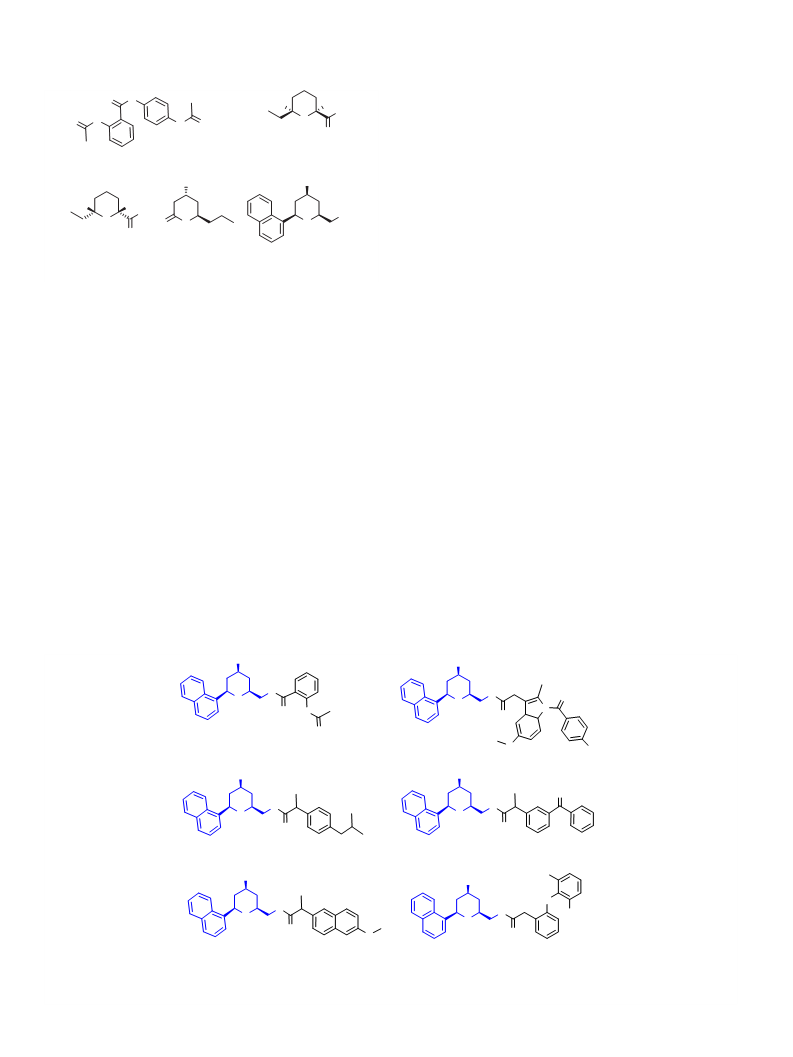
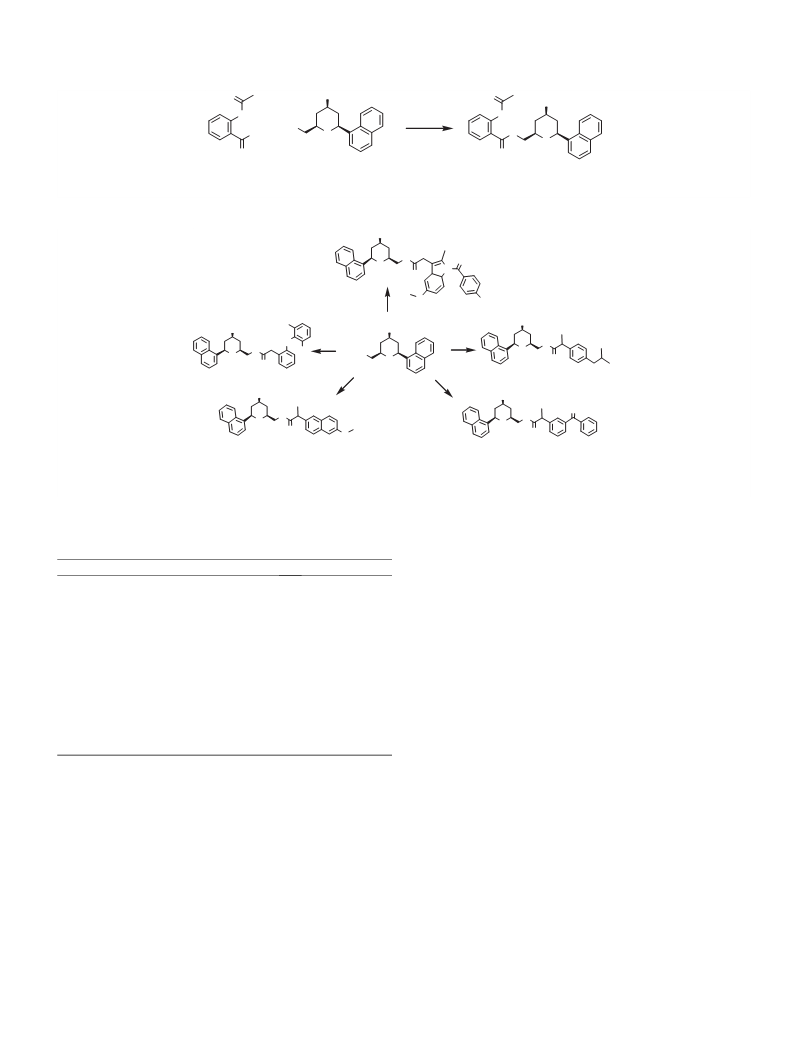
6
008
S. L. Capim et al. / Bioorg. Med. Chem. 21 (2013) 6003–6010
aromatic); 1735 (C@O of ester); 1681 (C@O of amide); 2924 (C–H
4.1.6. Synthesis of cis-(± ±-4-chloro-6-(naphthalen-1-yl±-
tetrahydro-2H-pyran-2-yl±methyl 2-(6-methoxynaphthalen-2-
yl±propanoate (8±
3
1
sp ); 3429 (N–H). H NMR (CDCl
sxt, 2H, J = 12 Hz); 2.12 (m, 2H); 2.36 (s, 3H); 2.55 (dd, 1H,
J = 4 Hz); 3.68 (t, 2H, J = 4 Hz); 3.72 (m, 3H); 3.86 (m, 1H); 4.06
3
, 400 MHz) d: 1.25 (t, 1H); 1.81
(
The reaction was performed by stirring a solution of naproxen
(
(
4
1
1
m, 1H); 4.24 (m, 2H, J = 4 Hz); 5.09 (m, 1H); 6.79 (m, 2H); 7.66
2
(2.0 g) in SOCl (5.0 mL) and the mixture was stirred at room temꢀ
1
3
m, 12H). C NMR (101 MHz; CDCl
3
) d: 13.37; 30.18; 38.24;
perature at 80 °C for 3 h. After the end of reaction (verified by chroꢀ
matography using hexane:ethyl acetate (7:3) as solvent), the
resulting mixture on evaporation afforded naproxen chloride in
95% yield. 1 mmol of naproxen chloride and 1 mmol of alcohol
(±)ꢀ3 were stirred in 10 mL of THF and 0.5 mL of TEA at 80 °C for
7 days. After the end of reaction (verified by chromatography),
2.77; 54.97; 55.52; 66.59; 74.92; 75.64; 101.23; 111.53;
12.31; 114.92; 122.74; 125.50; 128.94; 128.99; 130.49;
31.06; 133.64; 133.73; 136.01; 155.94; 168.17; 170.59. Anal.
Calcd for C35
6
H33Cl
2 5
NO , C, 68.18; H, 5.07; N, 2.27. Found: C,
8.19; H, 5.08; N, 2.22.
