1
10
W. Turek, A. Krowiak / Applied Catalysis A: General 417–418 (2012) 102–110
of propene according to E1cB mechanism and dehydrogenation to
acetone).
Therefore, the possibility of dehydration of isopropyl alcohol to
diisopropyl ether is determined by the distance between adjacent
cations, their accessibility for reactants and acidity.
catalyst contains Brønsted acid centres, the reaction products
are mainly propene and diisopropyl ether. If the catalyst con-
tains redox centres and it does not contain Brønsted acid centres,
the main reaction product is acetone. For catalysts with Lewis
acid and basic centres the amounts of propene and acetone are
similar.
4
. Conclusions
7) The conditions of diisopropyl ether formation over Lewis acid
centres were determined. The possibility of this reaction is
determined by:
The results presented above show that the test reaction of iso-
propyl alcohol conversion can be used not only to determine the
type of centres over which the reaction proceeds, but also to eval-
uate the strength of active centres.
- the distance between adjacent cations (Lewis acid centres). If the
distance is higher, the probability of dehydration to diisopropyl
ether decreases,
1
) It was demonstrated that Brønsted and Lewis acid centres in
oxide catalysts can be distinguished on the basis of catalytic
activity in the reaction of dehydration to propene. It is possi-
ble because the dehydration to propene proceeds over Brønsted
acid centres according to a different mechanism than over Lewis
acid centres. As a result the rate of reaction over Brønsted acid
centres is significant at lower temperatures than over Lewis acid
centres.
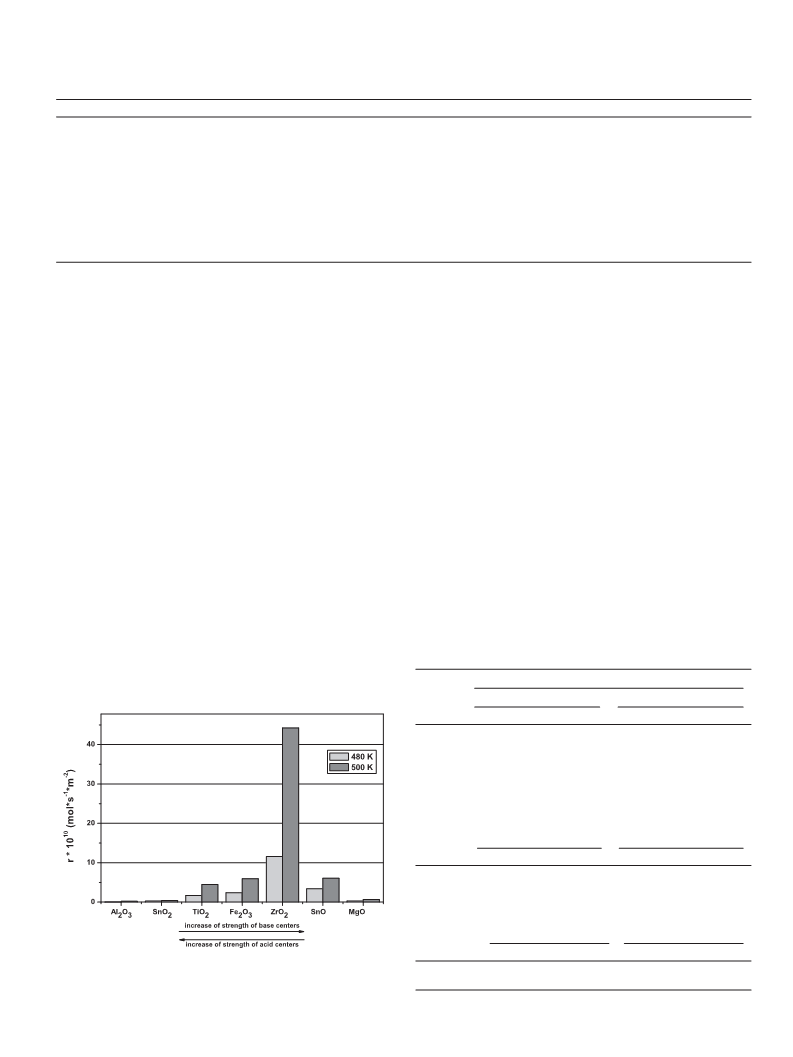
) It was also found that the rate of dehydration to propene is a
measure of the strength of acid and basic centres participating
in the reaction. In the case of catalysts with Lewis acid centres
the rate of dehydration increases when the strength of basic cen-
tres is higher. In the case of catalysts with Brønsted acid centres
the rate of dehydration increases with the strength of acid cen-
tres. It is important that the evaluation of the strength of Lewis
and Brønsted acid centres on the basis of the rate of dehydration
to propene does not depend on the amount of formed propene,
therefore it is independent of the number of active centres par-
ticipating in the reaction.
