S. Pyen et al.
Molecular Catalysis 448 (2018) 71–77
catalytic reactions. Finally, we discussed the variation in the catalyst
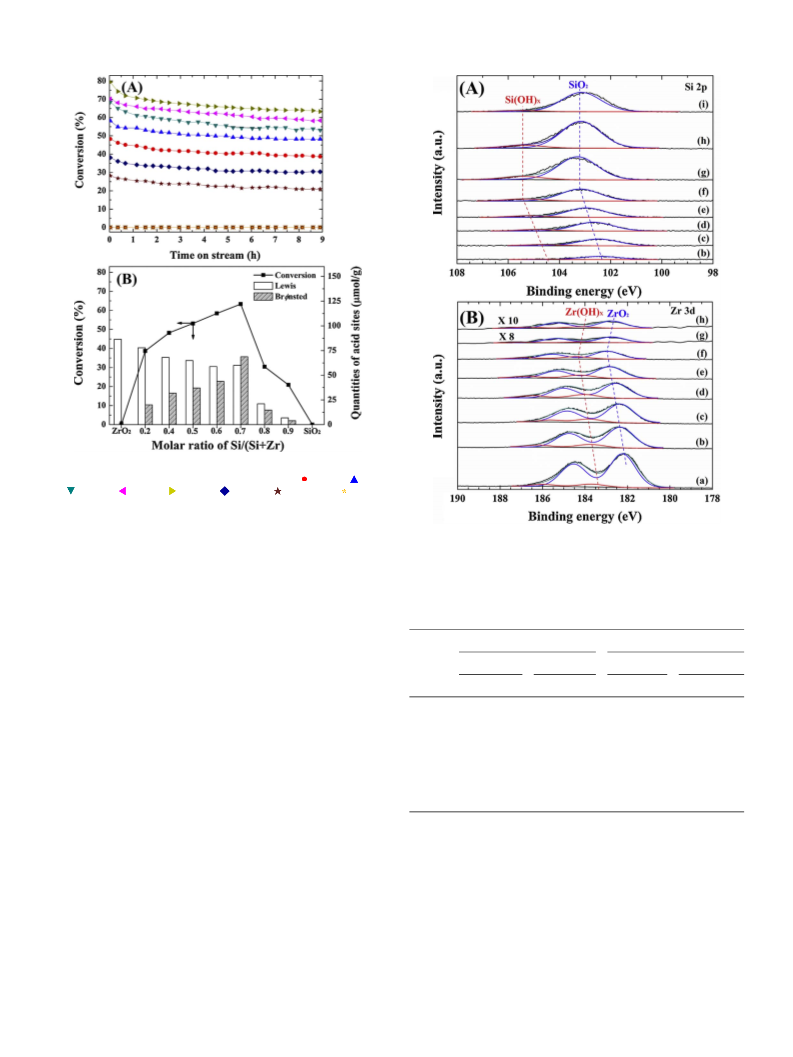
mixed oxide catalysts were identified by XPS using a PHI Quantera-II
acidic properties for the SiO
the atomic composition, and examined how such acidic functions op-
erated in the above catalytic reactions.
2
-ZrO
2
mixed oxide system in the context of
(ULVAC-PHI)
photoelectron
spectrometer
(Al Kα
radiation;
hν = 1486.6 eV), and XPS spectra were calibrated using the binding
energy of adventitious carbon (i.e., C 1s, 284.6 eV). To further interpret
the XPS results, peak-fitting and deconvolution of the O 1s, Zr 3d5/2
,
2. Experimental
and Si 2p spectra were carried out using the Gaussian–Lorentzian curve-
fitting method after background subtraction by the Shirley method.
