6
0
M. Sebah et al. / Journal of Molecular Catalysis A: Chemical 374–375 (2013) 59–65
2
.1.1. PAPSQa
stirred under reflux for 12 h. On cooling to room temperature the
solid was filtered and then washed extensively with distilled water,
methanol and diethyl ether. The solid was then dried at 120 C for
HCl (160 mL of 5 M) was added to 20 g of the colourless liq-
◦
uid (from Section 2.1) and then stirred under reflux for 5 h. On
cooling to room temperature the white solid was filtered and
washed extensively with distilled water and methanol. The solid
was dried under reduced pressure at 120 C for 4 h to afford a white
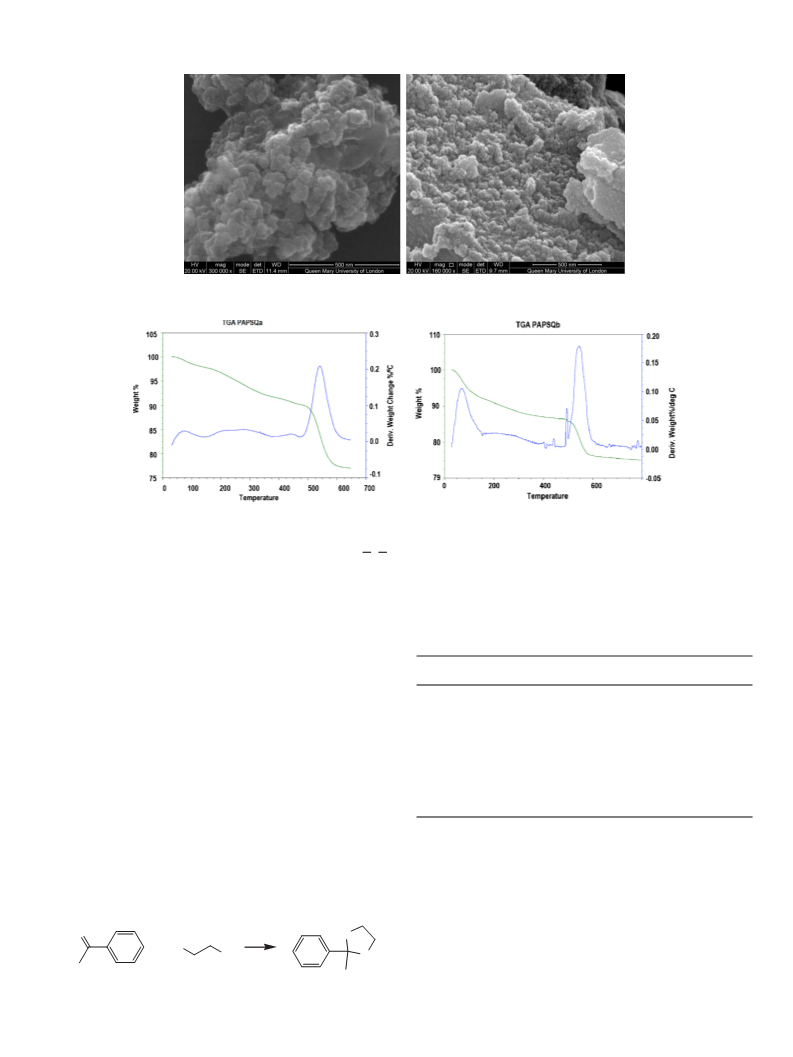
powder PAPSQa (10.9 g, 89.7%). Surface area: 65 m g
4 h to yield a white powder PAPSQb (2.9 g, 79%).
