Journal of Natural Products
Note
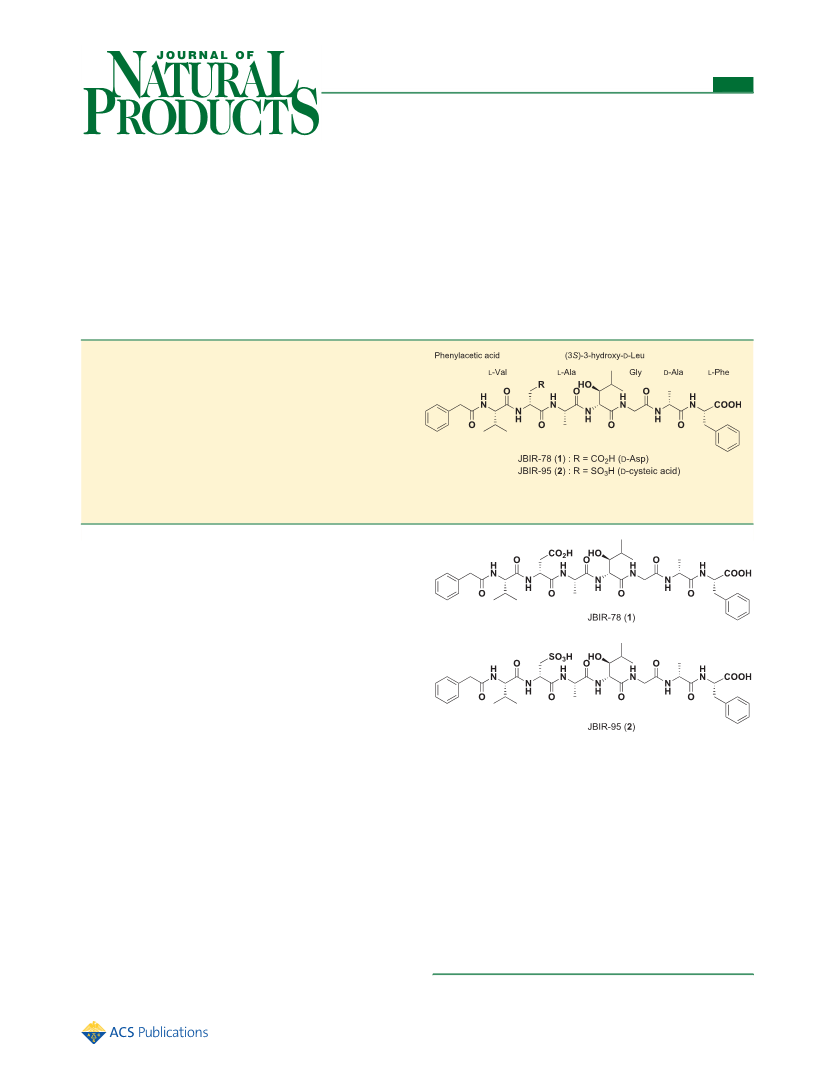
position was deduced to be S. Combining these results, the
absolute configuration of β-Hle was determined to be (S)-β-
hydroxy-D-leucine. The absolute configurations of the amino
acids in 2 were established in the same manner except for that
of the Cya moiety. The absolute configuration of Cya in 2 was
preparative reversed-phase HPLC using an XBridge C18 column
developed with 50% MeOH−H O containing 0.1% formic acid at a
2
flow rate of 10 mL/min to isolate 1 (5.4 mg, t = 30.1 min) and 2
R
(
14.2 mg, t = 12.4 min).
JBIR-78 (1): colorless amorphous solid; [α]
R
25
D
+30.7 (c 0.1,
MeOH); UV (MeOH) λ (log ε) 257 (3.76) nm; IR (KBr) ν
α
max
max
13
determined using N -(5-fluoro-2,4-dinitrophenyl)-L-valinamide
−1
1
1
650, 1560, 1180 cm ; H NMR (600 MHz, DMSO-d ) and
C
6
(
FDVA) instead of FDAA for Marfey’s method, because FDAA7
NMR (150 MHz, DMSO-d ), see Table 1; HRESIMS m/z 826.4000
6
+
derivatives of L-Cya and D-Cya were not separated by HPLC.
Accordingly, the Cya moiety was proven to be in the D-form.
Therefore, the absolute structures of 1 and 2 were conclusively
established.
[M + H] (calcd for C H N O , 826.3987).
