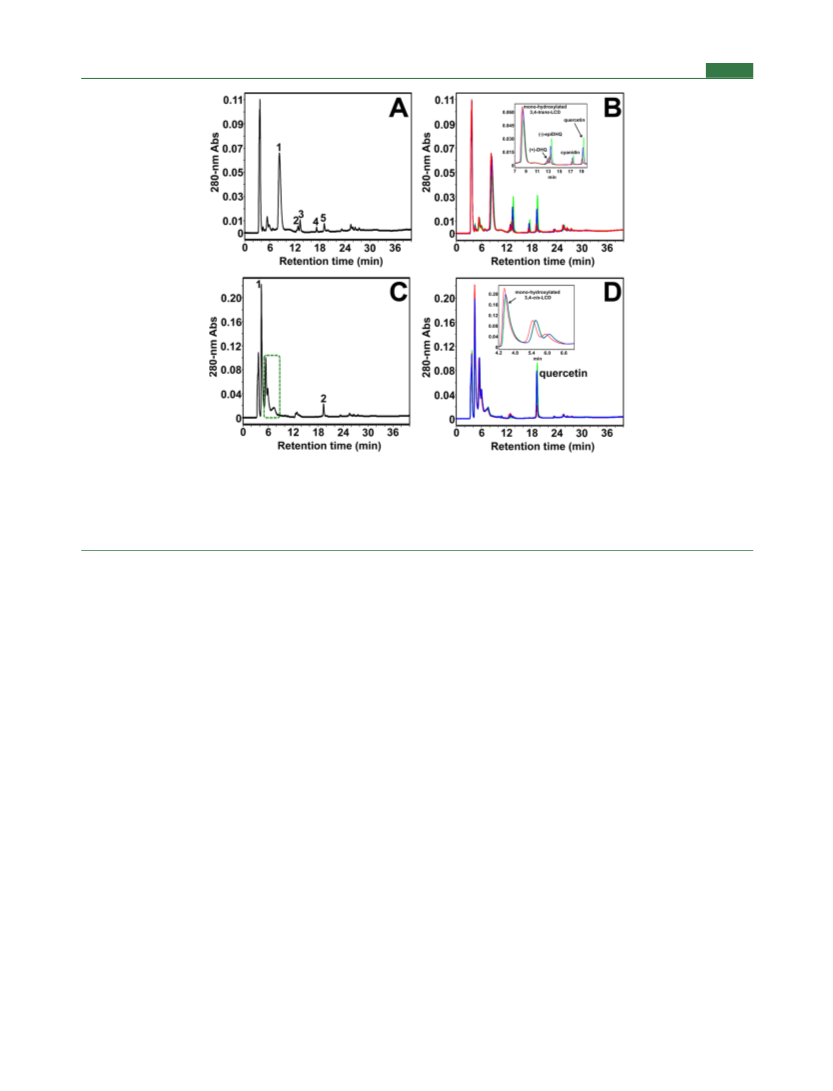
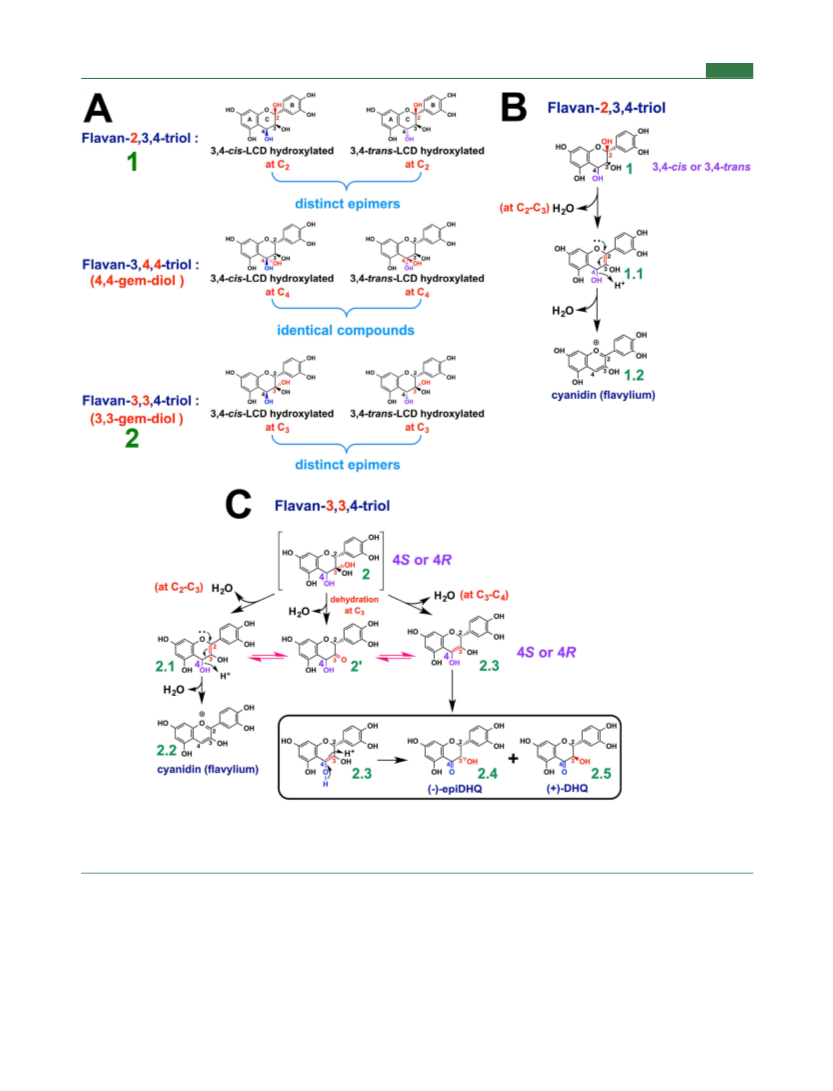
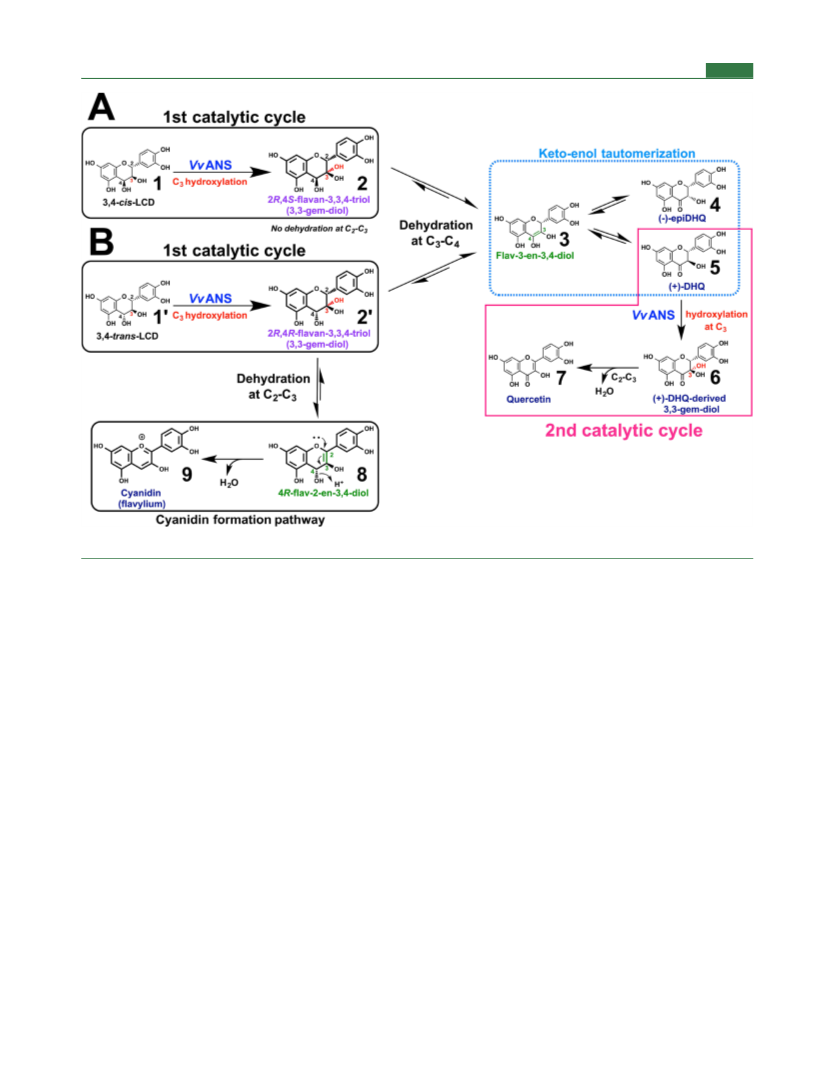
Journal of Agricultural and Food Chemistry
Article
(15) Fimognari, C.; Lenzi, M.; Hrelia, P. Chemoprevention of
cancer by isothiocyanates and anthocyanins: mechanisms of action
and structure-activity relationship. Curr. Med. Chem. 2008, 15, 440−
447.
(16) Lin, B. W.; Gong, C. C.; Song, H. F.; Cui, Y. Y. Effects of
anthocyanins on the prevention and treatment of cancer. Br. J.
Pharmacol. 2017, 174, 1226−1243.
