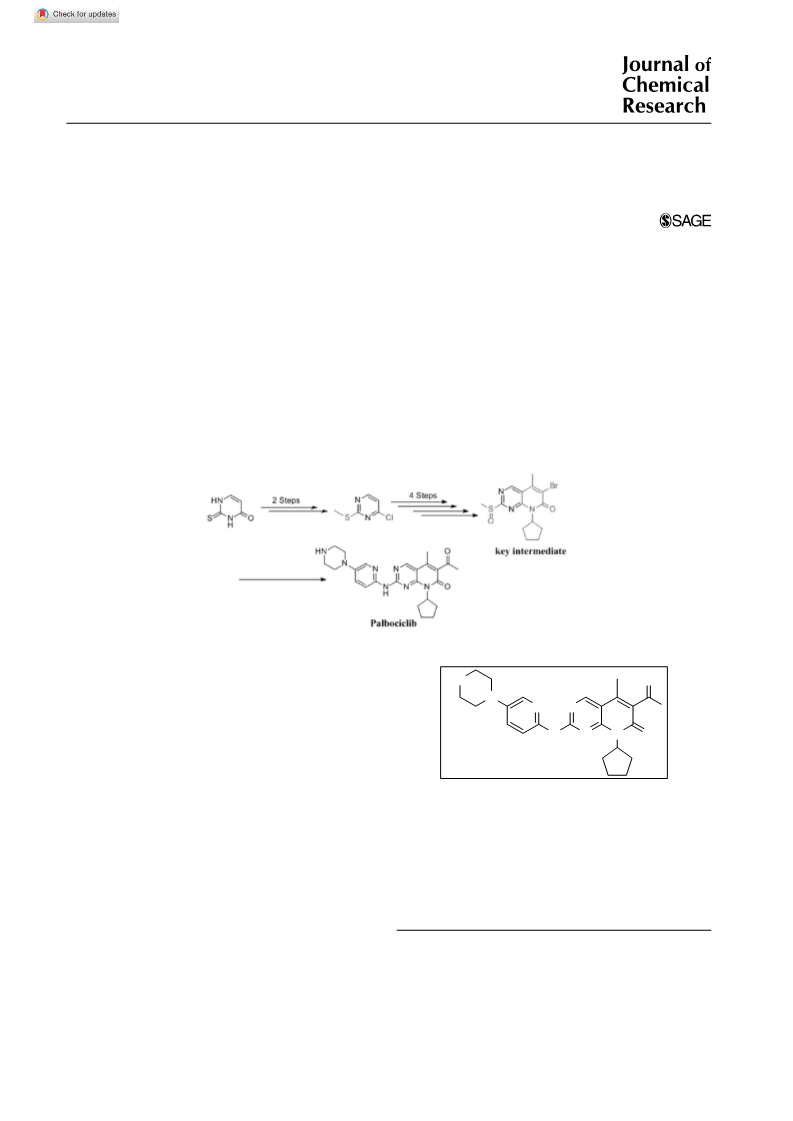
1
8
Journal of Chemical Research 43(1-2)
1
point meter and were uncorrected. Nuclear magnetic reso- 72.5%); m.p. 48.5–50.1°C. H NMR (300MHz, CDCl ) δ
3
1
3
nance (NMR) spectra were recorded on a Bruker Avance 8.54 (s, 1H), 2.55 (s, 3H). C NMR (75MHz, CDCl ) δ
3
+
DPX-300MHz/500MHz instrument in CDCl with tetra- 172.1, 159.5, 114.1, 14.7. HRMS ([M+H] ): m/z calcd for
3
methylsilane (TMS) as an internal reference and the chemi- C H BrClN S: 240.9025; found: 240.90203.
5
4
2
cal shifts (δ) were reported in parts per million (ppm).
High-resolution mass spectra (HRMS) were obtained from
Agilent 1100 LC/MS Spectrometry Services.
5
-Bromo-N-cyclopentyl-2-(methylthio)
pyrimidin-4-amine (17)
To a solution of 4-chloro-5-bromo-2-(methylthio)pyrimidine
2
-Methylthio-4-ketopyrimidine (14)
(16, 11.02g, 46.0mmol) in dioxane (40mL), DIPEA
(8.15mL, 46.0mmol) and cyclopentylamine (5.44mL,
To a solution of thiouracil (13, 10g, 78.1mmol), tetrabutyl-
ammonium bromide (12.59g, 39.1mmol), and K CO
55.2mmol) were added into the solution. After completion of
2
3
the addition, the reaction mixture was stirred at 100°C for 4h.
After quenching with water, the solution was extracted with
(16.2g, 117mmol) in dimethylformamide (DMF), dimethyl
carbonate (30mL) was slowly added dropwise when the
temperature was raised to 120°C. After addition, the mixture
was stirred at 120°C for 6h. After filtering K CO , water was
added to the filtrate which was then extracted with ethyl ace-
tate. The organic phases were combined, dried over Na SO ,
and concentrated in vacuo. The residue was purified by pass-
ing the organic extract through a silica gel column using
dichloromethane (DCM)/MeOH (75:1) to give the product
ethyl acetate and saturated NaHCO solution three times. The
3
combined organic phases were dried over MgSO , filtered,
4
2
3
and concentrated in vacuo. The residue was purified by pass-
ing the organic extract through a silica gel column using PE/
EA (15:1) to give product 17 as a yellow oil (11.99g, 90.4%).
2
4
1
H NMR (300MHz, CDCl ) δ 8.03 (s, 1H), 5.24 (s, 1H), 4.38
3
(dd, J=13.6, 6.8Hz, 1H), 2.48 (s, 3H), 2.09 (td, J=11.7,
6
2
5
.0Hz, 2H), 1.77–1.62 (m, 4H), 1.49 (dt, J=12.3, 6.2Hz,
1
4 as a white solid (10.27g, 92.6%); m.p. 204.4–205.3°C.