the resulting mixture was extracted with CH
2
Cl
2
(3 Â 20 mL). Pure
4
.1.4. Synthesis of cis-(± ±-4-chloro-6-(naphthalen-1-yl±-
product was obtained from the crude reaction by column chromaꢀ
tography through silica gel, using AcOEt:hexane as solvent in a raꢀ
tio of 3:7. The solvent mixture was concentrated under reduced
tetrahydro-2H-pyran-2-yl±methyl 2-(4-isobutylphenyl±
propanoate (6±
À1
The reaction was performed by stirring a solution of ibuprofen
pressure to afford 8 in 74% yield; IR (KBr, cm ): 825 and 800
(
9.7 mmol) in SOCl
2
(5.0 mL) and the mixture was stirred at room
(C–H aromatics); 1327–1178 (C–O); 1600 and 1500 (C@C aromatꢀ
ics); 1730 (C@O ester); 2970 (C–H sp ). H NMR (CDCl , 400 MHz)
3
d: 0.84 (t, 2H); 1.24 (m, 1H); 1.59 (m, 5H); 1.71 (m, 2H); 3.44 (m,
1H); 3.87 (m, 1H); 4.01 (s, 3H); 4.04 (m, 1H); 4.10 (m, 2H); 7.28
(m, 2H); 7.52 (m, 4H); 7.70 (m, 5H); 8.17 (d, 2H). 13C NMR
3
1
temperature at 80 °C for 3 h. After the end of reaction (verified by
chromatography), the resulting mixture evaporated obtained 95%
of ibuprofen chloride. 1 mmol of ibuprofen chloride and 1 mmol
of alcohol (±)ꢀ3 were stirred in 10 mL of THF and 0.5 mL of TEA
at 80 °C for 7 days. After the end of reaction (verified by chromaꢀ
tography using hexane:ethyl acetate (7:3) as solvent), the resultꢀ
3
(101 MHz; CDCl ) d: 10.29; 18.32; 18.43; 21.91; 25.93; 28.97;
44.41; 45.30; 56.97; 63.98; 66.44; 113.88; 113.94; 116.73;
123.88; 123.97; 126.12; 126.13; 127.39; 127.49; 127.81; 129.45;
129.50; 130.99; 131.03; 136.41; 136.63; 152.75; 174.50. Anal.
ing mixture was extracted with CH
2
Cl
2
(3 Â 20 mL). Pure product
was obtained from the crude reaction by column chromatography
through silica gel, using AcOEt:hexane as solvent at a ratio of 3:7.
The solvent mixture was concentrated under reduced pressure to
afford 6 in 70% yield; IR (KBr, cm ): 779 and 736 (C–H aroꢀ
matic); 1377–1330 (C–O); 1512 and 1454 (C@C aromatic); 1732
4
Calcd for C32H29ClO : C, 74.92; H, 5.70. Found: C, 74.85; H, 5.62.
À1
4.1.7. 4.1.7.Synthesis of cis-(± ±-4-chloro-6-(naphthalen-1-yl±-
tetrahydro-2H-pyran-2-yl±methyl 2-(2-(2,6-dichlorophenylamino±
phenyl±acetate (9±
3
1
(
C@O of ester); 2954 (C–H sp ). H NMR (CDCl
3
, 400 MHz) d:
0
2
1
3
3
1
1
.86 (dd, 6H, J = 4 Hz); 1.51 (dd, 3H); 1.92 (m, 5H); 2.38 (dd,
H, J = 4 Hz); 2.52 (m, 1H); 3.81 (m, 2H); 4.26 (m, 3H); 5.05 (d,
The reaction was performed by stirring a solution of alcohol 3
(0.53 mmol) in CH Cl (2.0 mL), triethylamine (0.2 mL), 4ꢀtolueneꢀ
2 2
sulfonyl chloride (1.3 mmol) and the mixture was stirred at 0 °C for
30 min. and 12 h at room temperature. After the end of reaction
(verified by chromatography using hexane:ethyl acetate (7:3) as
H, J = 12 Hz); 7.01 (m, 2H); 7.22 (dd, 2H, J = 4 Hz); 7.53 (m,
H); 7.89 (m, 3H). 1 C NMR (101 MHz; CDCl
3
3
) d: 18.31; 22.40;
0.11; 38.32; 42.87; 55.11; 66.18; 66.33; 75.04; 75.70; 122.88;
23.33; 125.54; 126.14; 127.18; 128.40; 128.96; 129.29;
30.13; 133.72; 136.17; 136.24; 137.42; 140.60, 174.51. Anal.
2 2
solvent), the resulting mixture was extracted with CH Cl
(3 Â 20 mL). The organic phase was dried with anhydrous sodium
sulfate and concentrated under reduced pressure where tosylate
derivative of alcohol 3 was obtained a 95% yield. After, 0.1 mmol
of tosyl derivative of alcohol 3 was stirred with 0.15 mmol of dicꢀ
Calcd for C29
3
H33ClO , C, 74.90; H, 7.62. Found: C, 74.86; H, 7.57.
