RSC Advances
DOI: 10.1039/C6RA19771J
Entry
Fatty acids
Fatty alcohol
Conv.Yield
30 We kindly acknowledge Director IIP for his kind permission to
publish these results. We are thankful to the Analytical Science
Division of IIP for providing help in the analyses of the
samples. SK kindly acknowledges CSIR, New Delhi for
providing fellowship.
b
c
(%) (%)
O
1
2
.
.
OH
95.78 95.2
96.83 96.1
(
(
CH
2
2
)
7
7
(CH
2
)
7
OH
OH
O
OH
OH
CH
)
(CH )
2 7
O
3
4
5
.
.
.
95.67 95.4
96.89 96.5
90.10 89.8
96.86 96.2
35 References
(
(
(
CH
2
CH
2
CH
2
)
7
)
7
)
7
(CH
(CH
(CH
2
)
7
OH
OH
OH
1
.
I. Diaz, C. MarquezꢀAlvarez, F. Mohino, J. PerezꢀPariente and E.
Sastre, J. Catal. 2000,193, 295ꢀ302.
O
,
(
CH
2
)
7
2 7
(CH )
OH
2
)
7
2. A. Demirbas, Biodiesel fuels from plant oils via catalytic and nonꢀ
catalytic supercritical alcohol transesterifications and other methods: a
survey, Energy Convers. Manage.,2003, 44, 2093–2109.
O
OH
4
4
0
5
2 7
)
3
.
A. Campanella, E. Rustoy, A. Baldessari, M. A. Baltanas, Lubricants
6
7
8
.
.
.
Acid oil (Major componentꢀC18:1;
other componentꢀ C18:1, C18:0,
C16:0)
OH
from chemically modified plant oils, Bioresour. Technol., 2010, 101
2
45–254.
4
.
S. Z. Erhant, B. K. Sharma, J. M. Perez, Ind. Crops Prod., 2006, 24
Acid oil (Major componentꢀC18:1;
other componentꢀ C18:1, C18:0,
C16:0)
96.77 96.2
97.10 96.8
292–299.
OH
5
6
.
.
S. Jumat, S. Nadia, Y. Emad, J. King Saud Univ.
J. M. Marchetti and A. F. Errazu, Biomass Bioenergy
95.
,
2012, 24, 11–17.
,
2008, 32, 892ꢀ
Acid oil (Major componentꢀC18:1;
other componentꢀ C18:2, C18:0,
C16:0)
8
7
.
Z. Helwani, M .R. Othman, N. Aziz, J. Kim and W. J. N. Fernando,
(CH
2
)
7
(CH
2
)
7
OH
50
App. Catal. A: Gen. 2009, 363, 1ꢀ10.
9
.
Acid oil (Major componentꢀC18:1;
other componentꢀ C16:0, C18:0,
C18:2)
86.23 85.5
88.34 88
90.99 90
8. H. Schuster, L. A. Rios, P. P. Wekjes and W. F. Hoelderich, App.
Catal. A: Gen. 2009, 348, 266ꢀ270.
9. M. J. Da Silva and A. L. Cardoso, J. Catal. 2013, Article ID 510509,
11 pages.
55 10. V. M. Mello, G. P. A. G. Pousa, M. S. C. Pereira, I. M. Dias and P. A.
Z. Suarez, Fuel Process. Tech. 2011, 92, 53ꢀ57.
11. W. D. Bassaert, D. E. De Vos, W. M. Van Rhijin, J. Bullen, P. J.
Grobet and P. A. Jacobs, J. Catal. 1999, 182, 156ꢀ164.
OH
1
1
0. Acid oil (Major componentꢀC18:1;
other componentꢀ C16:0, C18:0,
C18:2)
OH
OH
1. Acid oil (Major componentꢀC18:1;
other componentꢀ C16:0, C18:0,
C18:2)
1
2. I. Diaz, F. Mohino, J. P.Pariente and E. Sastre, App. Catal. A: Gen.
2001, 205, 19ꢀ30.
6
6
7
7
8
8
9
0
5
0
5
0
5
0
a
Reaction conditions: [TMG.HSO
4
]
IL (5 wt%), reaction time 6h,
13. Y. Leng, P. Jiang and J. A. Wang,. Catal. Commun. 2012, 25, 41ꢀ44.
14. A. S. Abbas and R.N. Abbas, Iraq. J. Chem. Pet. Eng. 2013, 14, 35ꢀ
o
b
temperature 150
C; conversion of fatty acid to ester was calculated by
4
3.
means of the acid value (AV). The acid value of the reaction mixture was
determined by the acid base titration technique. The conversion of FFA
1
1
5. D. Konwar, P. K. Gogoi, G. Borah, R. Baruah, N. Hazarika and R.
Borgohain, Ind. J. Chem. Tech. 2008, 15,75ꢀ78.