40
56
7
12
25
D
JBIR-95 (2): colorless amorphous solid; [α]
+12.0 (c 0.1,
MeOH); UV (MeOH) λmax (log ε) 257 (3.64) nm; IR (KBr) νmax
−
1 1
1
660, 1540, 1230, 1180, 1050 cm ; H NMR (500 MHz, DMSO-d )
6
13
and C NMR (125 MHz, DMSO-d ) see Table 1; HRESIMS m/z
Secondary metabolites produced by Kibdelosporangium were
quite rare; approximately 30 compounds classified into only
6
+
8
62.3655 [M + H] (calcd for C H N O S, 862.3657).
39 56 7 13
1
1
Determination of Amino Acid Configurations. Compound 1
four groups including antibiotic YL 02107Q-A analogues,
1
2
13
14
(1.0 mg) was hydrolyzed in 0.2 mL of 6 N HCl at 40 °C for 6 h. After
the reaction mixture was concentrated in vacuo, the dried residue was
separated by reversed-phase HPLC using a CAPCELL PAK C18 MGII
column (5.0 μm, 4.6 i.d. × 150 mm; Shiseido) developed with a
aridicins, kibdelones, and cycloviracins, have been
reported. To the best of our knowledge, 1 and 2 were the
first examples of peptides, which were aligned alternately with L
and D forms of amino acids, from actinomycetes. In addition,
although phenylacetyl N-terminal masked peptides from
Streptomyces sp. such as antibiotic YF 044P-D, antibiotic L
74580, antibiotic YM 47690, and JBIR-96 have been
gradient solvent system of 10−75% MeOH−H O containing 0.1%
2
formic acid at a flow rate of 1 mL/min for 15 min to yield two
15
fragments, 3 (t = 9.6 min) and 4 (t = 11.4 min). These fragments
R
R
1
6
17
18
−
were characterized by HRESIMS data ([M − H] , m/z 434.2015,
1
−
calcd for C H N O , 434.2040, for 3 and [M − H] , m/z 606.2784
2
0
28
5
6
reported, N-phenylacetylated peptides, 1 and 2, were isolated
for the first time from Kibdelosporangium. The antimicrobial
activity of 1 and 2 against Micrococcus luteus, Candida albicans,
and Escherichia coli was examined. Only 1 was shown to be
weakly active (inhibition zone, 7.1 mm) against M. luteus.
calcd for C H N O , 606.2775, for 4). Fragments 3 and 4 were
2
8
40
5
10
hydrolyzed in 0.2 mL of 6 N HCl at 120 °C for 14 h. After the reaction
mixtures were concentrated in vacuo, the residues were added to 0.1 M
NaHCO (200 μL) with 0.2 mg of FDAA or 0.2 mg of FDVA. The
3
solutions were heated at 75 °C for 30 min. The FDAA derivatives of
Phe, Asp, and Val were analyzed using an LC-MS system under the
following conditions: column, CAPCELL PAK C18 MGII column;
flow rate, 1 mL/min; solvent, 70% (for Phe), 40% (for Asp), or 60%
EXPERIMENTAL SECTION
■
General Experimental Procedures. Optical rotation was
measured on a Horiba SEPA-300 polarimeter. The UV spectra were
measured on a Beckman Coulter DU730 UV/vis spectrophotometer.
FT-IR spectra were obtained using a Horiba FT-720 spectropho-
tometer. 13C (150 and 125 MHz) and H (600 and 500 MHz) NMR
spectra were recorded on a Varian NMR System 600 and 500 NB CL.
(
for Val) MeOH−H O containing 0.1% formic acid. The FDAA
2
derivatives of Ala were analyzed with the LC-MS system using an
ACQUITY UPLC BEH C18 column (2.1 i.d. × 100 mm; Waters)
1
developed with 20% CH
CN−H O containing 0.1% formic acid at a
3
2
flow rate of 0.4 mL/min. The FDVA derivatives of Cya were also
analyzed with the LC-MS system using a CROWNPAK CR(+)
column (4.0 i.d. × 150 mm; DAICEL Chemical Industries) developed
The samples were measured in DMSO-d , and the solvent peak was
6
used for spectra calibration (δ 39.7, δ 2.49 ppm). HRESIMS data
C
H
were recorded on a Waters LCT-Premier XE mass spectrometer, and
HRESIMS/MS data were recorded on a Waters SYNAPT G2.
Reversed-phase medium-pressure liquid chromatography (MPLC) was
performed on a Purif-Pack ODS 100 column (Shoko Scientific).
Analytical and preparative reversed-phase HPLC were executed on an
XBridge C18 column (5.0 μm, 4.6 i.d. × 150 mm; Waters) and XBridge
C18 column (5.0 μm, 19 i.d. × 150 mm; Waters), respectively, with a
Waters 2996 photodiode array detector and a Waters 3100 mass
detector. The reagents and solvents were of the highest grade available.
