1
64
M. Ziolek et al. / Catalysis Today 187 (2012) 159–167
Scheme 1. Acetonylacetone transformation.
Table 2
catalysts which do not contain antimony species. The latter sam-
The results of acetonylacetone (AcOAc) cyclisation at 623 K.
ple ((Nb-V)-O25) catalyses the reaction mainly towards acetone
formation. Significant amounts of acetone are also produced on
Nb,(Sb-V)-O10. It is also very important to stress that both catalysts,
Nb,(Sb-V)-O10 mechanical mixture and (Nb-V)-O25 contain binary
oxides, antimony-vanadium and niobium-vanadium, respectively.
It seems that the presence of binary oxides of different composition
affects the activity in dehydrogenation of 2-propanol to acetone.
Moreover, the presence of antimony species considerably increases
the activity of the catalysts. This behaviour points out the role of
antimony species as a promoter in catalytic reactions in which elec-
tron transfer is involved. Such a reaction is dehydrogenation of
Catalyst
AcOAc
conv. [%]
Selectivity [%]
MCP/DMF
DMF*
MCP*
Blank experiment
Nb,Sb,V-O10
Nb,(Sb-V)-O10
0
7
9
8
7
–
–
–
45
40
49
24
55
60
51
76
1.22
1.50
1.04
3.17
(
(
Nb-Sb-V)-O10
Nb-V)-O25
*
MCP – 3-methyl-2-cyclopentenone; DMF – 2.5-dimethylfuran.
2
-propanol to acetone.
same for all samples. All, mechanical mixtures, ternary and binary
oxides exhibit both acidity (DMF production) and basicity (MCP
formation) on the surface. However, the selectivity differs signifi-
cantly depending on the chemical composition of catalysts. Among
the mechanical mixtures and ternary oxides (Nb-Sb-V)-O10 only
Nb,(Sb-V)-O10 containing binary Sb-V system presents slightly
higher basic than acidic properties (MCP/DMF = 1.5), whereas the
One more conclusion can be drawn from the results presented
in Fig. 6. Antimony species are necessary for the creation of Lewis
acid–base pairs responsible for the production of ether from 2-
propanol. Generation of such active pairs is enhanced by the
presence of the ternary oxide ((Nb-Sb-V)-O10) in comparison with
mechanical mixture of oxides.
The difference in catalytic surface properties is even much
more pronounced in the oxidation of methanol. This reaction is
more complex and reflects the presence of acid centres involved
in the chemisorption of methanol and active oxygen species. As
follows from kinetic studies, the rate determining step of the selec-
tive oxidation of methanol is the abstraction of hydrogen from
methyl group of methanol adsorbed. It is determined by the sur-
face properties. Methoxy species formed in the first step can be
adsorbed on oxygen vacancies in metal oxides (Lewis acid centres).
The methoxy species adsorbed on a terminal M O vacancy react
towards formaldehyde by a transfer of a methyl H atom to the
neighbouring M O bond [44–46]. The methoxy species adsorbed
on the bridged oxygen (M-O-M) vacancy sites are likely to form
dimethyl ether, dimethoxymethane (methylal), and methyl for-
mate, which requires the presence of additional Lewis acid sites
[44]. Thus, most authors working on the selective oxidation of
methanol considered a significant role of acidity and basic oxygen
species in the metal oxide catalysts. Finally, basicity is responsi-
ble for total oxidation of methanol to CO2 as proposed by Tatibouet
[47]. The total oxidation of methanol to carbon dioxide can proceed
also in the direct oxidation of methanol involving electrophilic oxy-
gen species (radical species) [48] or by the readsorption of HCHO
and its secondary reaction as proposed by Wachs and Kim [49]. In
this study a good correlation of the acetone formation in 2-propanol
dehydrogenation and CO2 generation in methanol oxidation sug-
gests the significant role of basic oxygen in the total oxidation of
methanol.
binary oxide (Nb VO25) exhibits significant domination of basicity
demonstrated by the MPC/DMF ratio of 3.17.
The strongly basic character of (Nb-V)-O25 was confirmed by
the results of 2-propanol decomposition carried out at 523 and
9
5
73 K (Fig. 6). The 2-propanol decomposition is a test reaction
for identification of acidic (Brønsted or Lewis) and/or basic prop-
erties of solids [42]. Dehydration of alcohol to propene and/or
di-isopropyl ether requires acidic centres (Lewis or BrØnsted),
whereas the dehydrogenation to acetone occurs on the basic sites.