- accessibility of the centres for the substrate (isopropyl alcohol),
and
- strength of acid centres and adjacent basic centres, which can
interact with alcohol molecules chemisorbed at acid centres and
direct its transformation to propene or acetone.
References
[
[
1] V. Solinas, I. Ferino, Catal. Today 41 (1998) 179–189.
2] G. Ramis, P.F. Rossi, G. Busca, V. Lorenzelli, A. La Ginestra, P. Patrono, Langmuir
2
5
(1989) 917–923.
[3] D. Haffad, A. Chambellan, J.C. Lavalley, J. Mol. Catal. A: Chem. 168 (2001)
53–164.
1
[
4] F.M. Bautista, J.M. Campelo, A. Garcia, D. Luna, J.M. Marinas, A.A. Romero, M.T.
Siles, Catal. Today 78 (2003) 269–280.
[5] Y. Toda, T. Ohno, F. Hatayama, H. Miyata, Phys. Chem. Chem. Phys. 1 (1999)
615–1621.
6] S. Bernal, R. Garcia, F. Ramirez, J.M. Rodriguez-Izquierdo, Z. Phys. Chem. N. F.
38 (1983) 229–238.
1
[
1
[7] A. Ouqour, G. Coudurier, J.C. Vedrine, J. Chem. Soc. Faraday Trans. 89 (1993)
3151–3155.
[
8] J.M. Campelo, A. Garcia, J.F. Herencia, D. Luna, J.M. Marinas, A.A. Romero, J. Catal.
51 (1995) 307–314.
9] J.M. Vohs, M.A. Barteau, J. Phys. Chem. 95 (1991) 293–302.
1
3
) It was found that the value of activation energy of dehydration
to propene can indicate changes in the number of active cen-
tres during catalytic measurements. If at higher temperatures
an increase in the number of active centres occurs, the obtained
activation energy of dehydration to propene is comparatively
[
[10] M.A. Aramendía, V. Baráu, I.M. García, C. Jiménez, A. Marinas, J.M. Marinas, A.
Porras, F.J. Urbano, Appl. Catal. A: Gen. 184 (1999) 115–125.
[
[
11] H. Lauron-Pernot, F. Luck, J.M. Popa, Appl. Catal. 78 (1991) 213–225.
12] G.D. Yadav, A.D. Murkute, Langmuir 20 (2004) 11607–11619.
[13] G.D. Yadav, G.S. Pathre, J. Phys. Chem. A 109 (2005) 11080–11088.
[14] M.I. Zaki, N. Sheppard, J. Catal. 80 (1983) 114–122.
−1
high (over 150 kJ mol ). If higher temperature and the course
of reaction decrease the number of active centres the activation
[
[
15] G.A.H. Mekhemer, Appl. Catal. A: Gen. 275 (2004) 1–7.
16] J.A. Wang, X. Bokhimi, O. Novaro, T. Lopez, F. Tzompantzi, R. Gomez, J. Navarrete,
M.E. Llanos, E. Lopez-Salinas, J. Mol. Catal. A: Chem. 137 (1999) 239–252.