Fermentation. Kibdelosporangium sp. AK-AA56 was isolated from
a soil sample collected at Aomi, Tokyo Prefecture, Japan. The seed
medium comprised 1% starch (Kosokagaku), 1% Polypepton (Nihon
Pharmaceutical), 1% molasses (Dai-Nippon Meiji Sugar), and 1%
meat extract (Extract Ehlrich, Wako Pure Chemical Industries) and
was adjusted to pH 7.2 before sterilization. The production medium
consisted of 2% glycerol, 1% molasses, 0.5% casein (Kanto Chemical),
with 20% CH
3
CN−H O containing 0.4% TFA at a flow rate of 1 mL/
2
min. The retention times of the standard FDAA derivatives were as
follows: L-Phe, 5.0 min; D-Phe, 7.9 min; L-Ala, 3.4 min; D-Ala, 5.6 min;
L-Asp, 13.0 min; D-Asp, 18.7 min; L-Val, 6.2 min; and D-Val, 12.2 min.
The retention times of the standard FDVA derivatives of Cya were 7.0
and 6.4 min for L-Cya and D-Cya, respectively. The retention times of
the FDAA derivatives of 1 were as follows: Phe, 4.9 min; Ala1, 5.6 min;
Ala2, 3.6 min; Asp, 18.9 min; and Val, 6.4 min. Compound 2 was
hydrolyzed, derivatized with FDAA and FDVA, and analyzed in the
same manner as 1. The retention times of the FDAA and FDVA
derivatives of 2 were as follows: Phe, 4.9 min; Ala, 5.5 and 3.7 min;
Cya, 6.4 min; and Val, 6.4 min.
Compound 1 (1.0 mg) was reacted with (+)- or (−)-MTPA
chloride (10 μL) in pyridine (200 μL) at room temperature for 14 h.
The reaction mixture was concentrated to dryness, and the residue was
dissolved in 10 mL of EtOAc−H O (1:1). The (R)- or (S)-MTPA
2
0
.1% Polypepton, and 0.4% CaCO (Kozakai Pharmaceutical) and was
ester recovered in the organic layer was dried in vacuo and purified by
3
adjusted to pH 7.2 before sterilization. Strain AK-AA56 was cultivated
in 50 mL test tubes containing 15 mL of the seed medium. The test
tubes were shaken on a reciprocal shaker (320 rpm) at 27 °C for 3
days. Aliquots (2.5 mL) of the culture were transferred to 500 mL
Erlenmeyer flasks containing 100 mL of production medium and
cultured on a rotary shaker (180 rpm) at 27 °C for 5 days.
Purification of 1 and 2. The fermentation broth (2 L) of AK-
AA56 was centrifuged to obtain a mycelial cake, which was extracted
with acetone (500 mL). The extract was concentrated in vacuo, and the
residual aqueous concentrate was successively washed with EtOAc and
extracted with n-BuOH. The n-BuOH layer was then concentrated in
vacuo. The dried residue (660 mg) was subjected to reversed-phase
preparative reversed-phase HPLC using an XBridge C18 column (5.0
μm, 10 i.d. × 150 mm; Waters) with 75% MeOH−H
0.1% formic acid at a flow rate of 4 mL/min to yield the (R)- or (S)-
MTPA ester with t of 11.9 and 12.5 min, respectively.
O containing
2
R
Antimicrobial Activity. Antimicrobial activity against M. luteus
was measured using the paper disk method. M. luteus was cultured in
LB liquid medium consisting of 0.5% yeast extract (BD Biosciences),
1% tryptone (BD Biosciences), and 1% NaCl, at 28 °C for 24 h. A
paper disk (diameter 6 mm, Whatman) that contained 10 μg of 1 or 2
was placed on an LB agar plate including 0.2% of the liquid culture;
the plate was then incubated at 28 °C for 24 h. The antimicrobial
activity is expressed as the diameter (mm) of the inhibitory zone.
Erythromycin (10 μg) as a positive control showed an inhibition zone
of 25 mm against M. luteus.
MPLC using a MeOH−H O stepwise solvent system (0, 20, and 40%
2
MeOH). The 20% MeOH eluate (38 mg) was further purified by
2
83
dx.doi.org/10.1021/np2008279 | J. Nat. Prod. 2012, 75, 280−284

 Izumikawa, Miho
Izumikawa, Miho
 Takagi, Motoki
Takagi, Motoki
 Shin-Ya, Kazuo
Shin-Ya, Kazuo