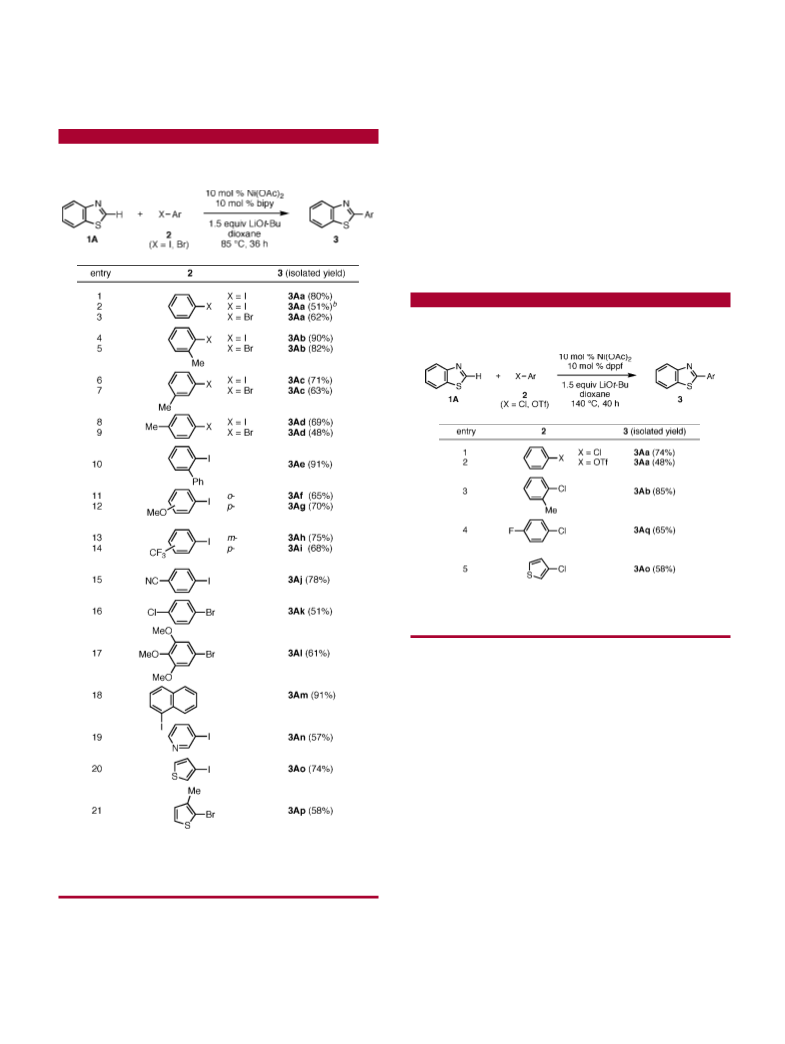1
3
Nakamura and Yu. We report herein our finding that a
nickel complex can catalyze the arylation of heteroarenes
(
efficient catalyst in the presence of LiOt-Bu. For example,
the cross-coupling product 2-phenylbenzothiazole (3Aa) was
obtained in 64% yield when the reaction was carried out in
THF at 85 °C for 18 h, representing our “early” standard
1
4,15
azoles) with aryl halides and triflates.
We began our study by examining various nickel salts,
ligands, and basic additives in the reaction of benzothiazole
1A) and iodobenzene (2a). After extensive screening, we
determined that Ni(OAc) /2,2′-bipyridyl (bipy) serves as an
1
6
conditions (Scheme 1).
(
2
(
7) Selected examples of Pd catalysis: (a) Pivsa-Art, S.; Satoh, T.;
Kawamura, Y.; Miura, M.; Nomura, M. Bull. Chem. Soc. Jpn. 1998, 71,
67. (b) Okazawa, T.; Satoh, T.; Miura, M.; Nomura, M. J. Am. Chem.
Scheme 1
.
