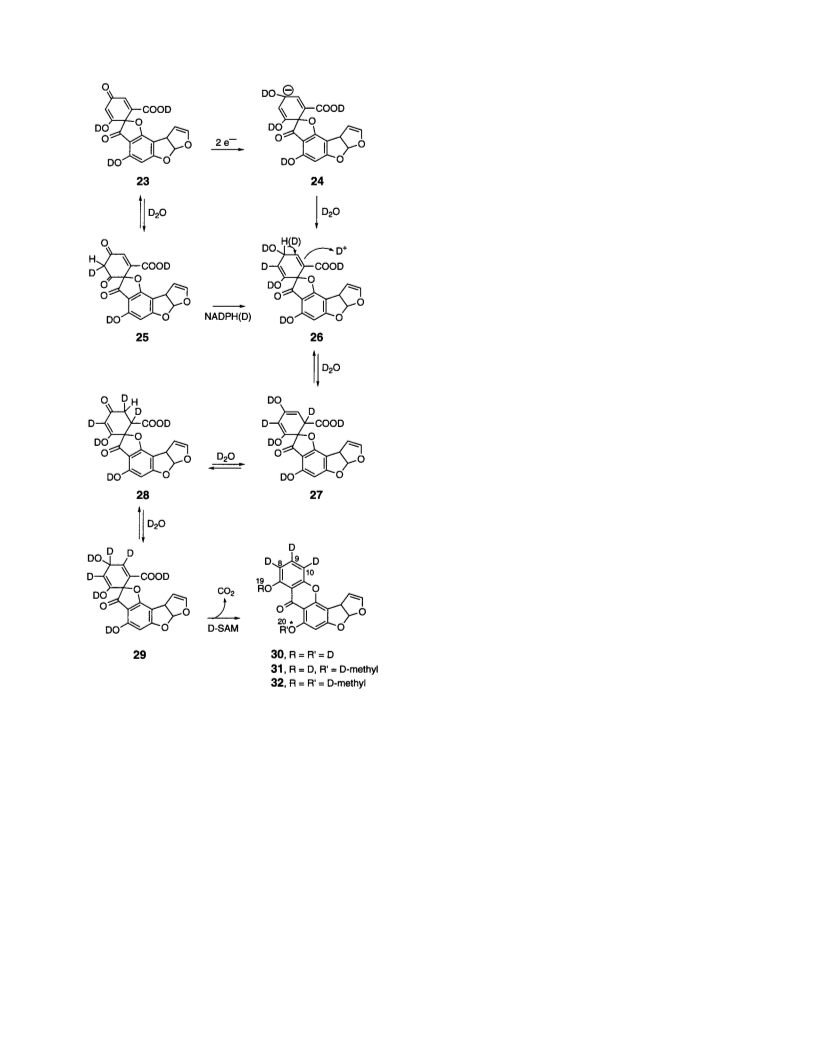
6238 J. Am. Chem. Soc., Vol. 120, No. 25, 1998
Watanabe and Townsend
the norsolorinic acid generating strain NOR-1 (ATCC 24690), and the
O-methylsterigmatocystin-producing mutant SRRC 2043 (ATCC 62882)
were obtained from the American Type Culture Collection (ATCC;
Rockville, MD). The A. parasiticus fatty acid synthase disruption
mutant strain DIS-2 was a generous gift from Professor J. E. Linz
(Michigan State University).
3. HPLC Analyses. Separation of aflatoxin and its intermediates
was achieved with a gradient that extended from 75% A/25% B f
40% A/60% B (A, 0.1% trifluoroacetic acid/water; B, acetonitrile) in
55 min. Samples were run at constant flow rate of 1 mL/min and
monitored at λ 310 nm.
NOR (2) f AFB1 (13). 1. Time-Course Analysis. SU-1 cell-
free extract devoid of small molecules was prepared as described above.
To 175 mL of extract was added 1.05 mL of 14C-labeled norsolorinic
acid (1 mg/mL, in acetone) and 1.4 mL of cofactor solution which
contained 1 mg of NADPH, 1 mg of SAM, and 1 mg of FAD in 1 mL
of distilled water. Aliquots (5 mL) were removed and quenched at
specific time points (see Figure 2) by vortexing with 1 mL of ethyl
acetate. The samples were microcentrifuged, and the organics were
transferred to fresh Eppindorf tubes and subsequently evaporated to
dryness. Reactions were resolubilized with 50 µL of acetonitrile and
analyzed by HPLC.
