pubs.acs.org/joc
N-Heterocyclic Carbene-Ruthenium Complexes for the Racemization
of Chiral Alcohols
Johann Bosson and Steven P. Nolan*
EaStCHEM School of Chemistry, University of St. Andrews, St. Andrews KY16 9ST, U.K.
Received January 21, 2010
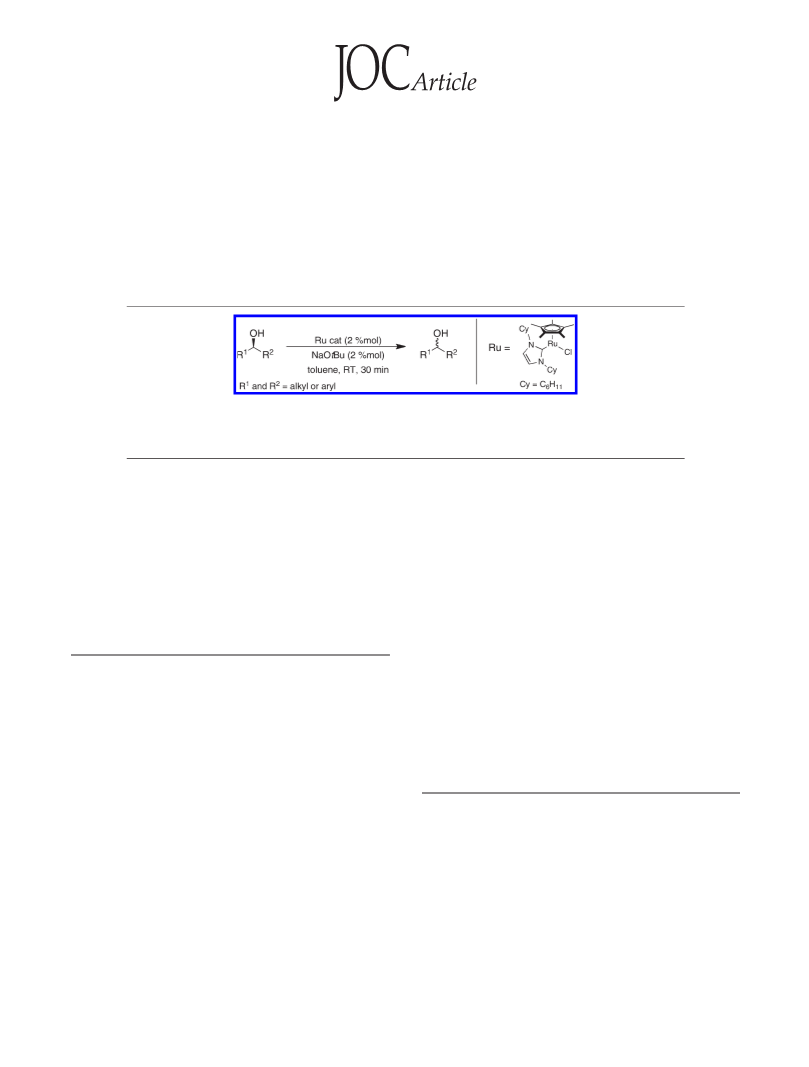
The activity of well-defined 16-electron ruthenium complexes bearing an N-heterocyclic carbene ligand
in the racemization of chiral alcohols is reported. Mechanistic considerations are also presented.
Introduction
The best performing catalysts enabling alcohol racemiza-
tion3 are based on ruthenium,4,5 and among these, the Shvo
catalyst (A, Figure 1)6,7 has been successfully applied to
Racemization reactions are intricate components of proto-
cols making use of chiral synthons in synthesis.1 More specifi-
cally, in the field of dynamic kinetic resolution (DKR), a
racemization catalyst is directly introduced into a kinetic
resolution reaction, allowing the complete conversion of the
racemic substrate into an enantiomerically pure derivative.2
Nevertheless, the development of new catalysts able to race-
mize substrates under mild conditions remains a challenge.
racemization reactions as part of DKR processes as dis-
€
8
closed by Backvall. The activity of the dimeric A involves,
upon thermal activation, the liberation of two ruthenium
species: an 18-electron ruthenium(II) complex A1 and a
16-electron ruthenium(0) complex A2 (Figure 1).6 Transfer
dehydrogenation of a chiral alcohol by A2 yields the corres-
ponding ketone which then undergoes a non-stereoselective
transfer hydrogenation reaction mediated by A1, leading to
the corresponding racemized alcohol.9 Park et al. have
developed a family of 18-electron ruthenium(II) complexes
analogous to the A1 monomer of the Shvo catalyst
and applied them to racemization in DKR reactions (B,
(1) Ebbers, E. J.; Ariaans, G. J. A.; Houbiers, J. P. M.; Bruggink, A.;
Zwanenburg, B. Tetrahedron 1997, 53, 9417–9476.
