Please do not adjust margins
Green Chemistry
Page 4 of 5
DOI: 10.1039/C6GC01880G
COMMUNICATION
Journal Name
11
, 616; (d) H. Liu, G. Chen, H. Jiang, Y. Li and R. Luque,
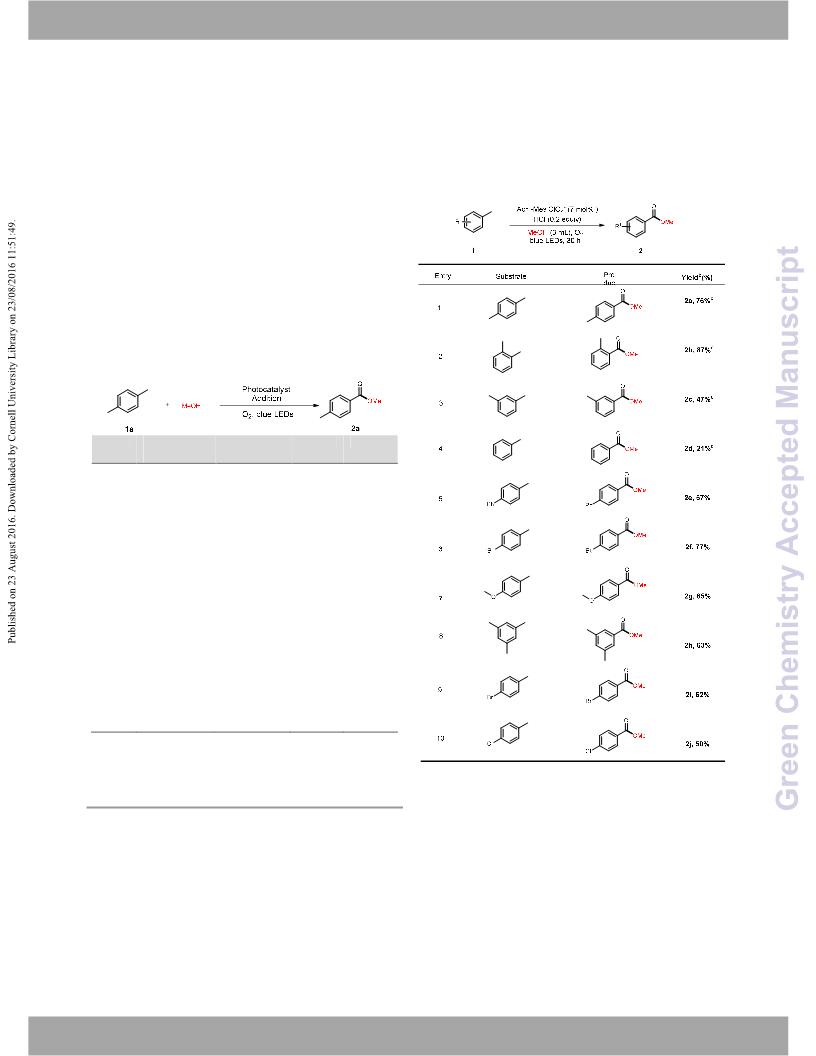
methyl aromatics, providing a green route for aromatic methyl
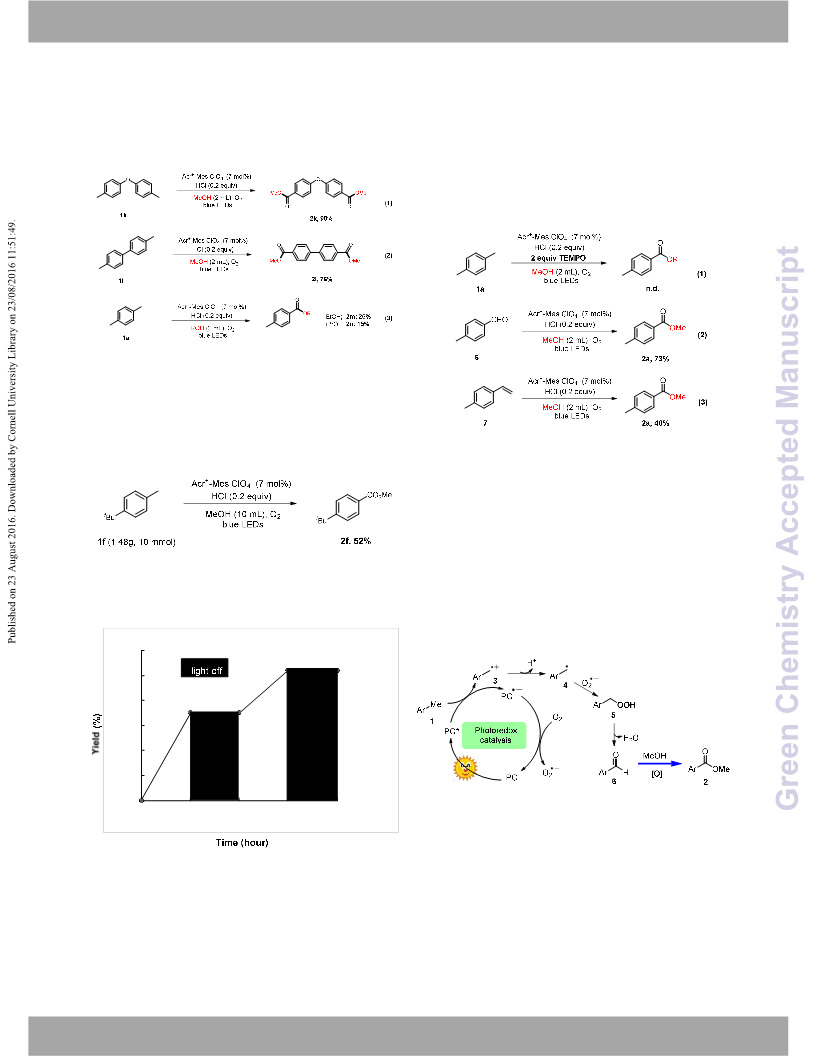
carboxylates. In addition, this protocol can be scalable to gram scale.
