Please do not adjust margins
ChemComm
Page 4 of 5
DOI: 10.1039/C8CC05494K
COMMUNICATION
Journal Name
In conclusion, we reported an exceptional method for
Angew. Chem. Int. Ed., 2014, 53, 2745-2749 (Angew. Chem.,
014, 126, 2783-2787).
C. Berg, T. Braun, M. Ahrens, P. Wittwer and R. Herrmann,
Angew. Chem. Int. Ed., 2017, 56, 4300-4304 (Angew.
Chem., 2017, 129, 4364-4368).
2
activating the green-house gas SF
agent. On using electron rich N–heterocyclic carbenes a
mixture of 2,2-difluoroimidazolines or 2,2-
6
and using it as fluorinating
9
difluoroimidazolidines and the corresponding 2-thio 10 C. Berg, N. Pfister, T. Braun and B. Braun-Cula, Chem. Eur. J.,
derivatives were produced by photolysis with UV light at 311
nm. This photochemically generated product mixtures can be
employed for the deoxyfluorination of various alcohols and
benzoic acid to give fluorinated organic molecules, which
generally can serve as building blocks for fluorinated materials
2018, 24, 7985-7990.
1 T. A. McTeague and T. F. Jamison, Angew. Chem. Int. Ed.,
1
1
1
1
2
1
016, 55, 15072-15075 (Angew. Chem., 2016, 128, 15296 –
5299).
2 a) M. Rueping, P. Nikolaienko, Y. Lebedev and A. Adams,
Green Chem., 2017, 19, 2571-2575; b) P. K. D. Sevenard, A. A.
Kolomeitsev and G.-V. Röschenthaler, DE 10220901, 2004.
3 F. Buß, C. Mück-Lichtenfeld, P. Mehlmann and F. Dielmann,
Angew. Chem. Int. Ed., 2018, 57, 4951-4955 (Angew. Chem.,
2
5
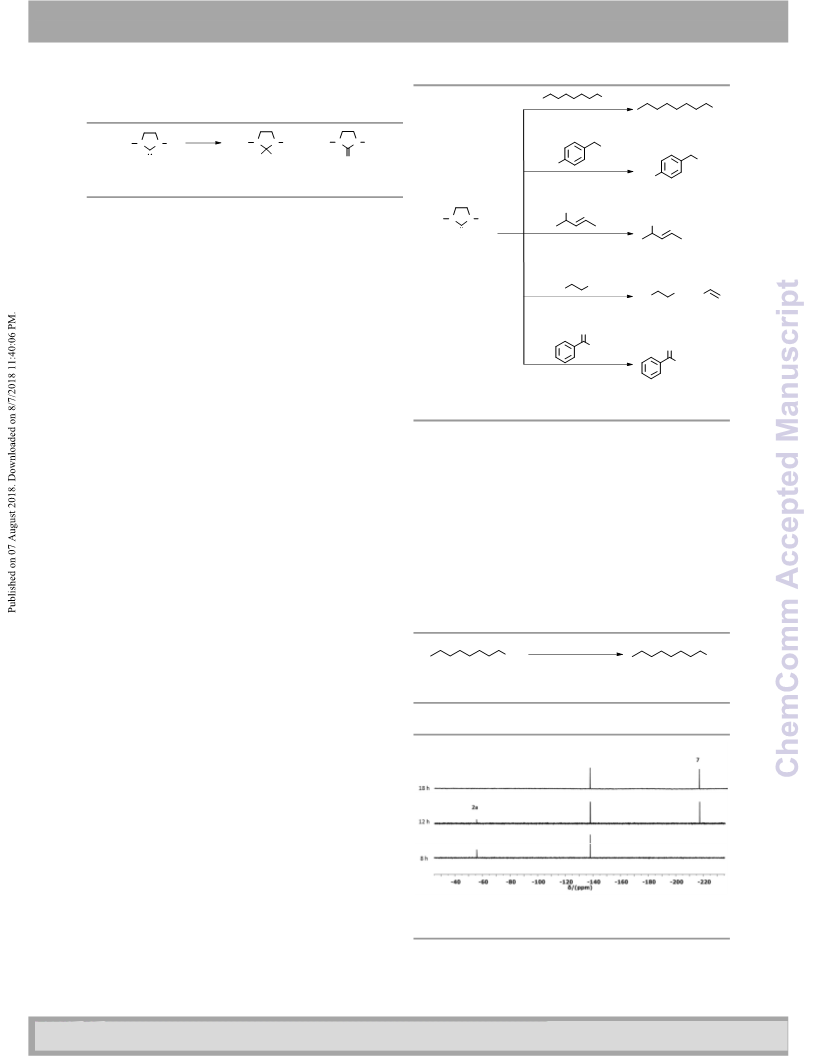
or bioactive compounds. Remarkably, an efficient one-pot
method was developed, which imparts the deoxyfluorination
of 1-octanol with SF
conditions. Thus, formally shuttling fluorides from SF
different organic substrates via an intermediate
difluoroimidazolidine opens up unprecedented opportunities
6
mediated by SIMes under photolytic
2
018, 130, 5045 –5049).