2.1. Catalyst preparation
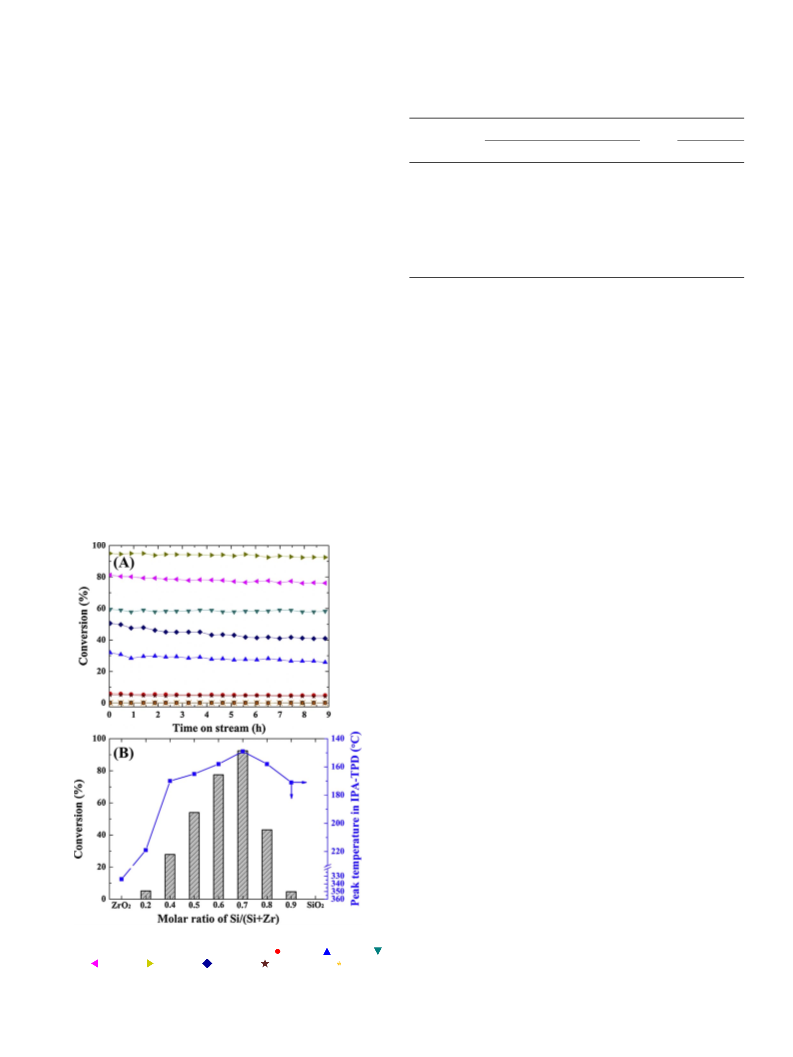
2.3. Catalytic activity tests
The Si-Zr mixed oxide catalysts were prepared via a co-precipitation
method with different molar ratios of Si/(Si + Zr) ranging from 0 to 1.
More specifically, the desired quantities of zirconia nitrate (ZrO
2.3.1. IPA decomposition
Prior to IPA decomposition experiments, all catalysts (0.1 g per re-
action) were pretreated under a flow of N
2
(50 cm3 min ) for 1 h at
−1
(
9
NO
8%, Acros Organics) were dissolved in mixture of 17 vol% deionized
water and 83 vol% ethanol at room temperature, and a 28 wt% solution
of NH OH (SK Chemicals) was added dropwise to give a pH of 9.5. The
3
)
2
∙2H
2
O, 99%, JUNSEI) and tetraethyl orthosilicate (C
8
H
20
O
4
Si,
400 °C in a fixed-bed quartz reactor. After pretreatment, the reactor was
cooled to 180 °C, and the isothermal reaction was allowed to proceed
2
for 9 h at a fixed partial pressure of 3 kPa IPA balanced with N (total
4
flow rate = 50 cm3 min ). The obtained dehydration products were
analyzed by an online gas chromatograph (Varian 3800) equipped with
a CP-Volamine capillary column (60 m × 0.32 mm) and an FID de-
tector. The conversion of IPA and the selectivities to propylene and di-
isopropyl ether (DIPE) were defined as follows:
−1
resulting suspension was aged at 75 °C for 48 h under reflux, after which
the obtained precipitate was washed with deionized water, and dried at
0 °C for 24 h. Finally, the dried samples were calcined at 500 °C for 6 h
under an air flow. The final products were labeled as xSi-Zr, where x
6
indicates the Si/(Si + Zr) molar ratio in the mixed oxide.