2
−1 31
Surface area: 80 m g
,
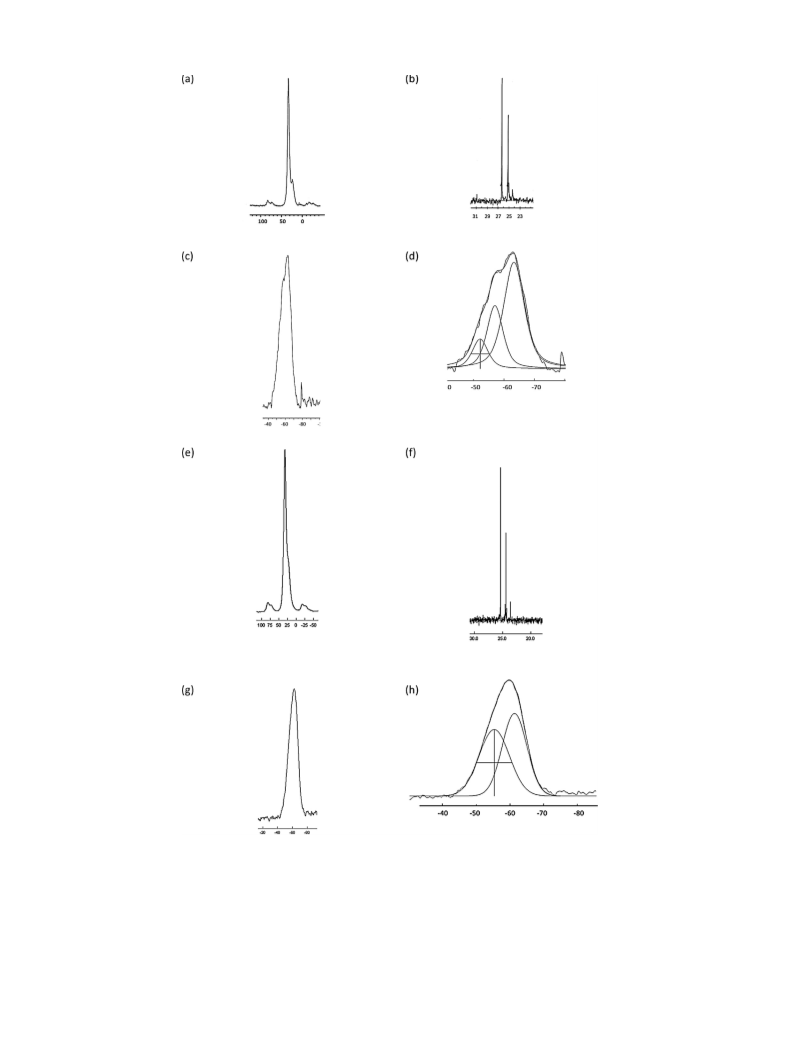
P NMR (NaOD/D O) ıP: 25.2, 26.3 ratio
2
◦
31
29
1:1.5; P CPMAS NMR ıP 32.2 (br s), Si MAS ı : −63 (br s) decon-
Si
2
−1 29
−1
,
Si MAS
voluted peaks at −58, −64, found P 5.1 mmol g Accessible acid by
31
−1
ı : −63 (br s) deconvoluted peaks at −53, −58, −63. P CPMAS
titration 3.3 mmol g
.
Si
NMR ıP 33 (PO(OH) , 90%) 24 (Si OH–O P(OH) , 10%). Found P
2
2
−
1
4
.96 mmol g
.
2.2. Catalysis
2
.1.1.1. Accessible acid measurement. In a typical measurement to
Quantity of catalyst used (mol%) was based on the available acid
determined by titration. Percentage conversions were determined
by H NMR spectroscopy or isolated products. Turnover numbers
determine the phosphonic acid first proton exchange capacity, a
sample of PAPSQa 0.2040 g was shaken in 50 mL of 0.5 M sodium
formate solution for 60 h. The formic acid produced was titrated
1
are based on three cycles in each case. Blank reactions were per-
formed in the absence of catalyst. The catalyst was recycled either
after filtration and washing or after removal of the product solution
and addition of fresh reagent and solvent. Leaching was assessed by
interrupting the reaction (ketalisation, esterification and transes-
terification) at partial conversion (1 h), separating the liquid phase
from the catalyst using a 0.2 m filter and measuring conver-
sion again at the specified reaction time. No evidence of leaching
was detected. The ketalisation, esterification and transesterifica-
tion experiments were also run using the equivalent quantity of
phosphoric acid as catalyst.
−
1
with 20 mM NaOH. Accessible acid in PAPSQa: 3.3 mmol g
.
2
.1.1.2. NMR PAPSQa dispersed in (NaOD/D O). PAPSQa was dis-
2
persed in NaOD/D O wherein the phosphonate is fully ionised
while Si-C bonds remain intact. This is a convenient way to confirm
presence of components (1) and (2) (see below)
2
Ha
C1
Ha
O
P
O
Hb
C2
Hb
O
Si
O
O
O
2.2.1. Protection of ketones (ketalisation)
(
1)
A mixture of acetophenone (4.8 g, 40.0 mmol, 4.7 mL), ethane-
,2-diol (6.7 g, 107.6 mmol, 6.0 mL) catalyst (30 mg, 0.25 mol%) in
1
O
Hf
O
P
toluene (30 mL) was refluxed 10 h using a Dean-Stark apparatus.
The reaction was followed by TLC. The reaction mixture was cooled
to room temperature, the catalyst filtered off and washed with
ether (20 mL). The ketal product 2-methyl-2-phenyl-1,3-dioxolane
was obtained as a white solid from the filtrate after washing with
water (3 × 20 mL) and drying over magnesium sulfate.
