Journal of the American Chemical Society
COMMUNICATION
arene-carbon and H to the RuꢀH could be a primary reason for
the marked difference in reactivity between the two catalyst
systems.
Sirlin, C.; Pfeffer, M. Organometallics 2008, 27, 5852–5859. (c) Baratta,
W.; Chelucci, G.; Magnolia, S.; Siega, K.; Rigo, P. Chem.—Eur. J. 2009,
15, 726–732.
(7) Related works; see: (a) Koike, T.; Ikariya, T. Organometallics
2005, 24, 724–730. (b) Sortais, J.-B.; Pannetier, N.; Holuigue, A.; Barloy,
L.; Sirlin, C.; Pfeffer, M.; Kyritsakas, N. Organometallics 2007,
26, 1856–1867.
(8) See the Supporting Information for details.
(9) Kitamura, M.; Tokunaga, M.; Ohkuma, T.; Noyori, R. Org. Synth.
1993, 71, 1–13.
(10) (a) Ikariya, T.; Ishii, Y.; Kawano, H.; Arai, T.; Saburi, M.;
Yoshikawa, S.; Akutagawa, S. J. Chem. Soc. Chem. Commun. 1985,
922–924. (b) King, S. A.; DiMichele, L. In Catalysis of Organic Reactions;
Scaros, M. G., Prunier, M. L., Eds.; Marcel Dekker: New York, 1994; pp
157ꢀ166. (c) Ohta, T.; Tonomura, Y.; Nozaki, K.; Takaya, H. Organo-
metallics 1996, 15, 1521–1523. (d) Mashima, K.; Nakamura, T.; Matsuo,
Y.; Tani, K. J. Organomet. Chem. 2000, 607, 51–56.
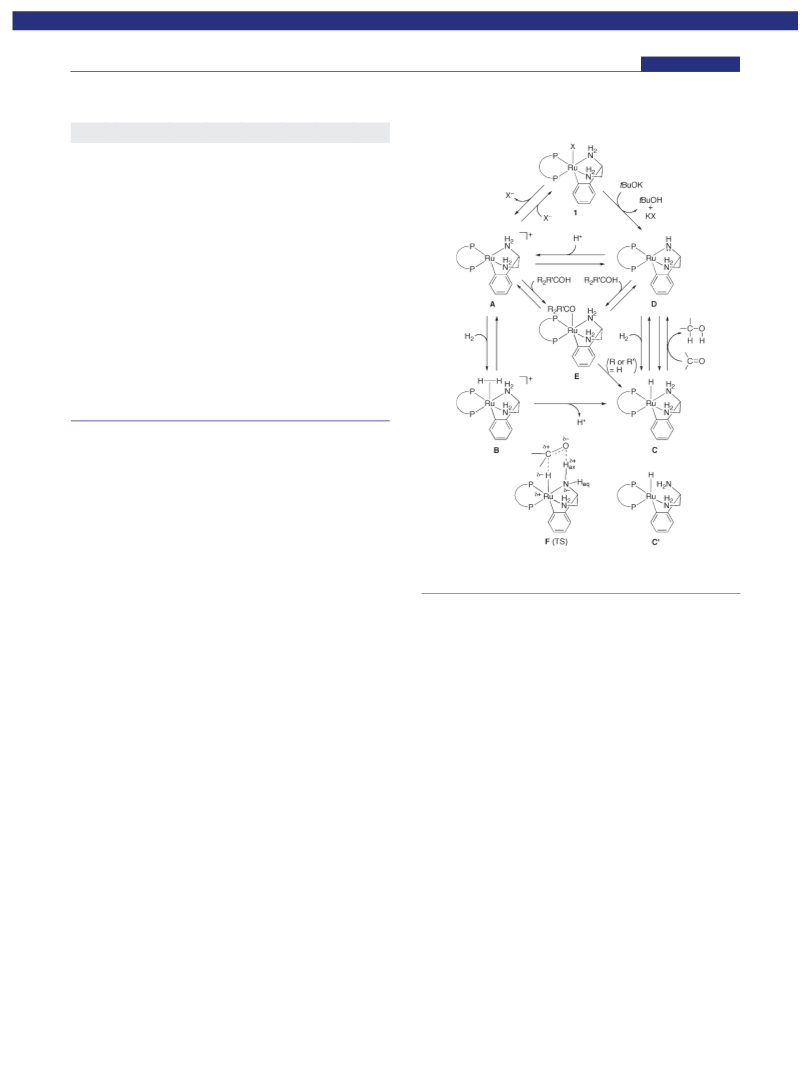
In conclusion, the newly devised ruthenabicyclic complexes
RuX(daipena)(xylbinap) (X = Cl, OTf) with base (t-C4H9OK,
DBU) exhibit remarkably high catalytic activity in the hydro-
genation of ketones. A turnover frequency of about 35 000 minꢀ1
is achieved in the best case. The enantioselectivity and scope for
the substrates are even superior to those of the previous RuCl2-
(xylbinap)(daipen)/t-C4H9OK system, which is one of the most
efficient catalysts. The catalytic cycle for this hydrogenation, in
which the ruthenabicyclic structure of the catalyst is maintained,
is proposed on the basis of the deuteration experiment and
spectroscopic analysis data.