We believe photo-mediated this oxidative reaction will help
chemists to design more and more interesting, useful, and
sustainable reactions in the near future.
ChemSusChem, 2012, 5, 1892
.
11. Selected reviews on photocatalysis (a) D. P. Hari and B. Konig,
Angew. Chem. Int. Ed., 2013, 52, 4734; (b) M. N. Hopkinson, B. Sahoo,
J. L. Li and F. Glorius, Chemistry, 2014, 20, 3874; (c) T. Koike and M.
Akita, Inorg. Chem. Front., 2014,
R. Stephenson, Chem. Soc. Rev., 2011, 40, 102; (e) D. A. Nicewicz and
T. M. Nguyen, ACS Catalysis, 2014, , 355; (f) C. K. Prier, D. A.
Rankic and D. W. MacMillan, Chem. Rev., 2013, 113, 5322; (g) D.
Ravelli, S. Protti and M. Fagnoni, Chem. Rev. 2016, DOI:
1, 562; (d) J. M. Narayanam and C.
This work was supported by the 973 Program
(2012CB725302), the National Natural Science Foundation of
China (21390400, 21025206, 21272180, and 21302148), and
the Research Fund for the Doctoral Program of Higher
Education of China (20120141130002) and the Ministry of
Science and Technology of China (2012YQ120060). The
Program of Introducing Talents of Discipline to Universities of
China (111 Program) is also appreciated.
4
,
10.1021/acs.chemrev.5b00662; (h) Y. Xi, H. Yi and A. Lei, Org.
Biomol. Chem., 2013, 11, 2387; (i) J. Xuan and W. J. Xiao, Angew.
Chem. Int. Ed., 2012, 51, 6828; (j) T. P. Yoon, M. A. Ischay and J. Du,
Nat Chem, 2010, 2, 527.
12. (a) K. Ohkubo, K. Mizushima, R. Iwata, K. Souma, N. Suzuki and S.
Fukuzumi, Chem. Commun., 2010, 46, 601; (b) G. Pandey and R. Laha,
Angew. Chem. Int. Ed., 2015, 54, 14875; (c) G. Pandey, R. Laha and D.
Singh, J. Org. Chem., 2016, DOI: 10.1021/acs.joc.6b00970; (d) G.
Pandey, S. Pal and R. Laha, Angew. Chem. Int. Ed., 2013, 52, 5146; (e)
K. Qvortrup, D. A. Rankic and D. W. MacMillan, J. Am. Chem. Soc.
2014, 136, 626; (f) J. B. Xia, C. Zhu and C. Chen, J. Am. Chem. Soc.
,
,
Notes and references
2013, 135, 17494; (g) Y. Yamada, K. Maeda, K. Ohkubo, K. D. Karlin
and S. Fukuzumi, Phys. Chem. Chem. Phys., 2012, 14, 9654; (h) H. Yi,
C. Bian, X. Hu, L. Niu and A. Lei, Chem. Commun., 2015, 51, 14046.
§ Lingling Zhang and Hong Yi contributed equally.
1. (a) R. L. Brutchey, I. J. Drake, A. T. Bell and T. D. Tilley, Chem.
Commun., 2005, 29, 3736; (b) R. Vanjari and K. N. Singh, Chem. Soc.
Rev., 2015, 44, 8062.
2.(a) J.ꢀB. Feng, D. Wei, J.ꢀL. Gong, X. Qi and X.ꢀF. Wu, Tetrahedron
Lett., 2014, 55, 5082; (b) S. Guin, S. K. Rout, A. Banerjee, S. Nandi
and B. K. Patel, Org. Lett., 2012, 14, 5294; (c) R. Vanjari, T.
Guntreddi and K. N. Singh, Org. Lett., 2013, 15, 4908; (d) Y. Wang, K.