4
.1.5. Synthesis of cis-(± ±-4-chloro-6-(naphthalen-1-yl±-
tetrahydro-2H-pyran-2-yl±methyl 2-(3-benzoylphenyl±
propanoate (7±
The reaction was performed by stirring a solution of ketoproꢀ
2
fen (2.0 g) in SOCl (5.0 mL) and the mixture was stirred at room
temperature at 80 °C for 3 h. After the end of reaction (verified by
chromatography), the resulting mixture on evaporation afforded
ketoprofen chloride in 95% yield. 1 mmol of ketoprofen chloride
and 1 mmol of alcohol (±)ꢀ3 were stirred in 10 mL of THF and
2 3
lofenac Potassium, 22 mg of K CO in 1 mL of DMF under microꢀ
wave irradiation (100 W) at 100 °C for 90 min. Pure product was
obtained from the crude reaction by column chromatography
through silica gel, using AcOEt:hexane as solvent in a ratio of
3:7. The solvent mixture was concentrated under reduced pressure
À1
to afford 9 in 90%; IR (KBr, cm ): 777 and 742 (C–H aromatics);
1303 (C–O); 1577 and 1502 (C@C aromatics); 1691 (C@O ester);
3
1
3
2918 (C–H sp ); 3348 (N–H). H NMR (CDCl , 400 MHz) d: 1.26
0
.5 mL of TEA at 80 °C for 7 days. After the end of reaction (veriꢀ
fied by chromatography using hexane:ethyl acetate (7:3) as solꢀ
vent), the resulting mixture was extracted with CH Cl
(t, 1H, J = 8 Hz); 1.81 (dd, 1H, J = 12 Hz); 2.09 (q, 2H, J = 12 Hz);
2.23 (m, 1H); 2.58 (t, 1H, J = 4 Hz); 2.61 (t, 4H, J = 4 Hz); 3.76 (m,
4H); 4.33 (m, 1H); 5.14 (d, 1H); 7.23 (m, 2H); 7.51 (m, 3H); 7.62
(d, 1H, J = 8 Hz); 7.85 (m, 2H); 8.00 (d, 1H, J = 8 Hz). 13C NMR
(101 MHz; CDCl ) d: 38.02; 38.15; 42.76; 55.39; 65.66; 75.70;
3
77.91; 118.24; 122.04; 122.89; 123.26; 123.90; 123.98; 125.37;
125.64; 126.29; 128.05; 128.60; 128.82; 128.96; 129.38; 130.32;
2
2
(
3 Â 20 mL). Pure product was obtained from the crude reaction
by column chromatography through silica gel, using AcOEt:hexꢀ
ane as solvent at a ratio of 3:7. The solvent mixture was concenꢀ
trated under reduced pressure to afford 7 in 72% yield; IR (KBr,
À1
cm ): 786 and 721 (C–H aromatics); 1315–1172 (C–O); 1593
130.90; 133.73; 136.09; 142.60; 175.93. Anal. Calcd for C30
H26Cl3ꢀ
and 1450 (C@C aromatics); 1658 (C@O) 1735 (C@O ester); 2958
NO : C, 64.94; H, 4.72; N, 2.52. Found: C, 65.07; H, 4.71; N, 2.51.
3
3
1
(
2
(
1
1
7
1
1
C–H sp ). H NMR (CDCl
.48 (m, 1H); 2.73 (m, 3H); 2.91 (m, 2H); 3.11 (m, 1H); 3.39
m, 1H); 3.74 (m, 1H); 4.66 (m, 1H); 5.01 (m, 2H); 5.31 (m,
3
, 400 MHz) d: 2.10 (d, 1H, J = 6 Hz);
4.2. Pharmacology
4.2.1. Animals
The experiments were carried out on male Albino–Swiss mice
(body weight 20–24 g). The animals were housed in wire mesh
cages in a room temperature and exposed to a 12 h light:12 h dark
cycle. The animals had free access to standard pellet diet, tap water
was given ad libitum. The protocol for this study was approved by
H); 5.47 (m, 2H); 8.73 (m, 16H). 13C NMR (101 MHz; CDCl
3
) d:
9.74; 27.37; 30.37; 39.75; 44.24; 46.67; 56.41; 65.48; 67.88;
6.32; 124.23; 124.60; 126.93; 127.52; 129.66; 129.81; 130.33;
30.52; 131.43; 132.93; 133.86; 135.08; 137.49; 138.81;
39.19; 142.03; 142.20; 175.27; 197.82. Anal. Calcd for C32
: C, 74.92; H, 5.70. Found: C, 74.85; H, 5.62.
H
29ꢀ
ClO
4

 Capim, Saulo L.
Capim, Saulo L.
 Goncalves, Gabriela M.
Goncalves, Gabriela M.
 Dos Santos, Gabriela C.M.
Dos Santos, Gabriela C.M.
 Marinho, Bruno G.
Marinho, Bruno G.
 Vasconcellos, Mario L.A.A.
Vasconcellos, Mario L.A.A.