17] H. Knözinger, R. Kohne, J. Catal. 5 (1966) 264–272.
18] V.S. Lusvardi, M.A. Barteau, W.E. Farneth, J. Catal. 153 (1995) 41–43.
19] A. Davydov, Molecular Spectroscopy of Oxide Catalyst Surface, John Wiley &
Sons Ltd, Chichester, 2003.
−1
energy is very low (ca. 40 kJ mol ).
[
[
[
4
) It was proved that in the case of dehydrogenation to acetone not
only basic properties of the surface should be taken into account,
because the reaction can also proceed over redox centres. Dis-
tinction of these active centres can be achieved on the basis of
the rate of dehydrogenation to acetone at a given temperature.
In this reaction redox centres are active at lower temperatures
than basic centres.
) It was also discovered, that the rate of dehydrogenation to ace-
tone is a measure of the strength of active centres participating
in the reaction. If the catalyst contains Lewis basic centres of
low and medium strength, the rate of dehydrogenation to ace-
tone increases with the strength of basic sites, whilst for strongly
basic catalysts the rate of reaction is determined by the strength
of acid centres. If the catalysts contain redox centres, the rate of
reaction increases with their reducibility.
[
20] M.A. Aramendía, V. Baráu, C. Jiménez, J.M. Marinas, A. Porras, F.J. Urbano, J.
Catal. 161 (1996) 829–838.
[
21] V.E. Diyuk, L.N. Grishchenko, V.K. Yatsimirskii, Theor. Exp. Chem. 44 (2008)
331–337.
[22] L.A. Gambaro, L.E. Briand, Appl. Catal. A: Gen. 264 (2004) 151–159.
[
[
[
23] G. Busca, Phys. Chem. Chem. Phys. 1 (1999) 723–736.
5
24] G. Busca, V. Lorenzelli, G. Ramis, R. Willey, Langmuir 9 (1993) 1492–1499.
25] P.F. Rossi, G. Busca, V. Lorenzelli, M. Waqif, O. Saur, J.C. Lavalley, Langmuir 7
(1991) 2677–2681.
[
26] J.W. Mellor, Comprehensive Treatise on Inorganic and Theoretical Chemistry,
Longmans, Green and Co, London, New York, Toronto, 1957, vol. VII, p. 42, 128,
3
88, 399, vol. XIII, p. 805, vol. XIV, p. 580.
[27] T. Matsuda, F. Uchijima, H. Sakagami, N. Takahashi, Phys. Chem. Chem. Phys. 3
2001) 4430–4436.
(
[
28] F. Uchijima, T. Takagi, H. Itoh, T. Matsuda, N. Takahashi, Phys. Chem. Chem.
Phys. 2 (2000) 1077–1083.
6
) It was found that the selectivity to propene, diisopropyl ether
and acetone depends mainly on the type of active centres on the
surface. Brønsted acid centres are active in dehydration reac-
tions at lower temperatures than Lewis acid and basic centres.
Redox centres are active in dehydrogenation at lower tem-
peratures than acid and basic Lewis centres. Therefore, if the
[29] M. Misono, N. Mizuno, Y. Konishi, K. Katamura, A. Kasai, K. Sakata, T. Okuhara,
Y. Yoneda, Bull. Chem. Soc. Jpn. 55 (1982) 400–406.
[
[
30] T. Komara, M. Misono, Chem. Lett. (1983) 1177–1180.
31] A. Gervasini, A. Auroux, J. Catal. 131 (1990) 190–198.
[32] K.C. Wough, M. Bowker, R.W. Petts, H.D. Vandervell, J. O’Malley, Appl. Catal. 25
1986) 121–128.
33] L. Nondek, J. Sedlacek, J. Catal. 40 (1975) 34–39.
(
[

 Turek, Wincenty
Turek, Wincenty
 Krowiak, Agnieszka
Krowiak, Agnieszka