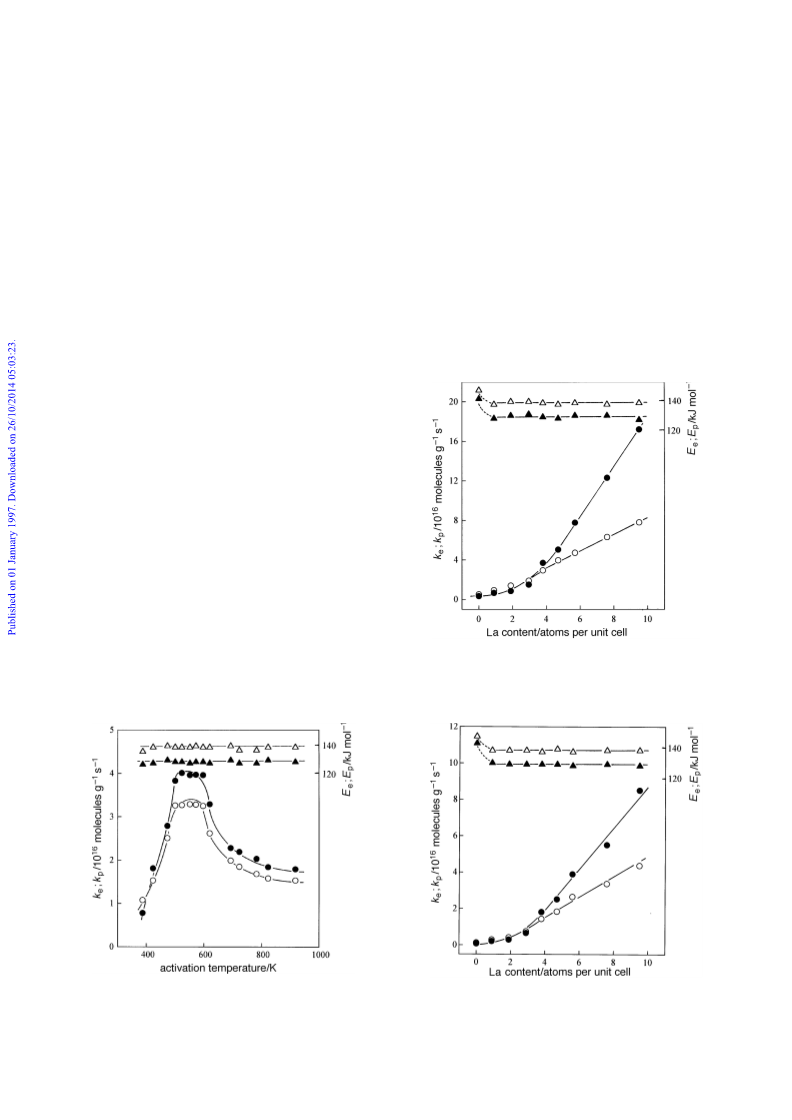
the same single-site ionic mechanism6,9 holds for LaY zeolites
as with HY zeolites, although Fig. 2 and 3 do not show the
single linear dependence of activity upon La content as pre-
viously observed for the H content of HY zeolites.10 Progres-
sive pyridine poisoning measurements showed that the speciÐc
activity of HY zeolite, expressed as (k ] k ) at 370 K, was
1.8 ] 1016 molecules g~1 s~1 for an accessible acid-site con-
centration of one per unit cell. We will consider whether this
Ðgure can be used to quantify accessible acid sites in the
present LaY catalysts, which were operating in identical reac-
tion conditions.
tion of hydroxy groups. This may be represented as:
La(H O)3` ] O ~ ] LaOH2` ] O H
(1)
2
z
z
However, dehyroxylation and an increase in the apparent
charge from 2] to 3] occurred13 at temperatures above 853
K, which can be represented as:
e
p
LaOH2` ] O H ] La3` ] O ~ ] H O
(2)
z
z
2
Clearly, such dehydroxylation would ultimately eliminate all
acidity, so that the second plateau of diminished activity is
either apparent, arising from the progressive slowing of acidity
loss, or arises from another acid centre stabilised by reaction
conditions. In this respect, bridged [LawOHwLa]5` ions
generated in the b-cages could account for the halving in both
acidity and activity:
The plots of k and k as a function of outgassing tem-
e
p
perature in Fig. 1 are similar in shape to that obtained by
Moscou14 for the concentration of acidic hydroxy groups in
the a-cages of rare-earth-exchanged Y zeolite as a function of
activation temperature. He found that the Ðrst plateau corre-
sponded to one acidic hydroxy group in an a-cage per rare
earth ion, whilst the second plateau occurred at half this
value. The mean value of (k ] k ) over the Ðrst plateau is
2LaOH2` ] 2O H ] [LawOHwLa]5`
z
] O H ] O ~ ] H O (3)
z
z
2
e
p
7.25 ] 1016 molecules g~1 s~1; division by 1.8 ] 1016 mol-
ecules g~1 s~1 (site per unit cell)~1 yields an acid-site concen-
tration of 4.03 per unit cell or 1.06 per La ion in LaY-3.8. For
the second plateau the average (k ] k ) for the last two
The low catalytic activity, yet high pyridine uptake, of samples
activated at high temperatures could be explained if pyridine
was sufficiently basic to form pyridinium ions with the H of
the bridged [LawOHwLa]5` formed in reaction (3):
e
p
experiments of 3.5 ] 1016 molecules g~1 s~1 yields 1.9 acid
sites per unit cell or 0.5 per La ion. Clearly, the use of the
speciÐc activity from HY zeolites gives results in accord with
those of Moscou,14 who used the techniques of gravimetry,
[LawOHwLa]5` ] Py ] [LawOwLa]4` ] PyH` (4)
We conclude that the activity for catalytic dehydration of
propan-2-ol to diisopropyl ether and propene at 370 K is a
valid measure of accessible BrÔnsted acid sites within the
supercages of LaY zeolite. Direct comparisons between
samples are possible because the activation energies for
product formation are independent of both the extent of
lanthanum exchange and the temperature of activation. Reac-
tion is considered to proceed by the same single-site mecha-
nism as with HY zeolites.6,9,10 However, progressive
poisoning measurements with pyridine measure the total con-
centration of acid sites throughout the zeolites, which gener-
ally exceeds that accessible to propan-2-ol. The fraction of
sites that is accessible depends on the activation temperature
and the nature of the lanthanum species within the b-cages
and hexagonal prisms.
