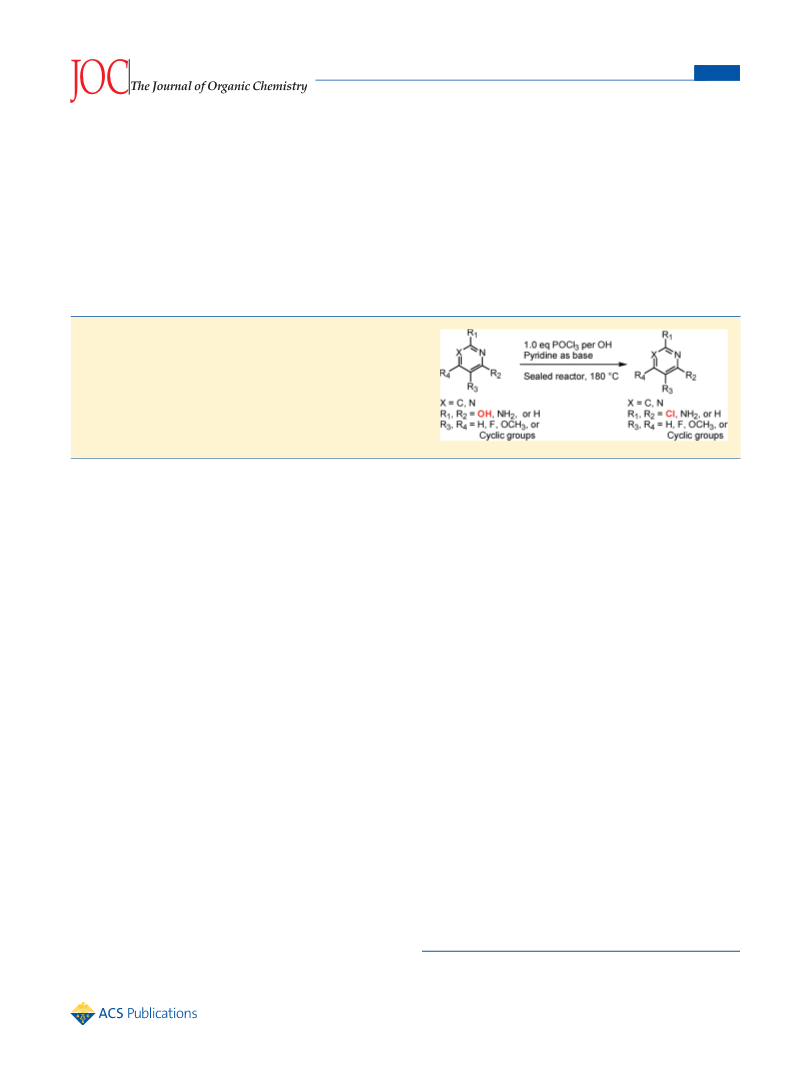
The Journal of Organic Chemistry
NOTE
literature reports. New compounds were fully characterized with 1H and
13C NMR and MS.
purity 95% (by HPLC, wavelength 230 nm); mp 118ꢀ120 °C (lit.20
118ꢀ120 °C); MS (EI, m/e) 197.9 (100), 199.9 (64). Spectroscopic data
matched literature reported data: 1H NMR.33
2,4-Dichloropyrido[3,2-d]pyrimidine (8): reaction scale 0.3
mol; yield 56.7 g (94.5%); purity 95% (HPLC 230 nm); mp
165ꢀ167 °C (lit.22 168ꢀ169 °C); MS (EI, m/e) 198.6 (100), 200.5
(64). Spectroscopic data (1H and 13C NMR in DMSO-d6) matched
literature reported data in DMSO-d634 and CDCl3.22
Caution: The safety parameters of the reaction protocols were not
systematically evaluated. Please exercise extreme caution and use proper
protective gear when operating with sealed reactors at high temperature.
