DOI: 10.1002/cssc.201000105
A Recyclable TEMPO Catalyst for the Aerobic Oxidation of Sulfides to
Sulfoxides
[
a]
Tamilselvi Chinnusamy and Oliver Reiser*
Nowadays a wide range of reagents is available for almost
every conceivable type of oxidation, however, in many instan-
ces the use of these reagents still has disadvantages such as
high costs, difficulties in handling, toxicity, or lack of stability.
Therefore, the development of catalytic systems that display a
high atom economy while using comparatively harmless oxi-
dants, such as dioxygen, hydrogen peroxide, or hypochlorite, is
desired.
[
a]
Table 1. Aerobic oxidation of sulfides with TEMPO (Scheme 1).
1
2
[b]
Entry
R
R
Time [h]
Yield [%]
[c]
1
2
3
4
5
6
7
8
Ph
Ph
Ph
Ph
Me
Me
1.5
4
4
6
5
58
94
96
91
89
92
93
79
81
68
0
C
2
H
5
iPr
2-Naphthyl
Me
Me
Me
Me
Ph
4-Me-C
4-OMe-C
4-Cl-C
PhCH
Ph
6
H
4
3
3
The selective oxidation of sulfides into the corresponding
sulfoxides is one of the most important transformations in or-
ganic synthesis because of its usefulness for the preparation of
6
H
4
6
H
4
15
12
24
24
7
[
d]
[
9
1
2
d]
0
Ph
Me
[1,2]
biologically and medicinally valuable intermediates.
The
1
1
4-CHO-C
6
H
4
chemoselective oxidation of sulfides to sulfoxides can be ach-
ieved directly with molecular dioxygen, requiring high pressure
12
C
C
3
2
H
H
7
C
C
3
2
H
H
7
98
92
86
13
5
5
6
[
3,4]
14
iPr
iPr
6
and elevated temperature conditions.
Consequently, a
[
a] Sulfide (1.0 mmol) in AcOH (2 mL), Mn(NO
(NO O (2.0 mol%), TEMPO (1.0 mol%), oxygen atmosphere, 408C.
b] Isolated yield. [c] Determined by HNMR. [d] 5 mol% each of TEMPO,
Mn(NO O, and Co(NO
3 2
)2·4H O (2.0 mol%), Co-
number of alternative methods have been developed for this
[
5–7]
3 2
)2·6H
transformation,
among them a few catalytic systems that
1
[
[8]
allow the use of atmospheric oxygen as secondary oxidant.
3
)
2·4H
2
3 2
)2·6H O
Relevant to this work, the catalytic oxidation of sulfides to sulf-
oxides by oxoammonium ions generated from TEMPO by a
one-electron transfer mechanism was achieved either with
[
9]
sodium hypochlorite or with hydrogen peroxides in the pres-
obtained in 94% yield, with no observable oxidation to the
corresponding sulfone. Control experiments revealed that all
components of the catalyst mixture as well as molecular
oxygen are necessary for complete turnover of the substrate,
although the oxidation also proceeded to an appreciable
extent (30%) in the absence of TEMPO. Moreover, as reported
[
10]
ence of Cu-salen catalysts.
In the course of our ongoing studies to develop environ-
mentally benign catalytic processes for the oxidation of alco-
[
11]
hols, we found that Minisci’s conditions, that is, the aerobic
oxidation of alcohols with TEMPO in the presence of catalytic
[
12]
[12]
amounts of manganese and cobalt nitrate, can be extended
to the oxidation of sulfides. We report here the results of this
study by applying unmodified TEMPO and, more importantly, a
supported TEMPO catalyst of equal activity that can be readily
recovered by simple filtration, and reused.
for the analogous oxidation of alcohols, acetic acid proved
to be the solvent of choice: no oxidation took place either in
dichloromethane or acetonitrile.
Under the optimized conditions, a representative range of
sulfides was evaluated. We were pleased to find that diaryl-
and dialkyl- as well as arylalkylsulfides were smoothly convert-
ed into the corresponding sulfoxides in high yields. This was
also true for electron-rich sulfides; further oxidation to sulfones
was not observed. It was necessary to prolong reaction times
and/or catalyst loading for some, especially electron-deficient,
sulfides (Table 1, entries 8–10), to the point that oxidation did
not proceed at all (Table 1, entry 11).
