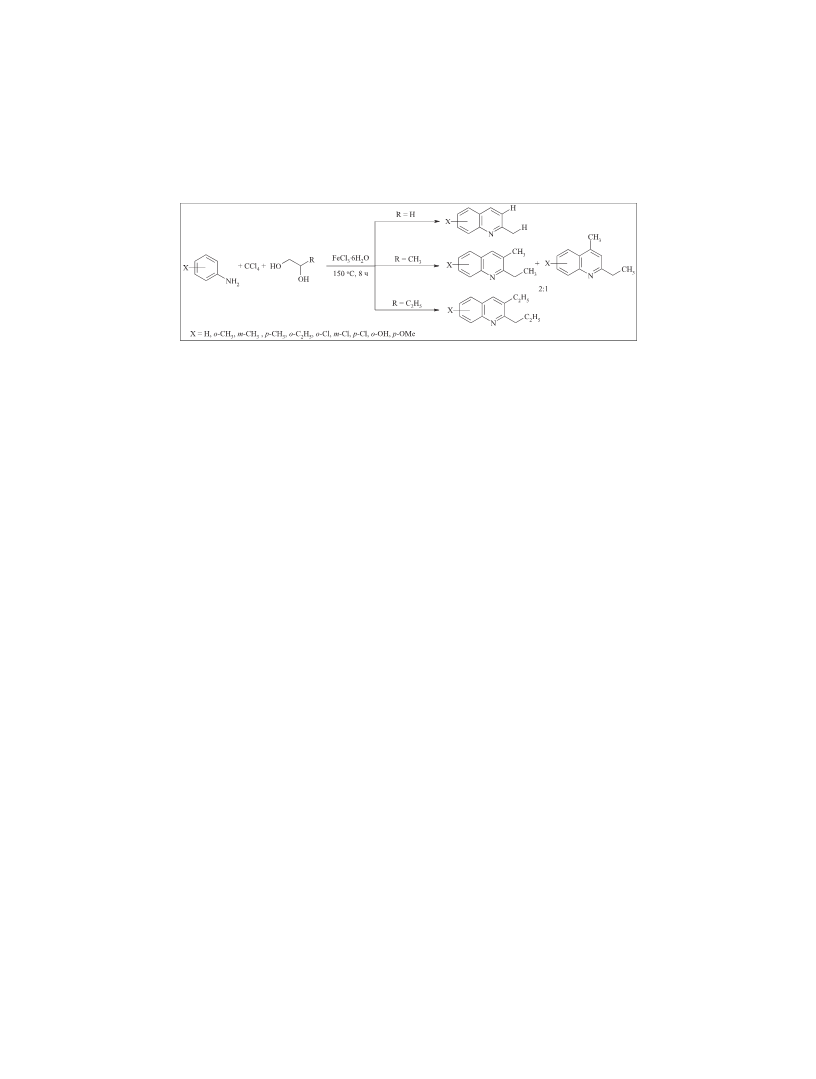
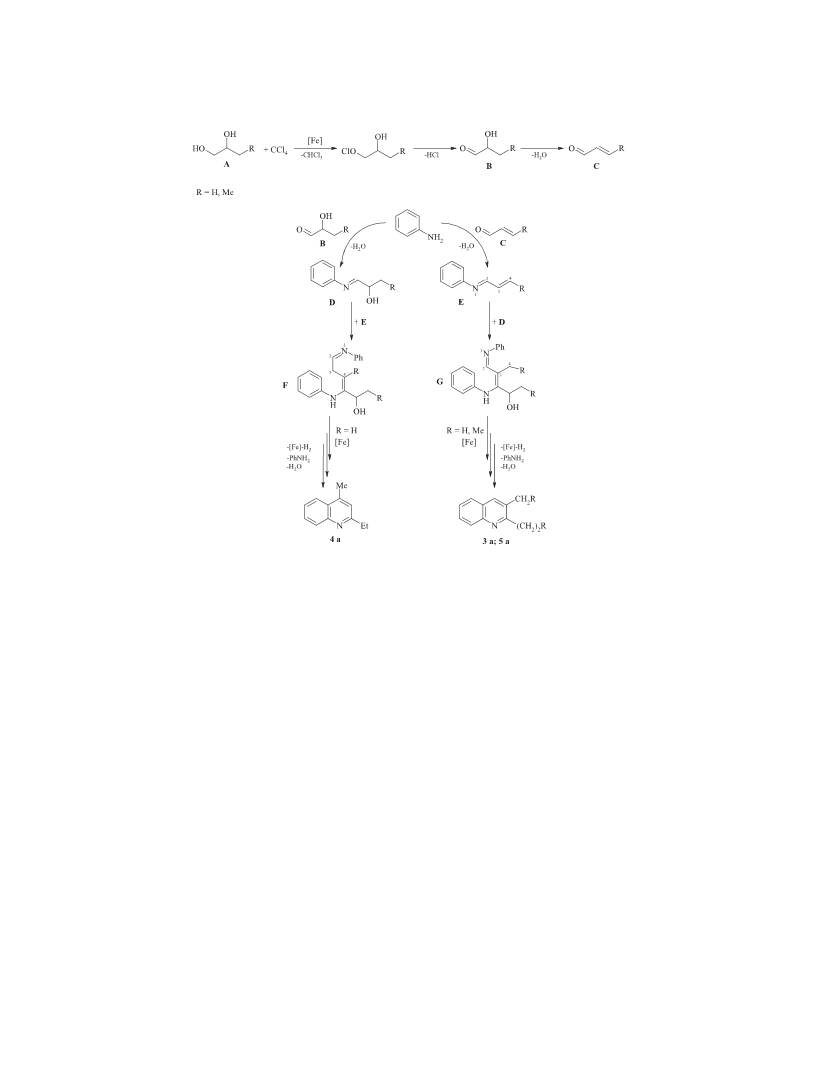
The synthesis of quinolines 2-5. The reactions were carried
out in a glass ampoule (V = 10 mL), placed in a stainless steel
microautoclaves (V = 17 mL) under constant stirring and
controlled heating.
[
6] Khusnutdinov, R. I.; Bayguzina, A. R.; Aminov, R. I.;
Dzhemilev, U. M. Russ J Org Chem 2012, 48, 690.
7] Sridharan, V.; Avendano, C.; Menendez, J. C. Tetrahedron
007, 63, 673.
[8] Watanabe, Y.; Shim, S. C.; Mitsudo T. Bull Chem Soc Jpn
981, 54, 3460.
9] AIST: RIO-DB Spectral Database for Organic Compounds
[
2
1
The ampoule was charged with FeCl · 6H O (FeCl3,
3
2
FeCl
2
· 4H
2
O, Fe(C
5
H
5
)
2
,
Fe(acac)
3
,
2 2 9
Fe(OAc) , Fe (CO) )
[
(
0.02 mmol), аniline (2 mmol), 1,2-diol (8 mmol), and carbon
SDBS
tetrachloride (4 mmol) in an argon flow. The sealed ampoule
[10] Zhang, Z.; Tan, J.; Wang, Z. Org Lett 2008, 10, 173.
was placed in an autoclave. The autoclave was air-tightly closed
[11] Uchiyama, K.; Ono, A.; Hayashi, Y.; Narasaka, K. Bull Chem
о
and heated at 150 С for 8 h under continuous stirring. After
Soc Jpn, 1998, 71, 2945.