F
IPA reacted
Conversion(%) =
× 100% ;
2
.2. Characterization
FIPA fed
(3)
Fi
XRD measurements were carried out on a D8 Discover with GADDS
Selectivity(%) =
× 100% ;
(
Bruker AXS) diffractometer using Cu Kα radiation. The BET surface
areas, total pore volumes, and pore diameters of the calcined samples
were determined from the physical adsorption of N at −196 °C using
an ASAP 2020 (Micromeritics) after sample pretreatment at 250 °C for
h under vacuum. The BET surface areas were determined using the
BET method in the P/P range of 0.05–0.2. In addition, the pore size
∑ Fi (product)
(
4)
where FIPA and F
i
are the molar flow rates of IPA and the product i (i.e.,
2
propylene or DIPE).
5
2
.3.2. Formic acid decomposition
The formic acid (HCOOH) decomposition reaction was carried out
with 0.1 g of the catalyst in the identical reaction system as described
for the IPA decomposition reaction. After pretreatment at 400 °C for 1 h
under a flow of Ar (100 cm min ), the reactor was cooled to 230 °C,
and the isothermal reaction was performed under a 5 kPa HCOOH/Ar
balanced feed stream (total flow rate = 100 cm min ). The resulting
decomposition products were analyzed by an online gas chromato-
graphy (Varian 3800) equipped with
0
distribution of each catalyst was calculated using the Barrett-Joyner-
Halenda (BJH) method from a desorption branch of the isotherm, and
the total pore volumes were calculated from the quantity of N
2
ad-
3
−1
sorbed at P/P = 0.99. Scanning electron microscopy (SEM) was car-
0
ried out on an LEO-1530 (Carl Zeiss) instrument equipped with an EDS
to observe the morphology and atomic composition of the catalyst.
Py-IR analysis was performed on a Nicolet 6700 spectrometer
3
−1
a
HayeSep
Q
column
(
Thermo Scientific). Prior to analysis, a portion of the sample (30 mg)
(
80 m × 100 mm) and a TCD detector. The conversion of formic acid
was calculated using the following equation:
was pressed into a self-supported disk and pretreated at 300 °C for 1 h
under vacuum. Subsequently, pyridine (5 μL) was injected for 15 min at
1
recorded with 32 scans and a resolution of 4 cm . From Py-IR analysis,
the Lewis and Brønsted acid densities were quantified using the bands
at 1444 and 1540 cm , respectively, using previously reported em-
50 °C then evacuated for 30 min under vacuum. All Py-IR spectra were
FHCOOH reacted
−1
Conversion(%) =
× 100% ;
FHCOOH fed
(5)
−
1
where FHCOOH is the molar flow rate of formic acid.
pirical formulae [28] (integrated molar extinction coefficients = 2.22
−1
3. Results and discussion
and 1.67 cm μmol
tively).
for the Lewis and Brønsted acid sites, respec-
3.1. General properties of SiO
2 2
-ZrO mixed oxides
2
C (Pyridine on Lewis acid sites) = 1.42∙IA (L) R /W;
(1)
(2)
2
Following the successful preparation of the Si-Zr mixed oxide cat-
alysts composed of various Si/(Si + Zr) molar ratios, we initially ex-
amined their XRD patterns after calcination at 500 °C for 6 h. As shown
C (Pyridine on Brønsted acid sites) = 1.88·IA (B)·R /W;
where C −is the acid site number on
a per-gram-catalyst basis
μmol gcat ), IA (L or B) the integrated absorbance of the band (1444
1
(
2
in Fig. S1A, the characteristic peaks of pure ZrO were detected at
−1
or 1540 cm ) ascribed to Lewis or Brønsted acid site, R the radius of
the catalyst disk (cm), and W the weight of the disk (mg).