Hc
Si
C4
Hd
O
O-
C2
O
O-
C3
C1
He
Ha
Hg
Hb
1
H NMR (CDCl3): ıH: 1.67 (s, 3H), 3.78 (t, 2H), 4.02 (t, 2H),
7
.25–7.56 (m, 5Harom).
Si
O
O
O
2.2.2. Esterification of oleic acid
Oleic acid (1 g, 3.54 mmol), catalyst (20 mg, 1.9 mol%) and
(
2)
ethanol (6 mL) were combined and the reaction mixture was
◦
ıP (PAPSQa) 26.8 (s, P (1)), 25.6 (s, P (2)) intensity ratio 1.5:1.
refluxed for 8 h then heated to 90 C for 2 h using an air condenser.
ıH (1) 0.65 (Ha, m, 2H), 1.36 (Hb, m, 2H); ıC (1) 7.6 (C1), 22.6 (d,
C2) (Assignments confirmed by HSQC [14]), ıH (2) 0.53 (m, Hb,
J(b,a),15.27 Hz, J(b,e), 3.43 Hz, J(b,g), 8.73 Hz), 0.73 (m, Ha, J(a,b),
The evaporated solvent was replaced with dry ethanol and the
mixture heated a further 2 h using an air condenser. Ether (30 mL)
was added to the cooled mixture and the catalyst was filtered off,
washed with diethyl ether and retained for recycling. The organic
solvents were removed from the filtrate. Conversion to the ethyl
oleate was determined by integration of the total alkene proton
signal at ıH 5.33 (m, 2H) against the ester methylene signal at ıH
4.11 (q, 2H, OCH CH ).
1
5.27 Hz, J(a,e), 10.45 Hz, J(a,g), 4.2 Hz), 0.99 (m, Hc, J(c,d), 5.22 Hz,
J(c,e), 9.76 Hz, J(c,f), 9.32 Hz, J(c,g), 4.27 Hz, J(c,P), 14.10 Hz), 1.27 (m,
Hd, J(d,f), 14.90 Hz, J(d,P), 17.62 Hz), 1.55 (m, He, J(e,g), 13.48 Hz),
1
.63(m, Hf, J(f,P), 16.2 Hz), 1.89 (m, Hg, J(e,g), 13.48 Hz), ıC (2) 11.6
C1), 21.1 (C2), 26.6 (C3), 31.0 (d, C4) (Assignments confirmed by
HSQC [14]).
(
2
3
ton spin system in component (2) are shown in Supplementary
Information Fig. A.
Supplementary data associated with this article can be
found, in the online version, at http://dx.doi.org/10.1016/j.molcata.
2.2.3. Transesterification of ethyl oleate
1
Ethyl oleate (1.55 g, 5 mmol), catalyst (50 mg, 3.3 mol%) and pen-
tanol (10 mL) were refluxed for 12 h. The reaction was followed
by TLC. On cooling, ether (30 mL) was added and the catalyst was
filtered off. The organic solvents were removed. Conversion was
determined by integration of the alkene proton signals at ıH 5.35
and the ester methylene signal at ıH 4.13 (t, 2H, OCH (CH ) CH ).
2
013.03.021.
2
2
3
3
2.1.2. PAPSQb
2.2.4. HMF synthesis
To 6.0 g of the mixture of 1 and 2 mole ratio 1.5:1 (from
Fructose (206 mg, 1.14 mmol) was dissolved in deionized water
(1.8 g) and PAPSQa (209 mg) was added to this solution. An organic
phase (2 g) containing MIBK and 2-butanol in 7:3 ratio was added to
the reaction vessel to obtain a biphasic mixture. The reaction mix-
Section 2.1) was added aqueous hydrochloric acid (1 M, 15 mL)
and methanol (180 mL). The resultant solution was placed in a
◦
polypropylene plastic bottle and warmed to 90 C for 16 h. The
◦
formed glass was ground to a powder. To this powder, hydrochloric
acid (5 M, 50 mL) was added slowly and the resultant mixture was
ture was heated to 130 C, while stirring at 600–700 rpm for 12 h.
◦
The reaction mixture was then cooled to 90 C without stirring,

 Sebah, Majda
Sebah, Majda
 Maddala, Sai P.
Maddala, Sai P.
 Haycock, Peter
Haycock, Peter
 Sullivan, Alice
Sullivan, Alice
 Toms, Harold
Toms, Harold
 Wilson, John
Wilson, John