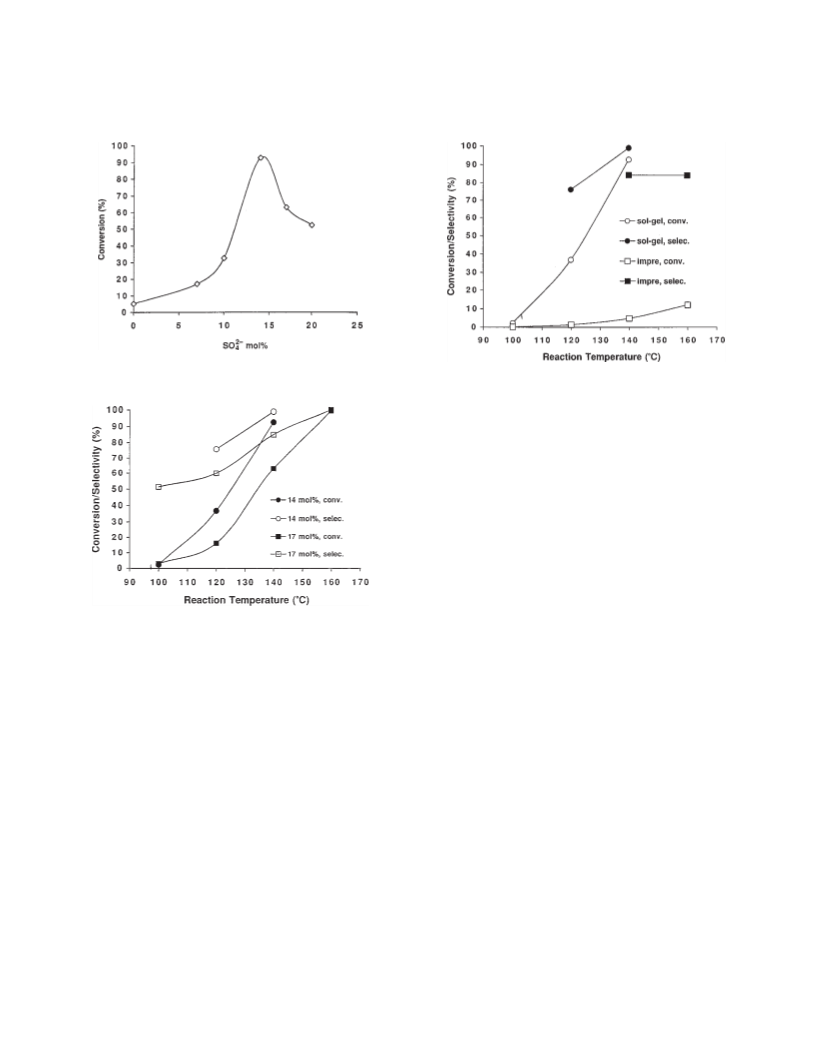
Zhuang and Miller
1221
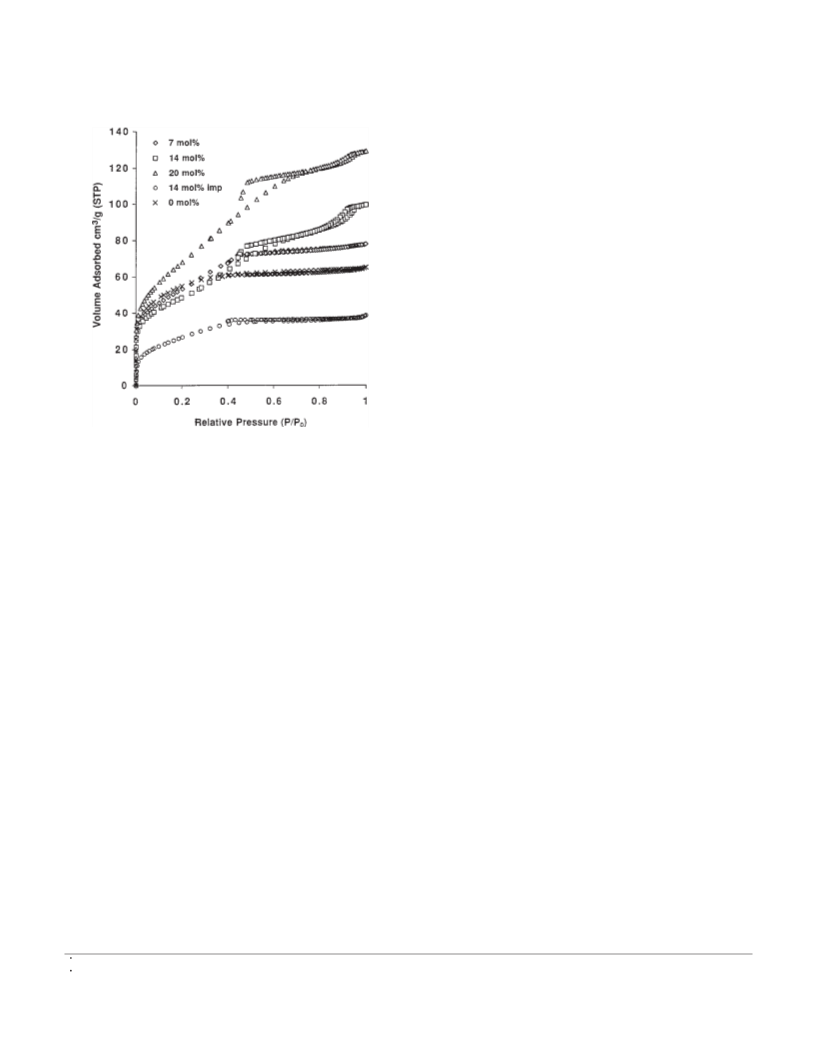
Fig. 1. N adsorption isotherms of the catalysts with different
SO4 content (based on synthetic concentrations).