2θ = 30.1, 35.3, 50.4, and 60.2°, which corresponded to a tetragonal
crystalline structure (JCPDS No. 50-1089). However, the Si-Zr mixed
oxides expressed a low degree of crystallinity, namely X-ray amorphous
phase. This is due to introduction of Si species into the Zr matrix sta-
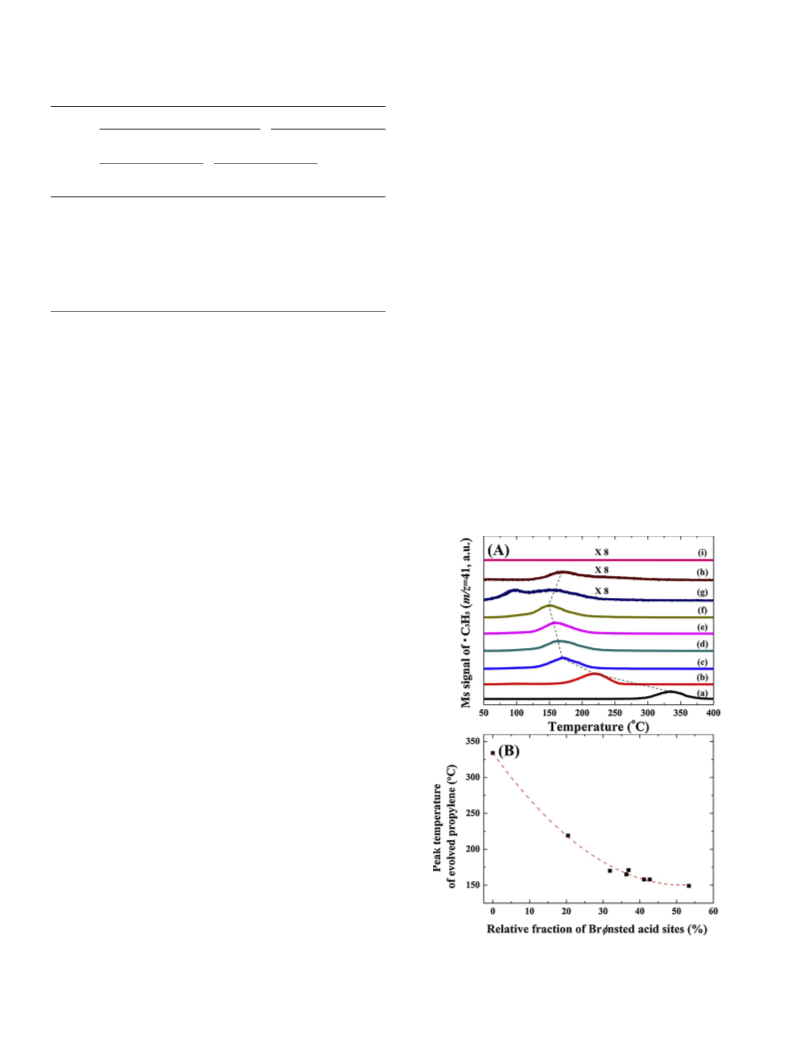
IPA-TPD measurement was performed using quadrupole mass
spectrometry (GSD301, Pfeiffer vacuum) according to the following
procedure. The sample (0.05 g) was loaded into a U-shaped quartz re-
bilized the tetragonal phase of ZrO
were calcined at a much higher temperature of 900 °C for 6 h, and all
SiO -ZrO mixed oxides exhibited the tetragonal ZrO crystallites
compared to pure ZrO showing mixed monoclinic and tetragonal
phases. In addition, the tetragonal characteristic peaks of ZrO became
less intense as the content of SiO increased (see Fig. S1B). It is known
that the ZrO can exist in monoclinic, tetragonal, and cubic crystal
structures, and the development of the crystalline structure is largely
2
. For confirmation, the samples
actor and pretreated at 300 °C for 1 h under
a
flow of Ar
3
−1
(
30 cm min ). After cooling to room temperature, IPA was adsorbed
2
2
2
3
−1
on the catalyst surface under a flow of 3% IPA/Ar (30 cm min ) for
2
30 min, followed by removal of the physisorbed IPA in an Ar flow for
2
3
0 min. Subsequently, the sample was heated to 400 °C with a heating
2
−1
3
−1
rate of 10 °C min
under a flow of Ar (30 cm min ), and evolved
2
mass fragments were recorded. The surface electronic states of the Si-Zr
72

 Pyen, Seunghee
Pyen, Seunghee
 Hong, Eunpyo
Hong, Eunpyo
 Shin, Mi
Shin, Mi
 Suh, Young-Woong
Suh, Young-Woong
 Shin, Chae-Ho
Shin, Chae-Ho