Discovery of Ni Catalysis and Influence of
Parameters
4
Soc. 2002, 124, 5286. (c) Mori, A.; Sekiguchi, A.; Masui, K.; Shimada, T.;
Horie, M.; Osakada, K.; Kawamoto, M.; Ikeda, T. J. Am. Chem. Soc. 2003,
1
25, 1700. (d) Park, C.-H.; Ryabova, V.; Seregin, I. V.; Sromek, A. W.;
Gevorgyan, V. Org. Lett. 2004, 6, 1159. (e) Chabert, J. F. D.; Joucla, L.;
David, E.; Lemaire, M. Tetrahedron 2004, 60, 3221. (f) Kalyani, D.; Deprez,
N. R.; Desai, L. V.; Sanford, M. S. J. Am. Chem. Soc. 2005, 127, 7330. (g)
Lane, B. S.; Brown, M. A.; Sames, D. J. Am. Chem. Soc. 2005, 127, 8050.
(
h) Campeau, L.-C.; Rousseaux, S.; Fagnou, K. J. Am. Chem. Soc. 2005,
127, 18020. (i) Daugulis, O.; Zaitsev, V. G. Angew. Chem., Int. Ed. 2005,
44, 4046. (j) Leclerc, J.-P.; Fagnou, K. Angew. Chem., Int. Ed. 2006, 45,
7781. (k) Campeau, L.-C.; Parisien, M.; Jean, A.; Fagnou, K. J. Am. Chem.
Soc. 2006, 128, 581. (l) Deprez, N. R.; Kalyani, D.; Krause, A.; Sanford,
M. S. J. Am. Chem. Soc. 2006, 128, 4972. (m) Lafrance, M.; Rowley, C. N.;
Woo, T. K.; Fagnou, K. J. Am. Chem. Soc. 2006, 128, 8754. (n) Lafrance,
M.; Fagnou, K. J. Am. Chem. Soc. 2006, 128, 16496. (o) Mariampillai, B.;
Alliot, J.; Li, M.; Lautens, M. J. Am. Chem. Soc. 2007, 129, 15372. (p)
Turner, G. L.; Morris, J. A.; Greaney, M. F. Angew. Chem., Int. Ed. 2007,
4
2
6, 7996. (q) Chiong, H. A.; Pham, Q.-N.; Daugulis, O. J. Am. Chem. Soc.
007, 129, 9879. (r) Chuprakov, S.; Chernyak, N.; Dudnik, A. S.;
Gevorgyan, V. Org. Lett. 2007, 9, 2333. (s) Lebrasseur, N.; Larrosa, I. J. Am.
Chem. Soc. 2008, 130, 2926. (t) Campeau, L.-C.; Schipper, D. J.; Fagnou,
K. J. Am. Chem. Soc. 2008, 130, 3266. (u) Campeau, L.-C.; Bertrand-
Laperle, M.; Leclerc, J.-P.; Villemure, E.; Gorelsky, S.; Fagnou, K. J. Am.
Chem. Soc. 2008, 130, 3276. (v) Gorelsky, S. I.; Lapointe, D.; Fagnou, K.
J. Am. Chem. Soc. 2008, 130, 10848.
Listed in Scheme 1 are some examples of variations from
the “early” standard conditions. In the absence of nickel,
essentially none of the desired coupling was observed. Nickel
(
8) Selected examples of Rh catalysis: (a) Bedford, R. B.; Coles, S. J.;
Hursthouse, M. B.; Limmert, M. E. Angew. Chem., Int. Ed. 2003, 42, 112.
b) Oi, S.; Watanabe, S.; Fukita, S.; Inoue, Y. Tetrahedron Lett. 2003, 44,
665. (c) Lewis, J. C.; Wiedemann, S. H.; Bergman, R. G.; Ellman, J. A.
complexes such as Ni(cod)
2
(55%) and NiCl
2
(55%) were
2
·6H O
(
8
moderately effective, but Ni(NO
3
)
2
·6H O and NiSO
2
4
Org. Lett. 2004, 6, 35. (d) Wang, X.; Lane, B. S.; Sames, D. J. Am. Chem.