2,4-Dichloropyrimidine (1). To a 1 L Teflon-lined stainless steel
reactor was added 161 mL of pyridine (2.0 mol) and 224 g of 2,4-
dihydroxypyrimidine (2.0 mol). With stirring, 360 mL of POCl3 (4.0
mol) was added. The reactor was sealed and heated to maintain at
180 °C for 2 h during which time the pressure inside rose to a maximum
of 0.12 MPa and then dropped to 0.02 MPa. After being cooled to rt, the
reaction mixture was poured into 600 mL of cold water (0ꢀ5 °C) with
stirring. The resulting oil was extracted into 1 L of ethyl acetate, which
was washed with saturated aqueous NaCl solution and dried over
Na2SO4. The organic phase was concentrated under reduced pressure,
and the resulting oil was distilled under vacuum (85ꢀ88 °C/10 mmHg)
to give 272 g of desired product (91%): purity 98.5% (HPLC, 230 nm);
MS (EI, m/e) 147.9 (100), 149.9 (64), 151.9 (10). Spectroscopic data
matched literature reported data: 1H NMR.25
2, 4-Dichloro-5-fluoropyrimidine (4): reaction scale 1.5 mol,
product was obtained as oil after removal of extraction solvent; yield 233
g (93%); purity 99% (by HPLC, wavelength 230 nm); MS (EI, m/e)
165.7 (100), 167.8 (64). Spectroscopic data matched literature reported
data: 1H and 19F NMR.26
Note: product extraction with organic solvent is not always required.
After quenching, one could directly separate the oily organic phase from
the aqueous phase and proceed to distillation after washing and drying.
This no-extraction protocol was used in our preparation of 2-chloropyr-
idine with good results (Table 2). 2-Chloropyridine (5): reaction scale
1.3 mol; yield 127.5 g (90%, distillation 165 ꢀ 173 °C); purity 96.8% (by
HPLC, wavelength 230 nm). Spectroscopic data matched literature
reported data: 1H NMR and MS.27,28
6-Bromo-4-chloropyrido[2,3-d]pyrimidine (11). To a 500 mL
Teflon-lined stainless steel reactor was added 80 mL of toluene, followed
by 60 mL of pyridine (0.75 mol) and 170 g of 6-bromo-pyrido[2,3-
d]pyrimidin-4-ol (0.75 mol). Then 73 mL of POCl3 (0.75 mol) was added
with stirring. The reactor was sealed and heated to 180 °C, and the pressure
reached 0.14 MPa. After 1 h, the pressure dropped to 0.09 MPa. Heating
was stopped after 2 h while the pressure was 0.03 MPa. After cooling, the
reaction mixture was poured into 500 mL of cold water and gave a light
yellow crystalline solid. The product was collected by filtration, washed
with 20 mL of toluene, and dried: yield 132 g (72%); purity 96.5% (HPLC,
1
230 nm); mp 152ꢀ154 °C; H NMR (CDCl3, δ, ppm) 9.33 (overlap,
2H), 8.80 (d, 1H); 13C NMR (CDCl3, δ, ppm) 162.2, 160.2, 157.10,
136.4, 120.6, 120.2; HRMS m/e 242.8932 [M]þ (calcd for C7H3BrClN3,
242.9919); MS (EI, m/e) 244.9 (100), 242.9 (84), 246.9 (25).
6-Bromo-2,4-dichloropyrido[2,3-d]pyrimidine (12): reaction
scale 0.75 mol with 9 h reaction time; yield 159 g (76%); purity 98%
(HPLC, 230 nm); mp 176ꢀ177 °C; 1H NMR (CDCl3, δ, ppm) 9.32 (d,
1H), 8.77 (d, 1H); 13C NMR (CDCl3, δ, ppm) 163.5, 161.1, 159.1, 157.9,
136.6, 120.6, 118.6; HRMS m/e 276.8783 [M]þ (calcd for C7H2BrCl2N3,
276.8809); MS (EI, m/e) 278.9 (100), 276.9 (67), 280.9 (46).
4-Chloropyrido[2,3-d]pyrimidine (9): reaction scale 1.3 mol
with 5 h reaction time; yield 153 g (73%); purity 97.5% (by HPLC,
wavelength 230 nm); mp 145ꢀ147.5 °C (lit.23 mp 240 °C, lit.9 mp
137 °C dec); MS (EI, m/e) 165.0 (100), 167.0 (32). Spectroscopic data
matched literature reported data: 1H NMR.13,23
2,4-Dichloro-5-methoxypyrimidine (2). To a 50 L ceramic
reactor was first added 6.8 L of pyridine (84.5 mol), followed by 12 kg
(84.5 mol) of 2,4-dihydroxy-5-methoxypyrimidine. Then 16.5 L of
POCl3 (169 mol) was slowly added over 15ꢀ20 min with stirring,
during which time the temperature of the mixture rose slightly from rt
(from 25ꢀ30 °C to ∼35 °C). This small temperature rise may be partly
due to water content of the 2,4-dihydroxy-5-methoxypyrimidine sample.