2. HPLC Analyses. To achieve separation of aflatoxin and its
intermediates, a linear gradient was employed that extended from 75%
A/25% B f 40% A/60% B (A, 0.1% trifluoroacetic acid/water; B,
acetonitrile) in 55 min. The solvent composition was then held at 40%
A/60% B from 55 to 59 min followed by a mixture of 20% A/80% B
which was initiated at 60 min from the point of injection. Samples
were analyzed by monitoring at λ 310 nm (flow rate 1 mL/min). These
lengthy run times were necessary to achieve separation of the broad
range of intermediates that vary in polarity and comprise the aflatoxin
pathway.
Media. The Adye and Mateles (AM) growth medium45 used to
culture the mycelial strains described above contained per liter sucrose,
50 g; potassium phosphate monobasic, 10 g; ammonium sulfate, 3 g;
magnesium sulfate (anhydrous) 1 g; and trace metals, 2 mL. The
replacement medium (RM)50 contained per liter (D2O) glucose or d7-
glucose, 1.62 g; potassium phosphate monobasic, 5 g; potassium
chloride, 0.5 g, magnesium sulfate (anhydrous), 0.25 g; and trace metals,
2 mL.
The AM growth medium (one liter, in 4-L Erlenmeyer flasks) was
autoclaved (140 °C, 20 psi) for 20 min and cooled to room temperature
prior to inoculation. The deuterated replacement medium was sterilized
by filtration through a 0.22 µm filtration unit (Corning; Corning, NY)
prior to use.
Culture Techniques. All fungi were grown on potato dextrose agar
plates50 for 4-7 days. The agar plates contained yeast extract, 2.5 g;
Bacto-agar, 2.5 g; potato dextrose agar, 19 g (Difco; Detroit, MI); and
distilled water, 500 mL (20 plates). The spores were suspended in a
solution of Tween 80 (0.05% v/v), 9 mL, which was subsequently
diluted in NaCl (0.85% w/v), 9 mL. A fraction of the spore suspension
(3 mL) was then transferred to 1 L of AM medium in 4-L Erlenmeyer
flasks unless stated otherwise. The flasks were incubated at 28-30
°C in the dark at 175 rpm for the requisite amount of time.
Cell-free Extract Preparation. 1. NOR f AFB1 Transformation.
A. parasiticus SU-1 cells were cultured for 84 h on AM growth medium
as described above. The mycelia were then vacuum filtered in a
Buchner funnel layered (4×) with cheesecloth and rinsed thoroughly
with distilled water. For each cell-free extract, 50 g of cells (damp
weight) were flash frozen in liquid nitrogen and pulverized in an
Osterizer blender with dry ice (cells were ground for 2 min in 30 s
cycles). The powdered mycelia were then suspended in 300 mL of
buffer [50 mM potassium phosphate, pH 7.5; 30% glycerol; 2 mM
â-mercaptoethanol; 100 µM phenylmethylsulfonyl fluoride (predis-
solved in a minimal amount of acetone); 100 µM benzamidine and 1
mM EDTA]. The mixture was stirred at 4 °C for 2 h and the cell
suspension was subsequently centrifuged for 20 min at 20 000g. The
decanted supernatant served as the crude cell-free extract. The protein
extract was then filtered through four layers of cheesecloth to remove
any residual cell debris, followed by dialysis (against 4 L of buffer)
with the Amicon RA 2000 apparatus employing a 10 000 molecular
weight cutoff membrane to produce the metabolite-free cell-free extract
in a volume of about 175 mL.
2. VA f OMST Transformation. A. parasiticus SRRC 2043 cells
(84 h) were used to prepare cell-free extracts utilized in NADPD-
labeling experiments. The extract was prepared as described above.
CFE Enzyme Assays. NOR f AFB1 Transformation. To the
metabolite-free SU-1 cell-free extract (5 mL) was added 20 µL of a 1
mg/mL solution (in acetone) of NOR (2), or any other aflatoxin
biosynthetic intermediate of interest and 40 µL of cofactor solution (1
mg NADPH; 1 mg SAM and 1 mg FAD in 1 mL of distilled water).
Reactions were typically incubated with mild shaking at 30 °C for ∼8
h to ensure maximal turnover of NOR (2) to AFB1 (13). Reactions
were quenched by vortexing with 1 mL of ethyl acetate. Emulsions
were eliminated by microcentrifugation of the samples at 14 000 rpm
for 2 min. The organic layer was then transferred to a fresh Eppindorf
tube and evaporated to dryness. Samples were solubilized with 20 µL
of ethyl acetate and subjected to TLC analysis or dissolved in 50 µL
of acetonitrile and analyzed by HPLC.