Soc. 2005, 127, 4996. (e) Wiedemann, S. H.; Lewis, J. C.; Ellman, J. A.;
Bergman, R. G. J. Am. Chem. Soc. 2006, 128, 2452. (f) Lewis, J. C.; Wu,
J. Y.; Bergman, R. G.; Ellman, J. A. Angew. Chem., Int. Ed. 2006, 45,
were by far less effective. We also determined that bidentate
nitrogen-based ligands furnish efficient catalysts. Whereas
bipy (64%), 4,4′-(t-Bu)
2
bipy (70%), and phenanthroline
1
589. (g) References 6a and 6b. (h) Proch, S.; Kempe, R. Angew. Chem.,
Int. Ed. 2007, 46, 3135. (i) Zhao, X.; Yu, Z. J. Am. Chem. Soc. 2008, 130,
136. (j) Berman, A. M.; Lewis, J. C.; Bergman, R. G.; Ellman, J. A. J. Am.
17
(
52%) promoted the coupling, diphosphines such as dppe
1
7
and dppf were less efficient under these conditions. The
coupling proceeded slightly better in dioxane (72%), but
DME, toluene, DMF, and NMP were unsuitable solvents.
The choice of basic additive turned out to be most critical:
when LiOt-Bu was replaced by other bases such as NaOt-
8
Chem. Soc. 2008, 130, 14926. (k) Lewis, J. C.; Bergman, R. G.; Ellman,
J. A. Acc. Chem. Res. 2008, 41, 1013.
(
9) Selected examples of Ru catalysis: (a) Oi, S.; Fukita, S.; Hirata, N.;
Watanuki, N.; Miyano, S.; Inoue, Y. Org. Lett. 2001, 3, 2579. (b) Oi, S.;
Ogino, Y.; Fukita, S.; Inoue, Y. Org. Lett. 2002, 4, 1783. (c) Ackermann,
L. Org. Lett. 2005, 7, 3123. (d) Ackermann, L.; Althammer, A.; Born, R.
Angew. Chem., Int. Ed. 2006, 45, 2619. (e) Ackermann, L.; Born, R.;
2 3 2 3
Bu, KOt-Bu, NaOMe, LiOH, Li CO , Cs CO , and DBU,
´
¨
Alvarez-Bercedo, P. Angew. Chem., Int. Ed. 2007, 46, 6364. (f) Ozdemir,
I.; Demir, S.; C¸ etinkaya, B.; Gourlaouen, C.; Maseras, F.; Bruneau, C.;
Dixneuf, P. H. J. Am. Chem. Soc. 2008, 130, 1156. (g) Ackermann, L.;
Mulzer, M. Org. Lett. 2008, 10, 5043. (h) Oi, S.; Funayama, R.; Hattori,
T.; Inoue, Y. Tetrahedron 2008, 64, 6051.
essentially none of the target product was produced. A
dramatic difference between LiOt-Bu and its sodium and
potassium analogues is notable. In view of efficiency, cost,
2
simplicity, and stability, we identified Ni(OAc) /bipy/LiOt-
(
10) Fujita, K.; Nonogawa, M.; Yamaguchi, R. Chem. Commun. 2004,
1
926.
(
11) Selected recent examples of other types of C-H bond arylation of
(13) (a) Norinder, J.; Matsumoto, A.; Yoshikai, N.; Nakamura, E. J. Am.
Chem. Soc. 2008, 130, 5858. (b) Wen, J.; Zhang, J.; Chen, S.-Y.; Li, J.;
Yu, X.-Q. Angew. Chem., Int. Ed. 2008, 47, 8897.
arenes: (a) Kakiuchi, F.; Matsuura, Y.; Kan, S.; Chatani, N. J. Am. Chem.
Soc. 2005, 127, 5936. (b) Stuart, D. R.; Fagnou, K. Science 2007, 316,
1
172. (c) Giri, R.; Maugel, N.; Li, J.-J.; Wang, D.-H.; Breazzano, S. P.;
(14) Selected examples of Ni-catalyzed cross-coupling using aryl
electrophiles: (a) Tamao, K.; Sumitani, K.; Kumada, M. J. Am. Chem. Soc.