Ether production requires the presence of pairs of Lewis acid–base
centres. Some authors [43] have reported that acetone formation
takes place on redox centres.
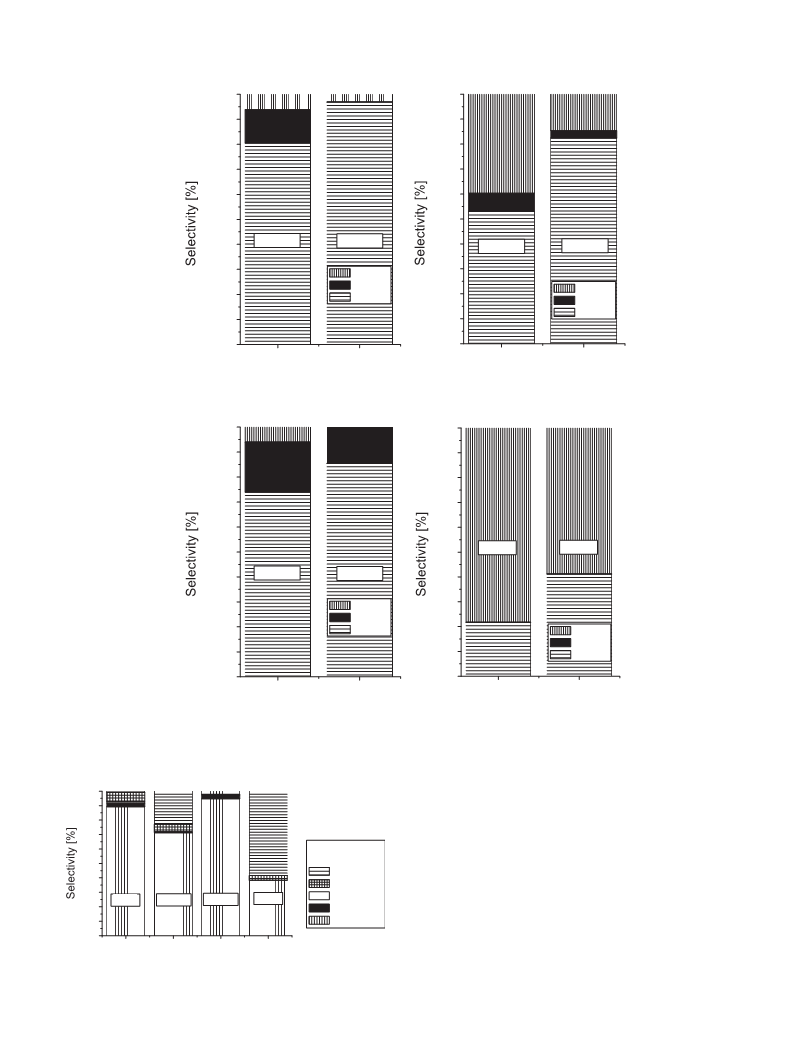
This test reaction allows estimation of surface properties of all
materials used. Diagrams in Fig. 6 contain values of 2-propanol con-
version at 523 and 573 K. It is clear that both mechanical mixture
of three metal oxides (Nb,Sb,V-O10) and ternary ((Nb-Sb-V)-O10)
oxide present almost the same very high activity in this reaction
demonstrated by 2-propanol conversion of ca 90% at 573 K. At the
lower temperature (523 K) the mechanical mixture of oxides is
less active than the ternary oxide. Especially interesting is a sig-
nificant difference in selectivity, which indicates differences in the
surface properties. On both materials the main product is propene,
which indicates highly acidic character of both samples. However,
on ternary oxide a considerable amount of ether is also produced,
which is not formed on mechanical mixture at 573 K. The formation
of ether, which requires the presence of pairs Lewis acid–base cen-
tres, proves the existence of such pairs on ternary oxide and their
higher activity than that in mixture of single oxides. Interestingly,
the activity of two other samples is much lower, especially of those
Fig. 7 reveals a very high selectivity to formaldehyde of all
catalysts containing three metals, but the highest selectivity to
this product is reached on ternary oxide ((Nb-Sb-V)-O10) and a

 Ziolek, Maria
Ziolek, Maria
 Golinska-Mazwa, Hanna
Golinska-Mazwa, Hanna
 Filipek, Elzbieta
Filipek, Elzbieta
 Piz, Mateusz
Piz, Mateusz