Choosing thioanisole as the model substrate, we investigat-
ed a catalyst mixture comprising TEMPO (1 mol%), Mn-
(
NO ) 4H O (2 mol%), and Co(NO ) 6H O (2 mol%) in acetic
3 2· 2 3 2· 2
acid at 408C (Scheme 1). After a reaction time of 4 h under am-
bient pressure of dioxygen (Table 1, entry 2) the sulfoxide was
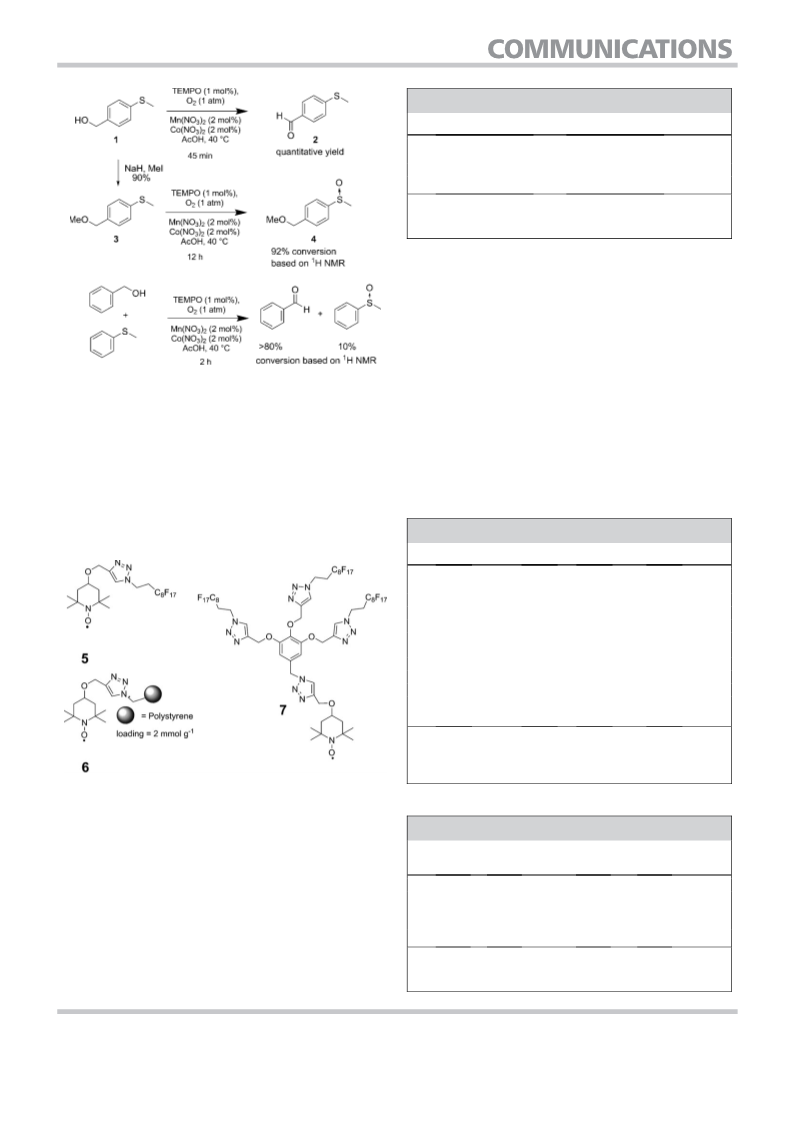
Because both alcohols and sulfides can be oxidized under
the reaction conditions described here, we subsequently inves-
tigated the chemoselectivity (Scheme 2). We found a high pref-
erence for alcohol oxidation when both functional groups
were present in a substrate. For example, oxidation of 1 gave
rise to 2 in quantitative yield, demonstrating that alcohol oxi-
dation is much more facile than sulfide oxidation under the ap-
plied reaction conditions. Nevertheless, oxidation of the sul-
fides occurred smoothly after protecting 1 as methyl ether 3,
suggesting that the hydroxymethyl group in 1 has not simply
deactivated the methyl sulfide by virtue of its electron-with-
Scheme 1. Aerobic oxidation of sulfides.
[a] T. Chinnusamy, Prof. O. Reiser
Institut fꢀr Organische Chemie
Universitꢁt Regensburg
Universitꢁtsstr. 31, 93053 Regensburg (Germany)
Fax: (+49)9419434121
E-mail: oliver.reiser@chemie.uni-regensburg.de
1
040
ꢀ 2010 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
ChemSusChem 2010, 3, 1040 – 1042

 Chinnusamy, Tamilselvi
Chinnusamy, Tamilselvi
 Reiser, Oliver
Reiser, Oliver