13
H); C NMR (75MHz, CDCl ) δ 170.3, 157.1, 154.8, 99.8,
1
3
H NMR (300MHz, CDCl ) δ 7.88 (d, J=6.5Hz, 1H), 6.23
(
3
+
2.9, 23.8, 14.4; HRMS ([M+Na] ): m/z calcd for
13
d, J=6.6Hz, 1H), 2.58 (s, 3H); C NMR (75MHz, CDCl )
3
+
C H BrN SNa : 311.9969; found: 311.99651.
+
10 14
3
δ 164.7, 162.8, 155.0, 111.1, 13.4; HRMS ([M+H] ): m/z
calcd for C H N OS: 143.0279; found: 143.02739.
5
6
2
8
-Cyclopentyl-5-methyl-2-(methylthio)
pyrido[2,3-d]pyrimidin-7(8H)-one (11)
4
-Chloro-2-(methylthio)pyrimidine (15)
To a solution of 5-bromo-N-cyclopentyl-2-(methylthio)pyrimi-
din-4-amine (17, 11.99g, 41.6mmol) and crotonic acid (5.37g,
To a solution of 2-methylthio-4-ketopyrimidine (14,
0.27g, 70.4mmol) in DCM (20mL) and DMF (10mL)
1
6
2.4mmol) in DMF (100mL), DIPEA(29.46mL, 166.4mmol)
was added. Then palladium dibenzylcarbonitrile (478mg,
.2mmol) and tri-o-tolylphosphorus (886mg, 3.0mmol) were
mixed solution, thionyl chloride (3.05mL, 42.2mmol)
solution was slowly added dropwise while heating. After
addition, the reaction mixture was stirred at 40°C for 3h.
After quenching with saturated NaHCO , the solution was
adjusted to a neutral pH and the solution was extracted
three times with DCM. The organic phases were washed
with saturated NaHCO solution and saturated NaCl solu-
tion and dried over anhydrous Na SO . After filtering to
remove the anhydrous Na SO , the solution was distilled at
1
added under a nitrogen atmosphere. After completion of the
addition, the reaction mixture was stirred at 130°C for 24h
under nitrogen protection. After 24h, acetic anhydride (15mL)
was added and the solution was stirred at 70°C for 4h. Then the
mixture was cooled, diluted with methyl tert-butyl ether
(MTBE; 100mL) and then extracted with NH Cl, NaHCO ,
3
3
2
4
4
3
2
4
and water. The organic phases were combined, dried over
4
0°C to remove solvents under atmospheric pressure to
MgSO , filtered, and concentrated in vacuo. The residue was
4
1
give product 15 as a yellow oil (10.19g, 87.6%). H NMR
purified by passing the organic extract through a silica gel col-
(
300MHz, CDCl ) δ 8.38 (d, J=5.2Hz, 1H), 6.99 (d,
3
umn using PE/EA (15:1) to give product 11 as a gray solid
1
3
J=5.2Hz, 1H), 2.53 (s, 3H); C NMR (75MHz, CDCl ) δ
1
calcd for C H ClN S: 160.9940; found: 160.99335.
1
3
(4.06g, 76.3%); m.p. 201.2–202.4°C. H NMR (300MHz,
+
74.0, 161.0, 158.0, 116.4, 14.3; HRMS ([M+H] ): m/z
CDCl ) δ 8.68 (s, 1H), 6.41 (s, 1H), 5.97–5.82 (m, 1H), 2.61 (s,
3
5
5
2
3H), 2.36 (d, J=20.9Hz, 5H), 2.06 (s, 2H), 1.87 (s, 2H), 1.69
1
3
(
1
(
d, J=10.3Hz, 3H); C NMR (75MHz, CDCl ) δ 163.0,
3
54.1, 144.0, 122.0, 111.0, 53.8, 28.4, 25.8, 17.2, 14.5.; HRMS
[M+Na] ): m/z calcd for (C H N OSNa ): 298.0990;
4
-Chloro-5-bromo-2-(methylthio)pyrimidine (16)
+
+
14
17
3
To a solution of 4-chloro-2-(methylthio)-pyrimidine (15, found: 298.09853.
0.19g, 63.4mmol) in MeOH (28mL) and MeCN (20mL)
mixed solution, NBS (13.55g, 76.1mmol) was added
2×3.76g). After the additions, the reaction mixture was
1
6
-Bromo-8-cyclopentyl-5-methyl-2-
methylsulfinyl)pyrido[2,3-d]pyrimidin-7(8H)
1)
(
(
stirred at room temperature. After quenching with saturated
Na SO solution, the solution was extracted with DCM and
(
2
3
saturated NaHCO solution three times. The combined To a solution of 8-cyclopentyl-5-methyl-2-(methylthio)
3
organic phases were dried over MgSO , filtered, and con- pyrido[2,3-d]pyrimidin-7(8H)-one (11, 4.06g, 14.7mmol)
4
centrated in vacuo. The residue was purified by passing the in MeCN (30mL), acetic acid (84µL, 1.47mmol), H O
2
organic extract through a silica gel column using PE/EA (265µL,14.7mmol), and NBS (7.87g, 44.2mmol) were
(
25:1) to give the product 16 as a white solid (11.02g, added. After completion of the addition, the reaction

 Li, Shuting
Li, Shuting
 Yang, Wanfeng
Yang, Wanfeng
 Ji, Min
Ji, Min
 Cai, Jin
Cai, Jin
 Chen, Junqing
Chen, Junqing