12] Shchepetov, A. E.; Khusnutdinov, R. A.; Bugay, D. E.;
completion of the reaction, the autoclave was cooled to room
temperature, the ampoule was opened, and the reaction mixture
was treated with diluted (10%) hydrochloric acid. The water
layer was separated, neutralized with 10% solution of sodium hy-
droxide, and extracted with dichloromethane. The organic layer
was filtered, and the solvent was distilled off. The residue was
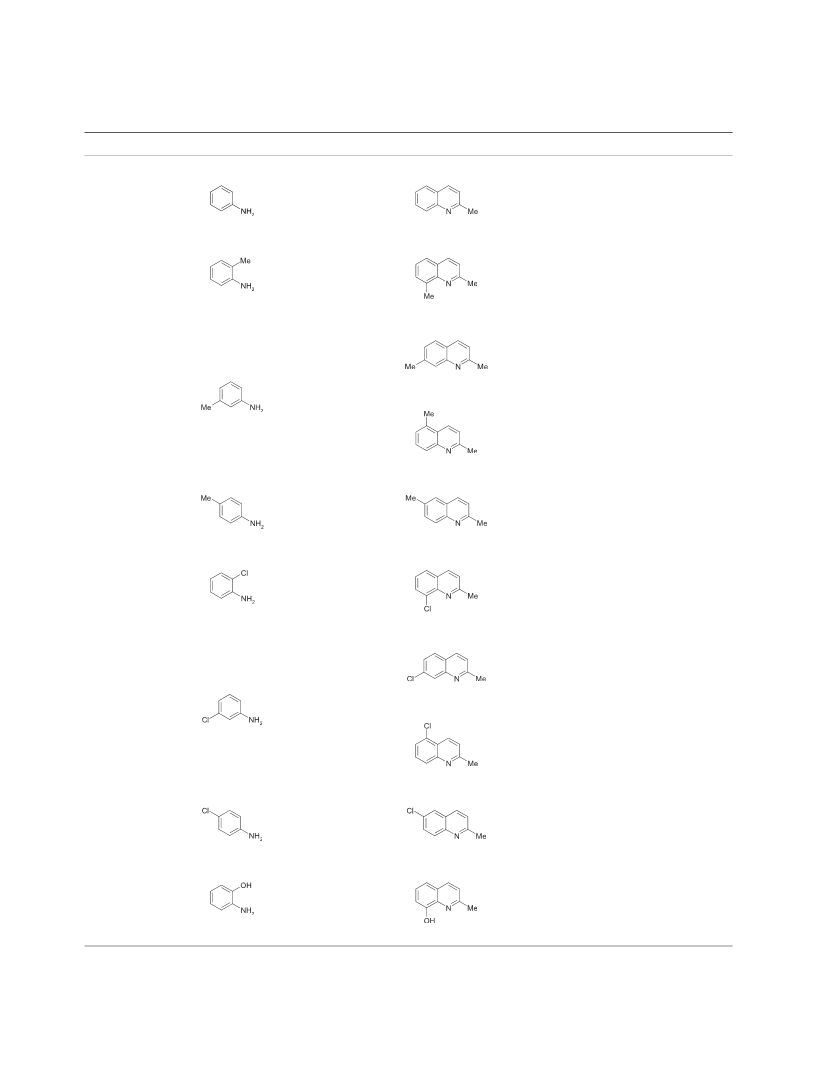
distilled in a vacuum or recrystallized from hexane (Table 3).
[
Rahmankulov, D. L. Bashkirskii Khimicheskii Zhurnal, 2007, 14, 30.
[13] Agronomov, A. E. Izbrannye glavy organicheskoi khimii,
Ucheb. posobie dlya vuzov [Selected Sections of Organic Chemistry.
Study Book for Higher School] 2-е ed., revised. and suppl. —Moscow.:
Khimiya, 1990. — 560 pp.
[14] Khusnutdinov, R. I.; Shcadneva, N. A.; Bayguzina, A. R.;
Lavrenteva, Y. Y.; Dzhemilev, U.M. Russ Chem Bull 2002, 11,
2074.
Detailed NMR spectra are given in the Supplementary Data file.
Journal of Heterocyclic Chemistry
DOI 10.1002/jhet

 Khusnutdinov, Ravil
Khusnutdinov, Ravil
 Bayguzina, Alfiya
Bayguzina, Alfiya
 Aminov, Rishat
Aminov, Rishat
 Dzhemilev, Usein
Dzhemilev, Usein