Yamaguchi and N. Mizuno, Angew. Chem. Int. Ed., 2012, 51, 7250; (e)
Y. Wu, P. Y. Choy, F. Mao and F. Y. Kwong, Chem. Commun., 2013,
13. K. Ohkubo, K. Mizushima, R. Iwata and S. Fukuzumi, Chem. Sci.
2011, , 715.
,
2
49, 689; (f) F. Xiong, C. Qian, D. Lin, W. Zeng and X. Lu, Org. Lett.
,
2013, 15, 5444; (g) Z. Yin and P. Sun, J. Org. Chem., 2012, 77, 11339.
3. (a) H. Liu, G. Shi, S. Pan, Y. Jiang and Y. Zhang, Org. Lett., 2013, 15
4098; (b) Z. Ni, Q. Zhang, T. Xiong, Y. Zheng, Y. Li, H. Zhang, J.
Zhang and Q. Liu, Angew. Chem. Int. Ed., 2012, 51, 1244; (c) D. A.
Powell and H. Fan, J. Org. Chem., 2010, 75, 2726; (d) S. K. Rout, S.
,
Guin, K. K. Ghara, A. Banerjee and B. K. Patel, Org. Lett., 2012, 14
,
3982; (e) P. Xie, C. Xia and H. Huang, Org. Lett., 2013, 15, 3370; (f) P.
Xie, Y. Xie, B. Qian, H. Zhou, C. Xia and H. Huang, J. Am. Chem.
Soc., 2012, 134, 9902; (g) J. Yuan, X. Ma, H. Yi, C. Liu and A. Lei,
Chem. Commun., 2014, 50, 14386.
4. S. K. Rout, S. Guin, W. Ali, A. Gogoi and B. K. Patel, Org. Lett., 2014,
16, 3086.
5. J. Otera, Esterification: Methods, Reactions, and Applications, Wileyꢀ
VCH, Weinheim, 2003.
6. (a) P. J. Garegg, L. Olsson and S. Oscarson, J. Org. Chem., 1995, 60
,
2200; (b) R. Gopinath, B. Barkakaty, B. Talukdar and B. K. Patel, J.
Org. Chem., 2003, 68, 2944; (c) R. Gopinath and B. K. Patel, Org.
Lett., 2000,
Catalysis A: Chemical
Chakraborty, Tetrahedron Lett., 2010, 51, 3521; (f) B. R. Travis, M.
Sivakumar, G. O. Hollist and B. Borhan, Org. Lett., 2003, , 1031; (g)
2, 577; (d) B. Karimi and J. Rajabi, Journal of Molecular
,
2005, 226 165; (e) P. Malik and D.
,
5
X.ꢀF. Wu and C. Darcel, Eur. J. Org. Chem., 2009, 2009, 1144.
7. (a) S. Arita, T. Koike, Y. Kayaki and T. Ikariya, Angew. Chem. Int. Ed.
2008, 47, 2447; (b) S. Gowrisankar, H. Neumann and M. Beller,
Angew. Chem. Int. Ed., 2011, 123, 5245; (c) C. Liu, J. Wang, L. Meng,
Y. Deng, Y. Li and A. Lei, Angew. Chem. Int. Ed., 2011, 50, 5144; (d)
,
R. L. Oliveira, P. K. Kiyohara and L. M. Rossi, Green Chem., 2009, 11
1366; (e) J. Williams, N. Owston, T. Nixon, A. Parker and M.
Whittlesey, Synthesis, 2009, 2009, 1578; (f) N. Yamamoto, Y. Obora
and Y. Ishii, J. Org. Chem., 2011, 76, 2937; (g) J. Zhang, G. Leitus, Y.
BenꢀDavid and D. Milstein, J. Am. Chem. Soc., 2005, 127, 10840; (h)
,
T. Zweifel, J.ꢀV. Naubron and H. Grützmacher, Angew. Chem. Int. Ed.
,
2009, 121, 567.
8. (a S. Tang, J, Yuan, C, Liu and A, Lei. Dalton Trans., 2014, 43
,
13460.
9. (a) J. Otera, Chem. Rev., 1993, 93, 1449; (b) E. Taarning, I. S. Nielsen,
K. Egeblad, R. Madsen and C. H. Christensen, ChemSusChem, 2008, 1,
75;
10. (a) S. Hirashima, T. Nobuta, N. Tada, T. Miura and A. Itoh, Org. Lett.
2010, 12, 3645; (b) L. Liu, L. Yun, Z. Wang, X. Fu and C.ꢀh. Yan,
Tetrahedron Lett., 2013, 54, 5383; (c) N. Tada, Y. Ikebata, T. Nobuta,
S. Hirashima, T. Miura and A. Itoh, Photochem Photobiol Sci, 2012,
,
4 | J. Name., 2012, 00, 1-3
This journal is © The Royal Society of Chemistry 20xx
Please do not adjust margins

 Zhang, Lingling
Zhang, Lingling
 Yi, Hong
Yi, Hong
 Wang, Jue
Wang, Jue
 Lei, Aiwen
Lei, Aiwen