Biosynthesis of Radiolabeled Norsolorinic Acid. A. parasiticus
NOR-1 cells were initially cultured on AM medium by inoculating six
500-mL Erlenmeyer flasks each containing 125 mL of medium with
375 µL of the spore suspension. The mycelia were allowed to culture
for 35 h at 30 °C, shaking at 175 rpm. At 35 h of growth the mycelial
suspensions were vacuum filtered on cheesecloth, rinsed thoroughly
with distilled water, and subsequently transferred into RM medium
(500-mL flasks each containing 125 mL of medium) supplemented with
5 g of glucose/L. To each flask was then added 80 µCi of acetic acid,
sodium salt (1 mCi/1 mL of distilled water). The cells were allowed
to grow for an additional 65 h at 30 °C, 175 rpm. A second addition
of radiolabeled substrate (80 µCi [1-14C]acetate/flask) was supplied 5
h after the first 40 h of growth. The mycelial mats were harvested by
vacuum filtration, rinsed with distilled water, flash frozen in liquid
nitrogen, and steeped in acetone overnight. Metabolites were also
extracted from the media with ethyl acetate. The organic extracts from
the cells and the mediium were consolidated and concentrated in vacuo.
Purification of the labeled NOR (2) was most easily achieved by direct
recrystallization from acetone. The material was recrystallized to
1
constant specific activity (0.18Ci/mol). Norsolorinic acid H NMR
(400 MHz, d6-DMSO, natural abundance) δ: 7.12 (s, 1H, H-4), 7.05
(d, J ) 2.4 Hz, 1H, H-5), 6.53 (d, J ) 2.4 Hz, 1H, H-7), 2.73 (t, J )
7.2 Hz, 2H, H-2′), 1.59 (m, 2H, H-3′), 1.25 (m, 4H, H-4′/5′), 0.81 (t,
J ) 6.8 Hz, 3H, H-6′).
Biosynthesis of Versicolorin A. Versicolorin A (9) was prepared
by culturing Wh-1 cells (ATCC 36537) in four 4-L Erlenmeyer flasks
each containing 1 L of AM growth medium as described above. At
84 h of growth, the mycelia were vacuum filtered, rinsed with distilled
water, and flash frozen in liquid nitrogen. The mycelia were then
steeped in acetone overnight, the organic extracts were concentrated
in vacuo, and lyophilized to dryness. Purification was achieved by
flash silica column chromatography (8:1.8:0.2: hexane/ethyl acetate/
acetic acid). Versicolorin A 1H NMR (400 MHz, d6-DMSO) δ: 12.29
(s, 1H, OH), 12.00 (s, 1H, OH), 11.12 (br s, 1H, OH), 7.18 (s, 1H,
H-4), 7.14 (br d, J ) 2.5 Hz, 1H, H-5), 6.94 (d, J ) 7.1 Hz, 1H, H-1′),
6.72 (t, J ) 2.4 Hz, 1H, H-4′), 6.59 (br d, J ) 2.5 Hz, 1H, H-7), 5.41
(t, J ) 2.4 Hz, 1H, H-3′), 4.77 (dt, J ) 7.1, 2.4 Hz, 1H, H-2′).
Cell-Free Preparation of Demethylsterigmatocystin (10). The
diafiltered SU-1 cell-free extract was prepared as described above. To
175 mL of cell-free extract were added 700 µL of versicolorin A (1
mg/mL in acetone) and 1.4 mL of cofactor solution which contained 1
mg of NADPH and 1 mg of FAD in 1 mL of distilled water. The
rection was allowed to incubate at 30 °C for 24 h. The metabolites
were extracted from the protein suspension with ethyl acetate and the
organic extracts were concentrated in vacuo. Isolation of DMST (10)
TLC Analyses. For chromatographic identification of OMST (12)
and AFB1 (13) a ternary mixture of 6:3:1 chloroform/ethyl acetate/
formic acid was utilized. All other intermediates were visualized with
a solvent mixture of 8:1.8:0.2 hexane/ethyl acetate/acetic acid. Afla-
toxin and its intermediates can be easily visualized under longwave
UV light.
(50) Brobst, S. W.; Townsend, C. A. Can. J. Chem. 1994, 72, 200-
207.

 Watanabe, Coran M. H.
Watanabe, Coran M. H.
 Townsend, Craig A.
Townsend, Craig A.