Characterization of the catalysts
2
2
−
N adsorption analysis was measured by using a Coulter
2
SA 3100 instrument at –196°C. Samples were degassed un-
der vacuum at 200°C for 1 h immediately prior to N ad-
2
sorption analysis. From the isotherm, Brunauer–Emmett–
Tellar (BET) surface area, pore volume, and pore size distri-
bution were calculated by using the Barret, Joyner, and
Halenda (BJH) method (15).
DRIFTS spectra were acquired using a Spectra Tech
DRIFTS accessory (The Collector) in an ATI Mattson Re-
search Series FT-IR spectrometer equipped with a Michelson
interferometer, a helium–neon laser, a KBr beam splitter, a
DTGS (deuterated triglycerol sulfate) detector with a spectra
–
1
range of 4000–400 cm , and a standard high-intensity
source. The diffuse-reflectance FT-IR were recorded under
vacuum after evacuating the samples at 100°C for 30 min.
The sample chamber was designed to be used in situ on the
collector DRIFTS accessory. Five hundred scans were col-
lected.
Isopropanol dehydration evaluation
The catalytic activity evaluation of the catalysts for
isopropanol dehydration was performed using a continuous
3
flow fixed-bed micro reactor made of Pyrex glass. The in-
ner diameter of the reactor was 6 mm, its length was 25 cm.
With each reaction, 200 mg fresh catalyst was put into the
middle of the reactor. A thermocouple was placed just below
the catalyst bed to monitor the reaction temperature, which
was manipulated by a temperature controller. Before the re-
action, the catalyst was heated to the reaction temperature
mance was evaluated for isopropanol dehydration, which is
known as an acid-catalyzed reaction. N2 adsorption and
DRIFTS were employed to characterize the catalysts.
Experimental
under a flow of N . The reactions were performed at temper-
2
ature range of 100–160°C at 1 atm (1 atm = 103.325 kPa).
The reactant, isopropanol, was introduced into the reactor by
One-pot sol-gel synthesis of the catalysts
n-Butanol was obtained from Caledon Laboratories, all
other chemicals were obtained from Aldrich and used as re-
ceived.
a N bubbler, which was kept at a constant temperature
2
3
–1
(16). The flow rate of N was 100 mL min , giving a
2
–1
weight hour space velocity (WHSV) of the reactant as 1.8 h .
The products were analyzed by an on-line PerkinElmer
Sigma 3B gas chromatograph, employing a Porapaq R col-
umn, 800/100 mesh, 6 ft × 0.125 in, with a thermal conduc-
tivity detector (TCD). To analyze all of the products in the
exit stream from the reactor, the GC column was heated by
A series of sulfated ZrO –SiO (molar ratio, 75:25) with
2
2
sulfur contents up to 20 mol% were synthesized by the one-
pot sol-gel method. Briefly, ammonium sulfate, zirconium
propoxide, and tetraethyl orthosilicate were used as the pre-
cursors and 2,4-pentanedione (H-acac) as the complexing
3
3
agent (9). The appropriate amounts of zirconium propoxide
programming from 150 to 220°C for 15 min. The main
and tetraethyl orthosilicate were dissolved in the solvent,
n-butanol. The solution was heated to 60°C under stirring to
thoroughly mix the components, then it was cooled down to
room temperature, and H-acac was added. The alkoxides in
the solution were hydrolyzed over night by adding an aque-
ous solution of ammonium sulfate. A yellowish transparent
gel was obtained, which was dried at 100°C to remove the
solvent and then calcined at 500°C for 3 h in air to remove
the organics. The total concentration of the alkoxides in
n-butanol solution was 0.8 M. The molar ratio of the
complexing agent to the alkoxides was 0.5. The molar ratio
of hydrolyzing water to alkoxide was 11:1. A reference cata-
lyst was also prepared by impregnating a sol-gel synthesized
ZrO –SiO (75:25) with 1 M ammonium sulfate solution.
product was propylene, with minimal amounts of
diisopropylether as the byproduct. The activities were
stablized in ca. 20 min. After that time, two consecutive
analyses were carried out, which were within 5%. An aver-
age is reported throughout the Discussion section.
Results and discussion
Figure 1 shows the N adsorption isotherms of the cata-
2
lysts. The catalysts showed type IV isotherms, some with H2
hysteresis (15). By adding the (NH ) SO into the sol-gel
4
2
4
synthesis solutions, the N adsorption of the resultant cata-
2
lysts increased, especially at the intermediate and high rela-
tive pressure range. The pore size distributions of the
catalysts are displayed in Fig. 2. It can be seen that the tex-
tural structure of the catalysts were mesoporous. As the
2
2
The slurry was evaporated to dryness under stirring and then
4
calcined at 500°C for 1 h.
3
Q. Zhuang and J.M. Miller. Submitted.
The sulfate concentrations reported for the catalysts are based on synthetic concentrations in the uncalcined materials, not on analysis after
calcining.
4
©
2001 NRC Canada

 Zhuang
Zhuang
 Miller
Miller