Karl Fischer titration and reaction with LiAlH .
4
It is apposite to consider to what extent base poisoning
measurements assist in understanding the e†ect of lanthanum
content on catalytic activity shown in Fig. 2 and 3. From
these activities, expressed as (k ] k ), division by the constant
e
p
1.8 ] 10~16 molecules g~1 s~1 (site per unit cell)~1 yields the
acid-site concentrations given in Table 1. For catalysts acti-
vated at 523 K, there is reasonable agreement between acid-
site concentrations from calculation and from pyridine
poisoning with higher lanthanum contents. However, calcu-
lated values are considerably less than those from pyridine
poisoning at low lanthanum contents, where a high propor-
tion of sites are generated by aqueous treatment alone. For
catalysts activated at 823 K, the e†ect is even more pro-
nounced with the calculated concentration of acid-sites consis-
tently falling short of the lanthanum content. Nevertheless, the
mean ratio of calculated acid-site concentrations for catalysts
activated at 823 K to those for catalysts activated at 523 K is
0.49 for LaY-3.8 and catalysts of higher lanthanum concentra-
tion, but 0.35 for catalysts at lower concentrations. The ratio
of 0.49 is in accord with the e†ects of activation temperature
on the activity of LaY-3.8 given in Fig. 1. We suggest that the
failure of LaY zeolites to show a single linear increase in activ-
ity with lanthanum content can be attributed to di†erences in
both preferred location and activity between acid sites gener-
ated by aqueous treatment and by lanthanum exchange. At
low lanthanum content, a predominant and almost constant
concentration of acidity from aqueous treatment is located in
a-cages, whilst the majority of acid-sites arising from lantha-
num occupy b-cages. This leads to catalytic activity being
relatively insensitive to lanthanum content. At higher lantha-
num contents, accessible acidity from trivalent cation hydro-
lysis now predominates such that activity becomes much more
sensitive to lanthanum content.
References
1
P. A. Jacobs, Carboniogenic Activity of Zeolites, Elsevier,
Amsterdam, 1977, pp. 99È107.
2
R. Rudham and A. Stockwell, Catalysis, Vol. 1, Specialist Period-
ical Reports, The Chemical Society, London, 1977, pp. 111È114.
P. A. Jacobs, Catal. Rev. Sci. Eng., 1982, 24, 415.
S. J. Gentry and R. Rudham, J. Chem. Soc., Faraday T rans., 1,
1974, 70, 1685.
P. A. Jacobs, M. Tielen and J. B. Uytterhoeven, J. Catal., 1977,
50, 98.
R. Rudham and A. Stockwell, Catalysis by Zeolites, ed. B. Imelik,
C. Naccache, Y. Ben. Taarit, J. C. Vedrine, G. Cordurier and H.
Praliaud, Elsevier, Amsterdam, 1980 p. 113.
K. V. Topchieva and H. S. Tkhoang, Kinet. Catal. (U.S.S.R.),
1973, 14, 1320.
3
4
5
6
7
8
9
R. A. Schoonheydt, L. J. Vandamme, P. A. Jacobs and J. B. Uyt-
terhoeven, J. Catal., 1976, 43, 292.
R. Rudham and A. Stockwell, Acta. Phys. Chem. Szeged., 1978,
24, 281.
10 R. Rudham, A. I. Spiers and A. W. Winstanley, Zeolites, 1991, 11,
850.
11 L. V. C. Rees and T. Zuyi, Zeolites, 1986, 6, 201.
12 E. F. T. Lee and L. V. C. Rees, Zeolites, 1987, 7, 143.
13 E. F. T. Lee and L. V. C. Rees, Zeolites, 1987, 7, 545.
14 L. Moscou, Adv. Chem. Ser., 1971, no. 102, p. 337.
In explaining the generation of acid-sites, we follow Lee and
Rees12 who showed that calcination of LaY at the tem-
peratures of our Ðrst plateau led to the migration of lantha-
num ions into the b-cages and a reduction in the apparent
charge from 3] to 2], together with the irreversible forma-
Paper 6/07615G; Received 8th November, 1996
1448
J. Chem. Soc., Faraday T rans., 1997, V ol. 93

 Rudham, Robert
Rudham, Robert
 Spiers, Andrew I.
Spiers, Andrew I.