6. L. Geng, Y. Wang, G. Yu and Y. Zhu, Catal. Commun. 2011, 13, 26ꢀ
30.
5
(
reduction in acid value) was calculated using the following equation: XFFA
ꢀ a / a ; where a is the initial acidity and a is the acidity at time t (6 h);
Isolated yield
=
a
i
t
i
i
t
c
17. B. Aghabarari, N. Dorostkar, M. Ghiaci, S.G. Amini, E. Rahimi and
M. V. J. MarꢀtinezꢀHuerta, Taiwan Ins. Chem. Eng. 2014, 45, 431ꢀ
435.
1
8. J. Oh, S. Yang, C. Kim, I. Choi, J. H. Kim and H. Lee, App. Catal. A.
1
1
2
0
5
0
4. Conclusions
Gen. 2013, 455, 164ꢀ171.
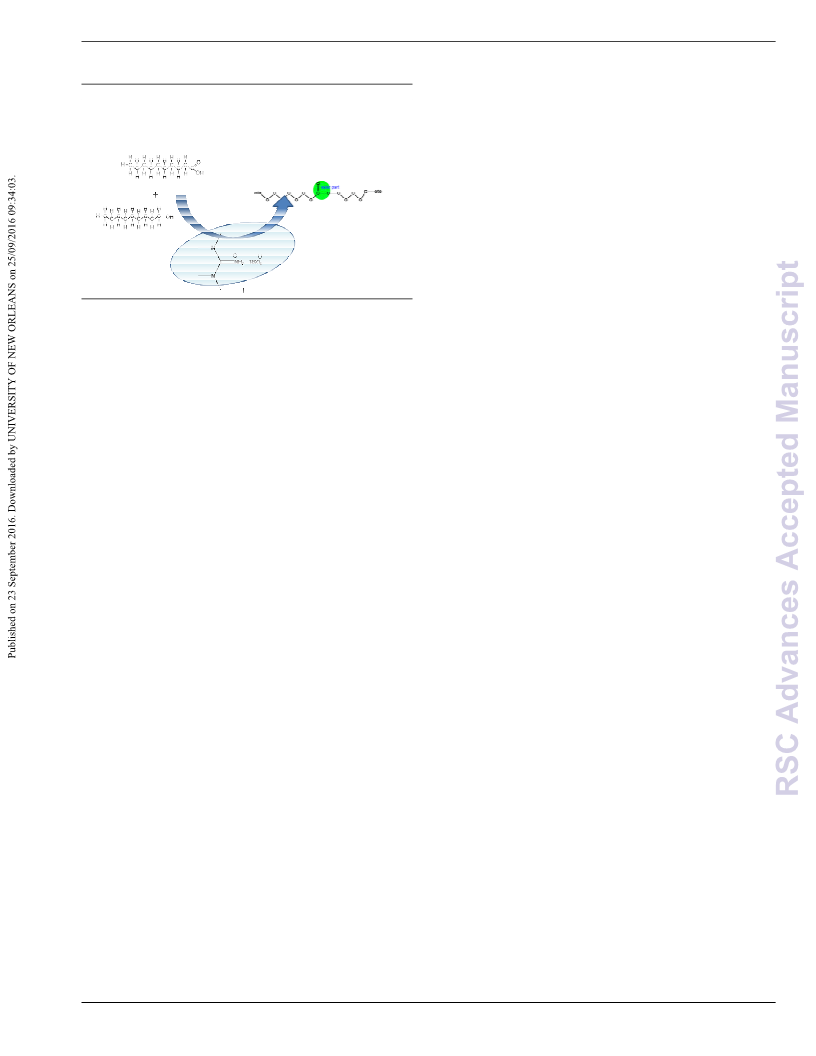
We have demonstrated for the first time the application of
tetramethylguanidine (TMG) derived ionic liquids as efficient and
reusable acid catalyst for the esterification of fatty acids with higher
alcohols to give higher alcohol esters of fatty acids for biolubricant
applications. These ionic liquids can readily be synthesized by
neutralizing equimolar amount of TMG with corresponding acids.
The synthesized 1,1,3,3ꢀtetramethylguanidinium hydrogen sulfate
1
9. B. Garg, T. Bisht and Y .C. Ling, Y. C. RSC Adv. 2014, 4, 57297ꢀ
57307.
20. J. H. Clark, T. J. Farmer, D. J. Macquarrie, Sherwood, J. Sustain.
Chem. Process. 2013, 1, 23, 1ꢀ3.
2
1. S. Kumar, R. K. Singh and S.L. Jain, RSC Adv. 2014, 4, 58238ꢀ
58242.
2
2. T. Welton, Chem. Rev. 1999, 99, 2071−2083.
23. J. Dupont, T. Itoh, P. Lozano, S. Malhotra; Environmentally Friendly
Syntheses Using Ionic Liquids; CRC Press, Taylor & Francis group,
2015.