’ ASSOCIATED CONTENT
(11) DM-SEGPHOS = (4,40-bi-1,3-benzodioxole)-5,50-diylbis(di-
(3,5-xylyl)phosphine), see: (a) Saito, T.; Yokozawa, T.; Ishizaki, T.;
Moroi, T.; Sayo, N.; Miura, T.; Kumobayashi, H. Adv. Synth. Catal. 2001,
343, 264–267. (b) Shimizu, H.; Nagasaki, I.; Matsumura, K.; Sayo, N.;
Saito, T. Acc. Chem. Res. 2007, 40, 1385–1393.
(12) The RuCl2(diphosphine)(diamine) complexes have the short-
er ClꢀRu bond length and the larger ClꢀRuꢀNꢀHax torsion angles;
for example: RuCl2[(R)-tolbinap][(R,R)-dpen] (2.41ꢀ2.43 Å; 20°),
RuCl2[(S)-tolbinap][(R)-dmapen] (2.41 Å; 15°), RuCl2[(S)-binap]-
[(R)-iphan] (2.42ꢀ2.43 Å; 6°). See: (a) Doucet, H.; Ohkuma, T.;
Murata, K.; Yokozawa, T.; Kozawa, M.; Katayama, E.; England, A. F.;
Ikariya, T.; Noyori, R. Angew. Chem., Int. Ed. 1998, 37, 1703–1707.
(b) Ooka, H.; Arai, N.; Azuma, K.; Kurono, N.; Ohkuma, T. J. Org. Chem.
2008, 73, 9084–9093. (c) Arai, N.; Akashi, M.; Sugizaki, S.; Ooka, H.;
Inoue, T.; Ohkuma, T. Org. Lett. 2010, 12, 3380–3383.
S
Supporting Information. Preparative methods and
b
properties of chiral ruthenabicyclic complexes 1, procedures
for asymmetric hydrogenation of ketones 3, NMR, GC, and
HPLC behavior, [R]D values of products, and the X-ray structure
of (SN,SP)-1c (CIF). This material is available free of charge via
’ AUTHOR INFORMATION
Corresponding Author
(13) The reaction temperature was immediately increased from 11
to 35 °C by the heat of reaction.
’ ACKNOWLEDGMENT
This work was supported by a Grant-in-Aid from the Japan
Society for the Promotion of Science (JSPS) (No. 21350048).
We thank Dr. Kimiko Hasegawa at the Rigaku Corporation
for her help in analyzing the X-ray diffraction data. We thank
also Messrs. Yoshihiro Yaguchi and Akihiro Kawaraya, Mrs.
Kyoko Zaizen, and the ruthenabicyclic catalysts (RUCY)
research members at the Takasago International Corporation
for measurement of NMR and mass spectra, and experimental
assistance.
(14) (a) Jiang, Q.; Jiang, Y.; Xiao, D.; Cao, P.; Zhang, X. Angew.
Chem., Int. Ed. 1998, 37, 1100–1103. (b) Li, W.; Hou, G.; Wang, C.;
Jiang, Y.; Zhang, X. Chem. Commun. 2010, 46, 3979–3981.
(15) Asymmetric hydrogenation of 3d with the diphosphine/diamineꢀ
RuCl2 complexes; see: Tsutsumi, K.; Katayama, T.; Utsumi, N.;
Murata, K.; Arai, N.; Ohkuma, T. Org. Process Res. Dev. 2009, 13, 625–628
and ref 12c.
(16) Asymmetric hydrogenation of 3e and 3f by using the BINAP/
IPHANꢀRuCl2 complexes; see: (a) Ohkuma, T.; Hattori, T.; Ooka, H.;
Inoue, T.; Noyori, R. Org. Lett. 2004, 6, 2681–2683. See also: (b) Zhang,
H.; Taketomi, T.; Yoshizumi, T.; Kumobayashi, H.; Akutagawa, S.;
Mashima, K.; Takaya, H. J. Am. Chem. Soc. 1993, 115, 3318–3319.