(17) Fernandes, I.; Azevedo, J.; Faria, A.; Calhau, C.; de Freitas, V.;
Mateus, N. Enzymatic hemisynthesis of metabolites and conjugates of
anthocyanins. J. Agric. Food Chem. 2009, 57, 735−745.
(18) Zha, J.; Koffas, M. A. G. Production of anthocyanins in
metabolically engineered microorganism: Current status and
perspectives. Synth. Syst. Biotechnol. 2017, 2, 259−266.
(19) Appelhagen, I.; Wulff-Vester, A. K.; Wendell, M.; Hvoslef-Eide,
A. K.; Russell, J.; Oertel, A.; Martens, S.; Mock, H. P.; Martin, C.;
Matros, A. Colour bio-factories: Towards scale-up production of
anthocyanins in plant cell cultures. Metab. Eng. 2018, 48, 218−232.
(20) Tohge, T.; Perez de Souza, L.; Fernie, A. R. Current
understanding of the pathways of flavonoid biosynthesis in model
and crop plants. J. Exp. Bot. 2017, 68, 4013−4028.
AUTHOR INFORMATION
■
Corresponding Author
*Tel.: +33 (0)6 63 07 53 48. Fax: +33 (0)5 40 00 24 38. E-
ORCID
̀
Notes
The authors declare no competing financial interest.
ACKNOWLEDGMENTS
■
We would like to thank our colleague Claude Manigand for his
technical advice with the chiral HPLC analysis. J.-r.Z. gratefully
acknowledges a three-year funding PhD scholarship from the
department of Bioengineering (Qilu University of Technology,
China).
(21) Saito, K.; Kobayashi, M.; Gong, Z. Z.; Yamazaki, M.; Tanaka, Y.
Direct evidence for anthocyanidin synthase as a 2-oxoglutarate-
dependent oxygenase: molecular cloning and functional expression of
cDNA from a red forma of Perilla frutescens. Plant J. 1999, 17, 181−
189.
REFERENCES
■
(1) Kong, J. M.; Chia, L. S.; Goh, N. K.; Chia, T. F.; Brouillard, R.
Analysis and biological activities of anthocyanins. Phytochemistry
2003, 64, 923−933.
(2) Steyn, W. J.; Wand, S. J. E.; Holcroft, D. M.; Jacobs, G.
Anthocyanins in vegetative tissues: a proposed unified function in
photoprotection. New Phytol. 2002, 155, 349−361.
̈
(22) Menssen, A.; Hohmann, S.; Martin, W.; Schnable, P. S.;
Peterson, P. A.; Saedler, H.; Gierl, A. The En/Spm transposable
element of Zea mays contains splice sites at the termini generating a
novel intron from a dSpm element in the A2 gene. EMBO J. 1990, 9,
3051−3057.
(3) Ferreira da Silva, P. F.; Paulo, L.; Barbafina, A.; Elisei, F.; Quina,
F. H.; Maca̧ nita, A. L. Photoprotection and the photophysics of
acylated anthocyanins. Chem. - Eur. J. 2012, 18, 3736−3744.
(4) Petrussa, E.; Braidot, E.; Zancani, M.; Peresson, C.; Bertolini, A.;
Patui, S.; Vianello, A. Plant flavonoids − Biosynthesis, transport and
involvement in stress responses. Int. J. Mol. Sci. 2013, 14, 14950−
14973.
(23) Martin, C.; Prescott, A.; Mackay, S.; Bartlett, J.; Vrijlandt, E.
Control of anthocyanidin biosynthesis in flowers of Antirrhinum
majus. Plant J. 1991, 1, 37−49.
(24) Weiss, D.; van der Luit, A. H.; Kroon, J. T. M.; Mol, J. N. M.;
Kooter, J. M. The petunia homologue of the Antirrhinum majus candi
and Zea mays A2 flavonoid genes; homology to flavanone 3-
hydroxylase and ethylene-forming enzyme. Plant Mol. Biol. 1993,
22, 893−897.
(25) Turnbull, J. J.; Sobey, W. J.; Aplin, R. T.; Hassan, A.; Firmin, J.
L.; Schofield, C. J.; Prescott, A. G. Are anthocyanidins the immediate
products of anthocyanidin synthase? Chem. Commun. 2000, 2473−
2474.
(26) Turnbull, J. J.; Nagle, M. J.; Seibel, J. F.; Welford, R. W. D.;
Grant, G. H.; Schofield, C. J. The C-4 stereochemistry of
leucocyanidin substrates for anthocyanidin synthase affects product
selectivity. Bioorg. Med. Chem. Lett. 2003, 13, 3853−3857.
(27) Nakajima, J. I.; Tanaka, Y.; Yamazaki, M.; Saito, K. Reaction
mechanism from leucoanthocyanidin to anthocyanidin 3-glucoside, a
key reaction for coloring in anthocyanin biosynthesis. J. Biol. Chem.