At this stage, the reaction components were still a mixture of solid and
liquid. The reactor was sealed and heated to reach an inside temperature
of 180 °C in 30ꢀ40 min; at this time, the pressure inside the reactor
reached its highest point of 0.12 MPa. After 30 min, the pressure inside
dropped to 0.09 MPa. The reaction was maintained at 180 °C for 2 h,
and the pressure dropped to 0.02 MPa. After cooling to rt, there was no
excess pressure inside and the reactor was opened. The resulting
reaction mixture existed as a slightly yellow liquid. It was poured into
20 L of cold water (0ꢀ5 °C) within 10 min with stirring. No significant
heat release was observed during quenching. The resulting white
crystalline solid was collected by filtration and washed with small
amount (∼500 mL) of 1:1 petroleum ether:ethyl acetate. This afforded
13.9 kg (92%) of product after drying: purity 97.5% (by HPLC,
wavelength 230 nm); mp 68ꢀ70 °C (lit.15 mp 67ꢀ68 °C). Spectro-
scopic data matched literature reported data: 1H NMR29 and MS.30
2-Amino-4-chloropyrimidine (3): reaction scale 0.3 mol; yield
37 g (95%); purity 96.5% (by HPLC, wavelength 230 nm); mp
156ꢀ158 °C (lit.31 mp 155ꢀ156 °C). Spectroscopic data matched
literature reported data: 1H NMR31 and MS.31
2,4-Dichloropyrido[2,3-d]pyrimidine (10): reaction scale 1.3
mol with 9 h reaction time; yield 190 g (75%); purity 98% (by HPLC,
1
wavelength 230 nm); mp 154ꢀ156 °C (lit.9 mp 157ꢀ158 °C); H
NMR (CDCl3, δ, ppm) 9.3 (br, s, 1H), 8.66 (dd, 1H), 7.75 (dd, 2H);
13C NMR (CDCl3, δ, ppm) 164.7, 159.8, 159.6, 159.0, 135.5, 124.7,
117.9; HRMS m/e 198.8998 [M]þ (calcd for C7H3Cl2N3, 198.9704);
MS (EI, m/e) 198.87 (100), 200.87 (64).
’ ASSOCIATED CONTENT
S
Supporting Information. NMR spectra of new com-
b
pounds or known compounds with no previous report of spectra.
This material is available free of charge via the Internet at http://
pubs.acs.org.
’ AUTHOR INFORMATION
Corresponding Author
*E-mail: zhihuasun@sues.edu.cn.
’ ACKNOWLEDGMENT
Support of this work is provided in part by the State Key
Laboratory of Drug Research, Chinese Academy of Sciences.
2,6-Dichloro-9H-purine (6): reaction scale 1.3 mol, 3 h reaction;
yield 206 g (88%); purity 94.5% (HPLC 230 nm); MS (EI, m/e) 187.9
(100), 189.9 (64), 191.9 (10); mp 176ꢀ178 °C (lit.7 mp 181 °C dec).
Spectroscopic data matched literature reported data: 1H NMR.32
2,4-Dichloroquinazoline (7). The reaction was carried out at
150 °C for 2 h under nitrogen: reaction scale 1.3 mol; yield 232 g (94%);
’ REFERENCES
(1) Gabriel, S. Chem. Ber. 1905, 38, 1690.
(2) Whittaker, N.; Jones, T. S. G. J. Chem. Soc. 1951, 1565.
(3) Hunds, A. Chlorination process for the preparation of chloro-
pyrimidines from hydroxypyrimidines or their tautomeric keto forms.
GB 2287466, 1995, CAN 124:176135.
4152
dx.doi.org/10.1021/jo2003715 |J. Org. Chem. 2011, 76, 4149–4153

 Sun, Zhihua
Sun, Zhihua
 Wang, Han
Wang, Han
 Wen, Kun
Wen, Kun
 Li, Ya
Li, Ya
 Fan, Erkang
Fan, Erkang