2
6
6
to
4 a) M. C. Leclerc, S. I. Gorelsky, B. M. Gabidullin, I. Korobkov
and R. T. Baker, Chem. Eur. J., 2016, 22, 8063-8067; b) S. D.
Paul Ursula and U. Radius, Chem. Eur. J., 2017, 23, 3993-
4009.
5 M. Paas, B. Wibbeling, R. Fröhlich and F. E. Hahn, Eur. J.
Inorg. Chem., 2006, 158-162.
6 a) P. Tang, W. Wang and T. Ritter, J. Am Chem. Soc., 2011,
133, 11482-11484; b) C. N. Neumann and T. Ritter, Acc.
Chem. Res, 2017, 50, 2822-2833.
2
7
6
to use SF as a versatile fluorination reagent.
1
1
We acknowledge the DFG (Deutsche Forschungsgemeinschaft)
for financial support as well as the graduate school SALSA
(School of Analytical Science Adlershof). We thank the Solvay
Fluor GmbH for a gift of SF , as well as Dr. Matthias Schwalbe,
6
1
1
7 A. Doddi, M. Weinhart, A. Hinz, D. Bockfeld, J. M.
Goicoechea, M. Scheer and M. Tamm, Chem. Commun.,
Dr. Iweta Pryjomska-Ray and Dr. Thoralf Krahl for their help
with the cyclic voltammetry and the luminescence emission
measurements.
2
017, 53, 6069-6072.
8 a) J. Bittner, J. Fuchs and K. Seppelt, Z. Anorg. Allg. Chem.,
988, 557, 182-190; b) T. Goettel James, N. Kostiuk and M.
1
Gerken, Angew. Chem. Int. Ed., 2013, 52, 8037-8040 (Angew.
Chem., 2013, 125, 8195 –8198).
Conflicts of interest
1
9 V. Balzani, G. Bergamini, S. Campagna and F. Puntoriero,
Photochemistry and Photophysics of Coordination
Compounds I, Springer, Berlin Heidelberg, 2007, 280, 1-36.
0 N. Kuhn, H. Bohnen, J. Fahl, D. Bläser and R. Boese, Chem.
Ber., 1996, 129, 1579-1586.
1 a) G. Meißner, K. Kretschmar, T. Braun and E. Kemnitz,
Angew. Chem. Int. Ed., 2017, 56, 16338-16341 (Angew.
Chem., 2017, 129, 16556-16559); b) G. Meißner, D. Dirican,
There are no conflicts to declare.
2
2
References
1
2
3
a) K. Seppelt, Chem Rev 2015, 115, 1296-1306; b) J. R. Case
and F. Nyman, Nature, 1962, 193, 473.
L. G. Christophorou, J. K. Olthoff and R. J. V. Brunt, IEEE
Electrical Insulation Magazine, 1997, 13, 20-24.
a) A. A. Lindley and A. McCulloch, J. Fluorine Chem., 2005,
C. Jäger, T. Braun and E. Kemnitz, Cat. Sci. Technol., 2017,
348-3354.