2001, 276, 25797−25803.
(5) Goupy, P.; Bautista-Ortin, A. B.; Fulcrand, H.; Dangles, O.
Antioxidant activity of wine pigments derived from anthocyanins:
hydrogen transfer reactions to the DPPH radical and inhibition of the
heme-induced peroxidation of linoleic acid. J. Agric. Food Chem. 2009,
57, 5762−5770.
(6) Khoo, H. E.; Azlan, A.; Tang, S. T.; Lim, S. M. Anthocyanidins
and anthocyanins: colored pigments as food, pharmaceutical
ingredients, and the potential health benefits. Food Nutr. Res. 2017,
61, 1361779.
(7) Giusti, M. M.; Wrolstad, R. E. Acylated anthocyanins from edible
sources and their applications in food systems. Biochem. Eng. J. 2003,
14, 217−225.
(8) Zhao, C. L.; Yu, Y. Q.; Chen, Z. J.; Wen, G. S.; Wei, F. G.;
Zheng, Q.; Wang, C. D.; Xiao, X. L. Stability-increasing effects of
anthocyanin glycosyl acylation. Food Chem. 2017, 214, 119−128.
(9) Li, D.; Wang, P.; Luo, Y.; Zhao, M.; Chen, F. Health benefits of
anthocyanins and molecular mechanisms: update from recent decade.
Crit. Rev. Food Sci. Nutr. 2017, 57, 1729−1741.
(28) Welford, R. W. D.; Clifton, I. J.; Turnbull, J. J.; Wilson, S. C.;
Schofield, C. J. Structural and mechanistic studies on anthocyanidin
synthase catalysed oxidation of flavanone substrates: the effect of C-2
stereochemistry on product selectivity and mechanism. Org. Biomol.
Chem. 2005, 3, 3117−3126.
(10) Liobikas, J.; Skemiene, K.; Trumbeckaite, S.; Borutaite, V.
Anthocyanins in cardioprotection: A path through mitochondria.
Pharmacol. Res. 2016, 113, 808−815.
(29) Wellmann, F.; Griesser, M.; Schwab, W.; Martens, S.;
(11) Aiyer, H. S.; Warri, A. M.; Woode, D. R.; Hilakivi-Clarke, L.;
Clarke, R. Anthocyanins in obesity-associated thrombogenesis.
Influence of berry polyphenols on receptor signaling and cell-death
pathways: implications for breast cancer prevention. J. Agric. Food
Chem. 2012, 60, 5693−5708.
̌
Eisenreich, W.; Matern, U.; Lukacin, R. Anthocyanidin synthase
from Gerbera Hybrida catalyzes the conversion of (+)-catechin to
cyanidin and a novel procyanidin. FEBS Lett. 2006, 580, 1642−1648.
(30) Welford, R. W. D.; Turnbull, J. J.; Claridge, T. D. W.; Prescott,
A. G.; Schofield, C. J. Evidence for oxidation at C-3 of the flavonoid
C-ring during anthocyanidin biosynthesis. Chem. Commun. 2001,
1828−1829.
(12) Belwal, T.; Nabavi, S. F.; Nabavi, S. M.; Habtemariam, S.
Dietary anthocyanins and insulin resistance: when food becomes a
medicine. Nutrients 2017, 9, 1111.
(31) Turnbull, J. J.; Nakajima, J. I.; Welford, R. W. D.; Yamazaki, M.;
Saito, K.; Schofield, C. J. Mechanistic studies on three 2-oxoglutarate-
dependent oxygenases of flavonoid biosynthesis. J. Biol. Chem. 2004,
279, 1206−1216.
(32) Busso, D.; Delagoutte-Busso, B.; Moras, D. Construction of a
set Gateway-based destination vectors for high-throughput cloning
(13) He, Y.; Hu, Y.; Jiang, X.; Chen, T.; Ma, Y.; Wu, S.; Sun, J.; Jiao,
R.; Li, X.; Deng, L.; Bai, W. Cyanidin-3-O-glucoside inhibits the UVB-
induced ROS/COX-2 pathway in HaCaT cells. J. Photochem.
Photobiol., B 2017, 177, 24−31.
(14) Wang, L. S.; Stoner, G. D. Anthocyanins and their role in
cancer prevention. Cancer Lett. 2008, 269, 281−290.
I
J. Agric. Food Chem. XXXX, XXX, XXX−XXX

 Zhang, Jia-Rong
Zhang, Jia-Rong
 Trossat-Magnin, Claudine
Trossat-Magnin, Claudine
 Bathany, Katell
Bathany, Katell
 Delrot, Serge
Delrot, Serge
 Chaudière, Jean
Chaudière, Jean