(c) Huang, H.; Okuno, T.; Tsuda, K.; Yoshimura, M.; Kitamura, M.
J. Am. Chem. Soc. 2006, 128, 8716–8717. (d) Ito, M.; Endo, Y.; Ikariya,
T. Organometallics 2008, 27, 6053–6055. (e) Li, Y.; Zhou, Y.; Shi, Q.;
Ding, K.; Noyori, R.; Sandoval, C. A. Adv. Synth. Catal. 2011, 353,
495–500.
(17) The reaction of 3h did not proceed with Cp*Ir(OTf)-
(MsDPEN), which is known as an excellent catalyst for hydrogenation
of R-hydroxy ketones, see: Ohkuma, T.; Utsumi, N.; Watanabe, M.;
Tsutsumi, K.; Arai, N.; Murata, K. Org. Lett. 2007, 9, 2565–2567.
See also: Kadyrov, R.; Koenigs, R. M.; Brinkmann, C.; Voigtlaender,
D.; Rueping, M. Angew. Chem., Int. Ed. 2009, 48, 7556–7559.
(18) The η2-H2 signal of RuH(η2-H2)[(R)-binap][(R,R)-dpen] was
observed at δ ꢀ0.66. See: Hamilton, R. J.; Leong, C. G.; Bigam, G.;
Miskolzie, M.; Bergens, S. H. J. Am. Chem. Soc. 2005, 127, 4152–4153.
(19) (a) Sandoval, C. A.; Ohkuma, T.; Mu~niz, K.; Noyori, R. J. Am.
Chem. Soc. 2003, 125, 13490–13503. See also: (b) Abdur-Rashid, K.;
Clapham, S. E.; Hadzovic, A.; Harvey, J. N.; Lough, A. J.; Morris, R. H.
J. Am. Chem. Soc. 2002, 124, 15104–15118.
’ REFERENCES
(1) Recent reviews: (a) Ohkuma, T.; Noyori, R. In The Handbook of
Homogeneous Hydrogenation; de Vries, J. G., Elsevier, C. J., Eds.; Wiley-
VCH: Weinheim, 2007; Vol. 3, pp 1105ꢀ1163. (b) Arai, N.; Ohkuma,
T. In Science of Synthesis: Stereoselective Synthesis 2; Molander, G. A., Ed.;
Thieme: Stuttgart, 2010; pp 9ꢀ57.
(2) Reviews: (a) Ohkuma, T. Proc. Jpn. Acad. Ser. B 2010, 86,
202–219. (b) Noyori, R.; Ohkuma, T. Angew. Chem., Int. Ed. 2001, 40,
40–73.
(3) XylBINAP = 2,20-bis(di-3,5-xylylphosphino)-1,10-binaphthyl,
DAIPEN = 1,1-di(4-anisyl)-2-isopropyl-1,2-ethylenediamine. DPEN =
1,2-diphenylethylenediamine.
(4) (a) Ohkuma, T.; Koizumi, M.; Doucet, H.; Pham, T.; Kozawa,
M.; Murata, K.; Katayama, E.; Yokozawa, T.; Ikariya, T.; Noyori, R.
J. Am. Chem. Soc. 1998, 120, 13529–13530. (b) Ohkuma, T.; Koizumi,
M.; Mu~niz, K.; Hilt, G.; Kabuto, C.; Noyori, R. J. Am. Chem. Soc. 2002,
124, 6508–6509.
(5) Mashima, K.; Kusano, K.; Ohta, T.; Noyori, R.; Takaya, H.
J. Chem. Soc., Chem. Commun. 1989, 1208–1210.
(6) Asymmetric reduction of alkyl aryl ketones with ruthenacyclic
catalysts; see: (a) Baratta, W.; Ballico, M.; Baldino, S.; Chelucci, G.;
Herdtweck, E.; Siega, K.; Magnolia, S.; Rigo, P. Chem.—Eur. J. 2008,
14, 9148–9160. (b) Pannetier, N.; Sortais, J.-B.; Dieng, P. S.; Barloy, L.;
10699
dx.doi.org/10.1021/ja202296w |J. Am. Chem. Soc. 2011, 133, 10696–10699

 Matsumura, Kazuhiko
Matsumura, Kazuhiko
 Arai, Noriyoshi
Arai, Noriyoshi
 Hori, Kiyoto
Hori, Kiyoto
 Saito, Takao
Saito, Takao
 Sayo, Noboru
Sayo, Noboru
 Ohkuma, Takeshi
Ohkuma, Takeshi