2 a) M. Hanack and J. Ullmann, J. Org. Chem., 1989, 54, 1432-
435; b) M. F. Kuehnel, D. Lentz and T. Braun, Angew. Chem.
Int. Ed., 2013, 52, 3328-3348 (Angew. Chem., 2013, 125
412-3433); c) T. Ahrens, J. Kohlmann, M. Ahrens and T.
Braun, Chem. Rev., 2015, 115, 931-972.
7,
3
2
126, 1457-1462; b) G. P. Stiller, T. von Clarmann, M. Höpfner,
1
N. Glatthor, U. Grabowski, S. Kellmann, A. Kleinert, A. Linden,
M. Milz, T. Reddmann, T. Steck, H. Fischer, B. Funke, M.
López-Puertas and A. Engel, Atmos. Chem. Phys., 2008, 8,
,
3
6
77-695.
2
3 H. Hayashi, H. Sonoda, K. Fukumura and T. Nagata, Chem.
Commun., 2002, 1618-1619.
4 Y. Kim and E. Lee, Chem. Commun., 2016, 52, 10922-10925.
5 a) H. Amii and K. Uneyama, Chem. Rev., 2009, 109, 2119-
4
5
a) P. J. Amal Joseph and S. Priyadarshini, Org. Process Res.
Dev., 2017, 21, 1889-1924; b) A. C. Sather and S. L.
Buchwald, Acc. Chem. Res., 2016, 49, 2146-2157; c) J. Miró,
C. del Pozo, Chem. Rev., 2016, 116, 11924-11966; d) M. G.
Campbell and T. Ritter, Chem. Rev., 2015, 115, 612-633.
2
2
2
183; b) P. Kirsch, Modern Fluoroorganic Chemistry:
Synthesis Reactivity, Applications, Wiley-
VCH, Weinheim, 2004.
a) H. Deubner and F. Kraus, Inorganics, 2017,
Demitras and A. G. MacDiarmid, Inorg. Chem., 1964,
199; c) P. Holze, B. Horn, C. Limberg, C. Matlachowski and S.
5
, 68; b) G. C.
3, 1198-
2
2
6 S. J. Ryan, S. D. Schimler, D. C. Bland and M. S. Sanford, Org.
Lett., 2015, 17, 1866-1869.
7 T. Böttcher, O. Shyshkov, M. Bremer, B. S. Bassil and G.-V.
1
Mebs, Angew. Chem. Int. Ed., 2014, 53, 2750-2753 (Angew.
Chem., 2014, 126, 2788-2791); d) B. G. Harvey, A. M. Arif, A.
Glockner and R. D. Ernst, Organometallics, 2007, 26, 2872-
Röschenthaler
,
Organometallics, 2012, 31, 1278–1280.
2879; e) R. Basta, B. G. Harvey, A. M. Arif and R. D. Ernst, J.
Am. Chem. Soc., 2005, 127, 11924-11925.
6
7
8
W.-T. Tsai, J. Fluorine Chem., 2007, 128, 1345-1352.
L. Zámostná and T. Braun, Nachr. Chem., 2016, 64, 829-835.
a) M. Wozniak, T. Braun, M. Ahrens, B. Braun-Cula, P.
Wittwer, R. Herrmann and R. Laubenstein, Organometallics,
2
018, 37, 821-828; b) L. Zámostná and T. Braun, Angew.
Chem. Int. Ed., 2015, 54, 10652-10656 (Angew. Chem., 2015,
27, 10798-10802); c) L. Zámostná, T. Braun and B. Braun,
1
4
| J. Name., 2012, 00, 1-3
This journal is © The Royal Society of Chemistry 20xx
Please do not adjust margins

 Tomar, Pooja
Tomar, Pooja
 Braun, Thomas
Braun, Thomas
 Kemnitz, Erhard
Kemnitz, Erhard