
ACS Catalysis p. 5812 - 5821 (2015)
Update date:2022-08-16
Topics:
 Vidal, Juan D.
Vidal, Juan D.
 Climent, Maria J.
Climent, Maria J.
 Concepcion, Patricia
Concepcion, Patricia
 Corma, Avelino
Corma, Avelino
 Iborra, Sara
Iborra, Sara
 Sabater, Maria J.
Sabater, Maria J.
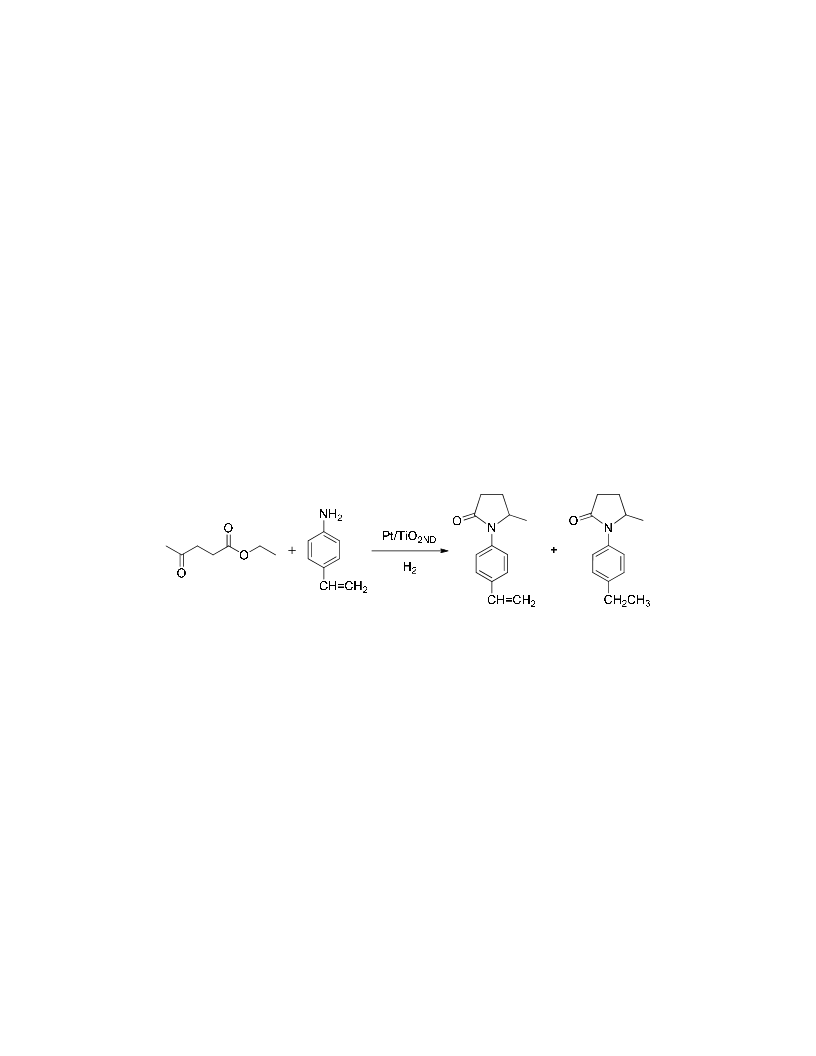
N-substituted-5-methyl-2-piyrrolidones have been obtained by reductive amination of ethyl levulinate with amines in the presence of H2 as reducing agent under solvent-free conditions. The process involves as a first step the formation of an imine intermediate followed by hydrogenation of the imine group and subsequent cyclization into pyrrolidone. Pt/TiO2 with Pt crystal faces decorated with TiOx is a very active and chemoselective catalyst, being possible to achieve high conversion and selectivity to the corresponding N-substituted-5-methyl-2-pyrrolidones even when other groups susceptible of hydrogenation such as vinyl, carbonyl, or cyano groups are present in the amine moiety. A kinetic study showed that the reaction-controlling step is the formation of the imine intermediate. The rate of formation is enhanced by the presence of protonic acid sites generated on the support by hydrogen dissociation on the metal, resulting in a true bifunctional catalyst for the reaction.
View More






Contact:+86-371-55981030
Address:Room 1571, Macalline Soho, No.1, Shangdu Road, Zhengzhou, Henan
Tianjin Anda North Industrial & Business Co.Ltd.
Contact:86-22-24999306
Address:No.11 Erwei Road,Dongli Development Area,Tianjin,China
website:http://www.tbbmed.com
Contact:86--21-50498136
Address:Room 6002, Building 7-1, No.160 Basheng Road,Pudong Area,Shanghai China
Shandong Zhongcheng Barium Salt Co., Ltd
Contact:+86-15725732638
Address:No.29 baoxi road, hi-tech zone, zibo, shandong
Contact:+86-579-85206992
Address:No 451 chouzhou north road ,room 1106 int'l business center , yiwu ,china
Doi:10.1039/c3ra43022g
(2013)Doi:10.1016/j.tetlet.2019.03.038
(2019)Doi:10.1055/s-2005-861877
(2005)Doi:10.1002/cssc.201402466
(2014)Doi:10.1039/d1cc00093d
(2021)Doi:10.1021/acs.inorgchem.5b00435
(2015)