1972, 94, 4374. (b) Corriu, R. J. P.; Masse, J. P. J. Chem. Soc., Chem.
Commun. 1972, 144. (c) Wenkert, E.; Michelotti, E. L.; Swindell, C. S.
J. Am. Chem. Soc. 1979, 101, 2246. (d) Dankwardt, J. W. Angew. Chem.,
Int. Ed. 2004, 43, 2428. (e) Tobisu, M.; Shimasaki, T.; Chatani, N. Angew.
Chem., Int. Ed. 2008, 47, 4866. (f) Guan, B.-T.; Wang, Y.; Li, B.-J.; Yu,
D.-G.; Shi, Z.-J. J. Am. Chem. Soc. 2008, 130, 14468. (g) Quasdorf, K. W.;
Tian, X.; Garg, N. K. J. Am. Chem. Soc. 2008, 130, 14422.
Saunders, L. B.; Yu, J.-Q. J. Am. Chem. Soc. 2007, 129, 3510. (d) Yang,
S.; Li, B.; Wan, X.; Shi, Z. J. Am. Chem. Soc. 2007, 129, 6066. (e) Shi, Z.;
Li, B.; Wan, X.; Cheng, J.; Fang, Z.; Cao, B.; Qin, C.; Wang, Y. Angew.
Chem., Int. Ed. 2007, 46, 5554. (f) Li, B.-J.; Tian, S.-L.; Fang, Z.; Shi,
Z.-J. Angew. Chem., Int. Ed. 2008, 47, 1115. (g) Yang, S.-D.; Sun, C.-L.;
Fang, Z.; Li, B.-J.; Li, Y.-Z.; Shi, Z.-J. Angew. Chem., Int. Ed. 2008, 47,
1
1
473. (h) Cho, S. H.; Hwang, S. J.; Chang, S. J. Am. Chem. Soc. 2008,
30, 9254.
(
12) (a) Do, H.-Q.; Daugulis, O. J. Am. Chem. Soc. 2007, 129, 12404.
(15) Ni-catalyzed aromatic C-H bond functionalization: (a) Clement,
N. D.; Cavell, K. J. Angew. Chem., Int. Ed. 2004, 43, 3845. (b) Nakao, Y.;
Kanyiva, K. S.; Oda, S.; Hiyama, T. J. Am. Chem. Soc. 2006, 128, 8146.
(c) Kanyiva, K. S.; Nakao, Y.; Hiyama, T. Angew. Chem., Int. Ed. 2007,
46, 8872. (d) Nakao, Y.; Kashihara, N.; Kanyiva, K. S.; Hiyama, T. J. Am.
Chem. Soc. 2008, 130, 2448. (e) Nakao, Y.; Kashihara, N.; Kanyiva, K. S.;
Hiyama, T. J. Am. Chem. Soc. 2008, 130, 16170.
(
b) Do, H.-Q.; Daugulis, O. J. Am. Chem. Soc. 2008, 130, 1128. (c)
Yoshizumi, T.; Tsurugi, H.; Satoh, T.; Miura, M. Tetrahedron Lett. 2008,
9, 1598. (d) Ackermann, L.; Potukuchi, H. K.; Landsberg, D.; Vicente, R.
4
Org. Lett. 2008, 10, 3081. (e) Do, H.-Q.; Kashif Khan, R. M.; Daugulis,
O. J. Am. Chem. Soc. 2008, 130, 15185. (f) Phipps, R. J.; Grimster, N. P.;
Gaunt, M. J. J. Am. Chem. Soc. 2008, 130, 8172.
1734
Org. Lett., Vol. 11, No. 8, 2009

 Canivet, Jerome
Canivet, Jerome
 Yamaguchi, Junichiro
Yamaguchi, Junichiro
 Ban, Ikuya
Ban, Ikuya
 Itami, Kenichiro
Itami, Kenichiro