
Physical Chemistry Chemical Physics p. 1077 - 1083 (2000)
Update date:2022-08-11
Topics:
 Uchijima, Fumiko
Uchijima, Fumiko
 Takagi, Tomoaki
Takagi, Tomoaki
 Itoh, Hidenobu
Itoh, Hidenobu
 Matsuda, Takeshi
Matsuda, Takeshi
 Takahashi, Nobuo
Takahashi, Nobuo
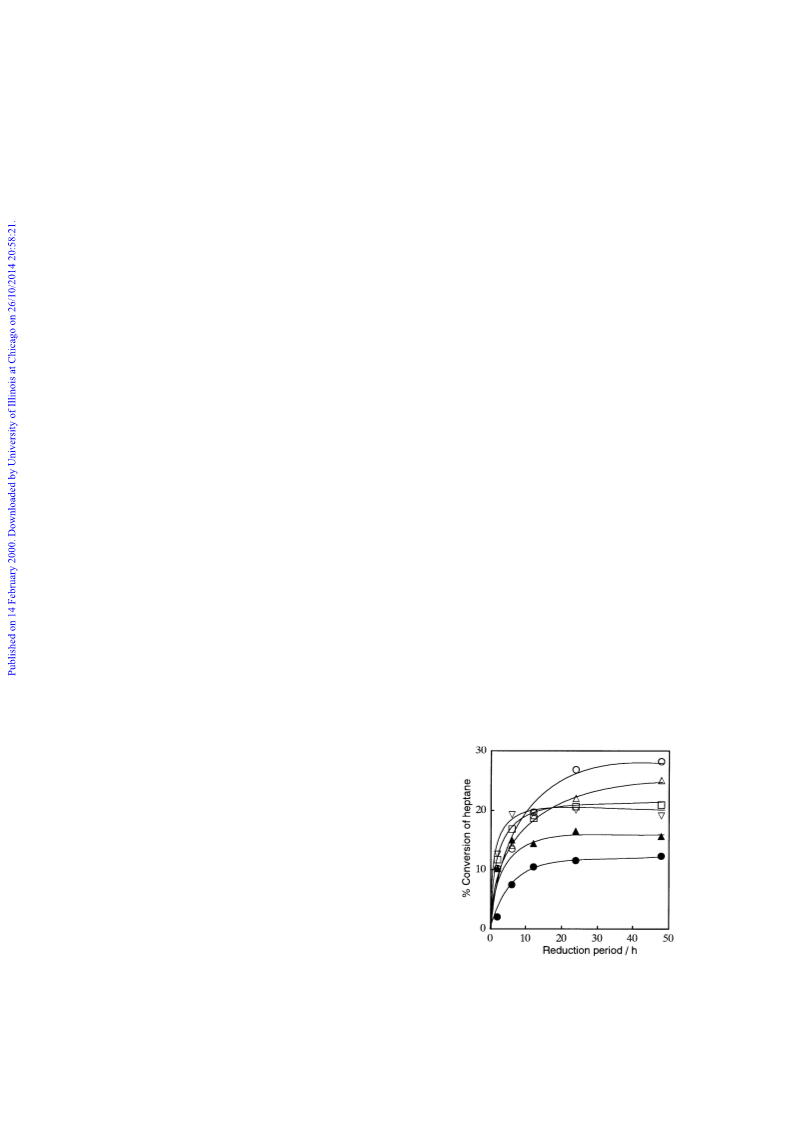
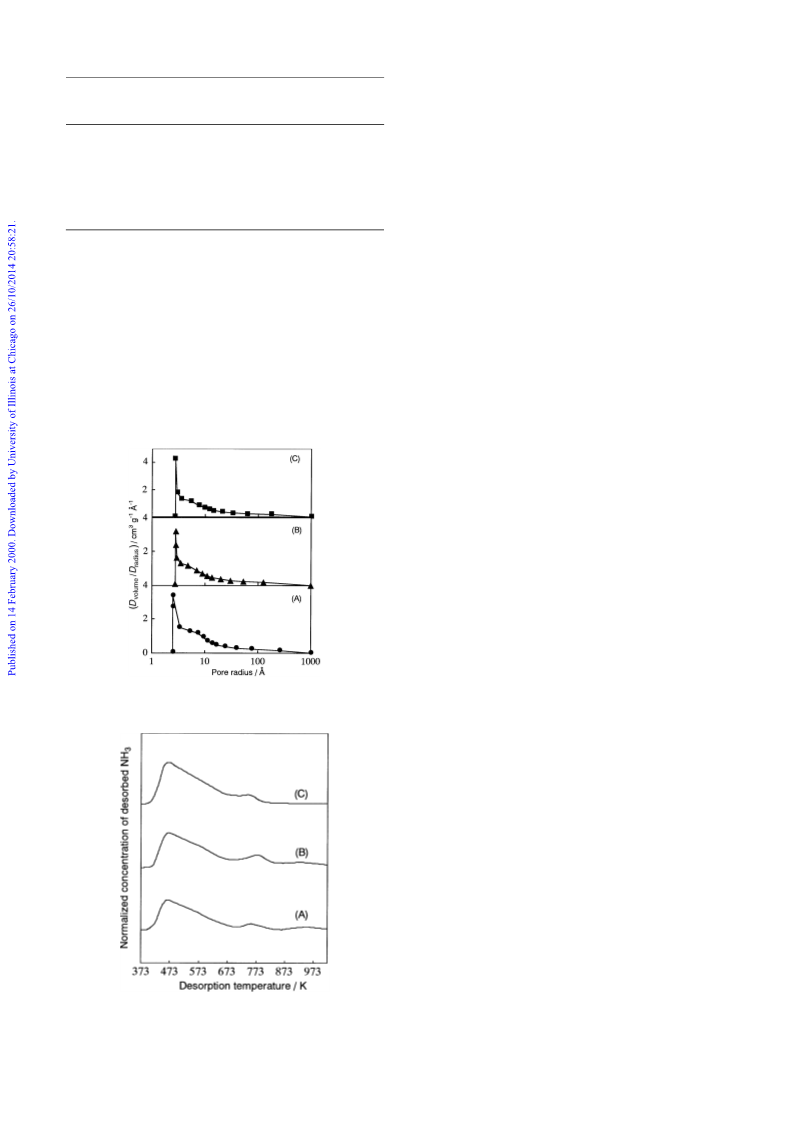
Effects of H2 reduction on the catalytic properties of MoO3 with Pt, Pd, Rh, Ir, or Ru for the conversions of heptane and propan-2-ol were studied. The catalytic activity of MoO3 with noble metal for the isomerization of heptane was strongly dependent on the period of H2 reduction at 623 K. This behavior was almost the same as that of MoO3 without noble metal. Among the catalysts tested, Pt/MoO3 was the most active for this reaction. The catalytic activities of MoO3 for dehydration and dehydrogenation of propan-2-o1 also increased in proportion with the period of H2 reduction. In the case of MoO3 with noble metal, a higher dehydration activity was obtained by a longer period of H2 reduction, while the dehydrogenation