€
(2) For reviews on DKR: (a) Huerta, F. F.; Minidis, A. B. E.; Backvall,
J.-E. Chem. Soc. Rev. 2001, 30, 321–331. (b) Pamies, O.; Backvall, J.-E.
Chem. Rev. 2003, 103, 3247–3261. (c) Pellissier, H. Tetrahedron 2008, 64,
1563–1601.
(3) For selected examples of alcohol racemization catalysts: (a) Dinh,
P. M.; Howarth, J. A.; Hudnott, A. R.; Williams, J. M. J.; Harris, W.
Tetrahedron Lett. 1996, 37, 7623–7626. For Al: (b) Berkessel, A.;
€
Figure 1).10 Backvall has reported11 a remarkable 18-electron
€
ruthenium(II) catalyst analogue of A1 (C, Figure 1) able to
racemize alcohol at rt within minutes.12 Racemization cata-
lysts derived from the Shvo catalyst are based on monomer
€
Sebastian-Ibarz, M. L.; Muller, T. N. Angew. Chem., Int. Ed. 2006, 45,
6567–6570. For Ir: (c) Jerphagnon, T.; Gayet, A. J. A.; Berthiol, F.; Ritleng,
V.; Mrsic, N.; Meetsma, A.; Pfeffer, M.; Minnaard, A. J.; Feringa, B. L.;
De Vries, J. G. Chem.;Eur. J. 2009, 15, 12780–12790. For Rh: (d) Marr,
A. C.; Pollock, C. L.; Saunders, G. C. Organometallics 2007, 26, 3283–3285.
For V: (e) Wuyts, S.; Wahlen, J.; Jacobs, P. A.; De Vos, D. E. Green Chem.
2007, 9, 1104–1108. For non-metal-catalyzed racemization: (f) Wuyts, S.;
De Temmerman, K.; De Vos, D. E.; Jacobs, P. A. Chem.;Eur. J. 2005, 11,
386–397. For a review on enzymatic racemization: (g) Schnell, B.; Faber, K.;
Kroutil, W. Adv. Synth. Catal. 2003, 345, 653–666.
(6) Shvo, Y.; Czarkie, D.; Rahamim, Y.; Chodosh, D. F. J. Am. Chem.
Soc. 1986, 108, 7400–7402.
(7) Karvembu, R.; Prabhakaran, R.; Natarajan, K. Coord. Chem. Rev.
2005, 249, 911–918.
€
(8) Larsson, A. L. E.; Persson, B. A.; Backvall, J.-E. Angew. Chem., Int.
Ed. 1997, 36, 1211–1212.
(4) For a review on racemization catalysts in DKR: (a) Ahn, Y.; Ko,
S.-B.; Kim, M.-J.; Park, J. Coord. Chem. Rev. 2008, 252, 647–658. For a
review on ruthenium-based racemization catalysts: (b) Karvembu, R.;
Prabhakaran, R.; Muthu Tamizh, M.; Natarajan, K. C. R. Chim. 2009, 12,
951–962. See also: (c) Mavrynsky, D.; Sillanpaa, R.; Leino, R. Organo-
metallics 2009, 28, 598–605.
(5) Other organometallic species can facilitate racemization processes.
For examples of Ir-based systems, see: (a) Stirling, M.; Blacker, A. J.; Page,
M. I. Tetrahedron Lett. 2007, 48, 1247–1250. (b) Blacker, A. J.; Stirling, M. J.;
(9) (a) Casey, C. P.; Singer, S. W.; Powell, D. R.; Hayashi, R. K.; Kavana,
M. J. Am. Chem. Soc. 2001, 123, 1090–1100. (b) Casey, C. P.; Beetner, S. E.;
Johnson, J. B. J. Am. Chem. Soc. 2008, 130, 2285–2295.
(10) (a) Choi, J. H.; Kim, Y. H.; Nam, S. H.; Shin, S. T.; Kim, M.-J.; Park,
J. Angew. Chem., Int. Ed. 2002, 41, 2373–2376. (b) Choi, J. H.; Choi, Y. K.;
Kim, Y. H.; Park, E. S.; Kim, E. J.; Kim, M.- J.; Park, J. J. Org. Chem. 2004,
69, 1972–1977.
€
(11) Martin-Matute, B.; Edin, M.; Bogar, K.; Backvall, J.-E. Angew.
Chem., Int. Ed. 2004, 43, 6535–6539.
ꢀ
Page, M. I. Org. Process Res. Dev. 2007, 11, 642–648. (c) Corberan, R.; Peris,
E. Organometallics 2008, 27, 1954–1958.
€
(12) Csjernyik, G.; Bogar, K.; Backvall, J.-E. Tetrahedron Lett. 2004, 45,
6799–6802.
DOI: 10.1021/jo1001005
r
Published on Web 02/19/2010
J. Org. Chem. 2010, 75, 2039–2043 2039
2010 American Chemical Society

 Bosson, Johann
Bosson, Johann
 Nolan, Steven P.
Nolan, Steven P.