24. Jiang, T.; Gao, H. X.; Han, B. X.; Zhao, G. Y.; Chang, Y. H.; Wu, W.
Z.; Gao, L.; Yang, G. Y. Tetrahedron Lett. 2004, 45, 2699ꢀ2701.
25. A. Shaabani, S. Samadi, Z. Badri, A. Rahmati, Catal. Lett. 2005, 104,
(TMG. HSO ) was found to have the highest activity among the
4
different ILs including (1,1,3,3ꢀtetramethylguanidinium acetate
(TMG.Ac), 1,1,3,3ꢀtetramethylguanidiniumꢀhydrogen phosphate
(TMG. H PO ) and 1,1,3,3ꢀtetramethylguanidinium trifluoroacetate
2 4
3
9ꢀ43.
(TMG. TFA). After the reaction, product was easily separated by
26. A. Shaabani and A. Rahmati, Catal. Lett. 2005, 100, 177ꢀ179.
2
7. A. Zhu, T. Jiang, B. Han, J. Huang, J. Zhang and X. Ma, New J.
Chem. 2006, 30, 736ꢀ740.
decantation and the recovered IL could be recycled for several runs
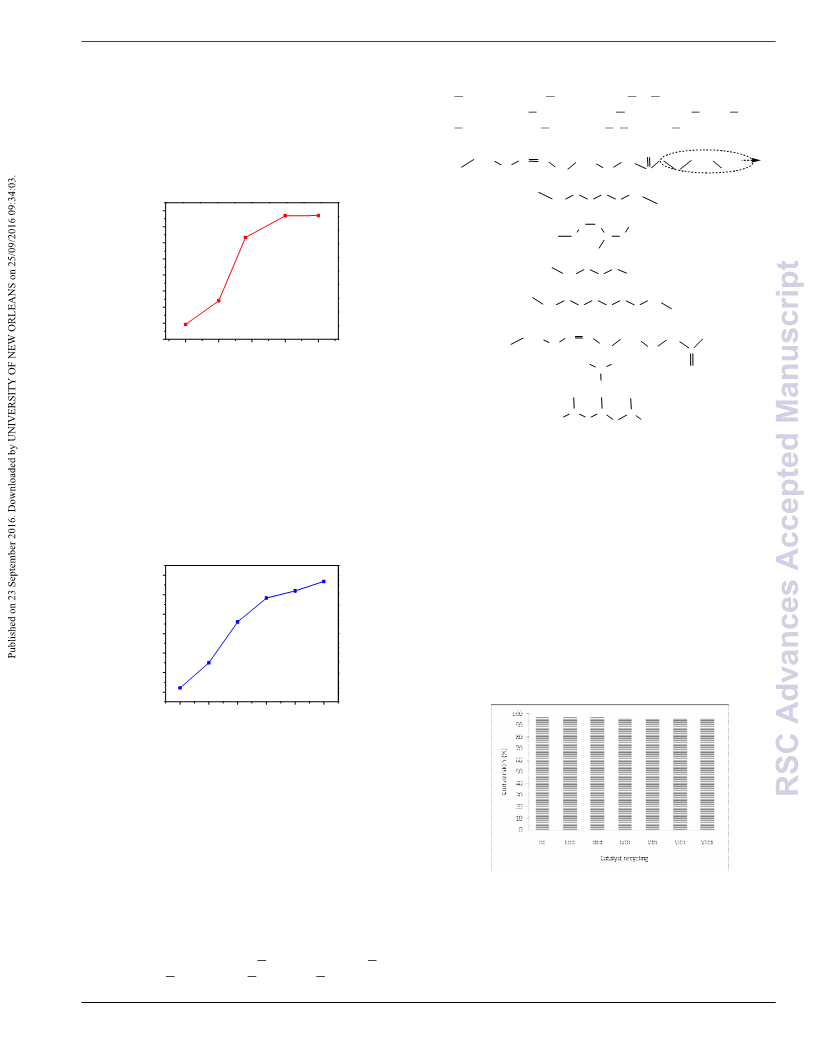
without any significant decrease in the catalytic activity.
28. H. Muthu, V. SathyaSelvabala, T. K.Varathachary, D. Kirupha
Selvaraj J. Nandagopal and S. Subramanian, Braz. J. Chem. Eng.
10, 27(4), 601ꢀ608.
2
5
,
0
95
Acknowledgements
4
| Journal Name, [year], [vol], 00–00
This journal is © The Royal Society of Chemistry [year]

 Porwal, Jyoti
Porwal, Jyoti
 Kumar, Subodh
Kumar, Subodh
 Kaul, Savita
Kaul, Savita
 Jain, Suman L.
Jain, Suman L.