activity was almost independent of the reduction period. Pt/MoO3 exhibited a high dehydration activity compared with the other catalysts, indicating the most acidic property of Pt/MoO3. We conclude from these results that the high isomerization activity of Pt/MoO3 can be attributed to its high acidity as well as to the hydrogenative and dehydrogenative properties of Pt metal.
View More



Contact:+86-717-6370352
Address:168 Chengdong Avenue, Yichang, Hubei 443003, P. R.China
Xinjiang Zhongtai Chemical Co., Ltd.
Contact:+86-991-8788172
Address:NO.78 XISHAN RD.URUMQI,CHINA
Contact:410-273-7300; 800-221-3953
Address:4609 Richlynn Dr., PO Box 369, Belcamp, MD, 21017-0369, USA
Contact:+86-513-80923002
Address:Suzhou Road 8,Beixin,Qidong,Jiangsu province,P.R.226221,China
Contact:18669908765
Address:Zibo City, Shandong Province, P.R.China
Doi:10.1016/S1872-2067(11)60521-4
(2013)Doi:10.1039/d0cc04935b
(2020)Doi:10.1002/cssc.201601595
(2017)Doi:10.3109/00498259409043216
(1994)Doi:10.1002/cbic.202000408
(2020)Doi:10.1016/j.tetlet.2009.11.074
(2010)