Halpenny and Mascharak
salt, RuCl
3
·3H
2
O. Potassium pentachloronitrosylruthenate(II) was
Table 1. Summary of Crystal Data and Intensity Collection and
3
Structural Refinement Parameters for 1·2CHCl and 2
purchased from Alfa Aesar and used without further purification.
NO gas was purchased from Spectra Gases Inc. and was purified
by passing through a long KOH column prior to use. The nitrosyls
1
2
formula
fw
cryst color, habit
T (K)
cryst syst
space group
a (Å)
b (Å)
c (Å)
R (deg)
29 6 5 5 28 4 5 4
C H23Cl N O Ru C H22BF N O Ru
[
(Me
2
bpb)Ru(NO)(Cl)], [(Me
2
bQb)Ru(NO)(Cl)], and [(pyca)
2
Ru-
835.29
680.39
red, blade
153(2)
P1
(
NO)(Cl)] (3) were synthesized by following the published proce-
1
8,34
150(2)
dures.
All solvents were purified and/or dried by standard
jP1j
techniques and distilled.
triclinic
triclinic
Syntheses of Compounds. [(Me
2
bpb)Ru(NO)(OBz)] (1). A
in 5 mL of MeCN
was added to a stirred solution of 0.14 g (0.268 mmol) of
11.6052(6)
11.8863(6)
14.0099(7)
77.5810(10)
66.2400(10)
72.0440(10)
1673.43(15)
2
7.3221(17)
12.982(3)
15.497(4)
98.244(3)
99.337(3)
101.954(3)
1397.9(6)
2
solution of 0.057 g (0.274 mmol) of AgBF
4
1
8
[
2
(Me bpb)Ru(NO)(Cl)] in 20 mL of MeCN, and the reaction
ꢀ
(deg)
mixture was heated to reflux for 8 h. The precipitate of AgCl that
separated during this period was then removed by filtration through
a sintered-glass frit with a Celite pad on top. Next, a batch of 0.050
g (0.348 mmol) of sodium benzoate was added to the filtrate, and
the reaction mixture was heated to reflux for 4 h. Finally, the clear
red-brown solution was concentrated to 10 mL and filtered through
a sintered-glass frit with a Celite pad on top, as above. The filtrate
was then stored at -20 °C for 16 h, resulting in precipitation of a
fine red powder that was collected by filtration. Crystalline solid
γ (deg)
3
V (Å )
Z
d
calcd (g cm-3)
1.658
1.616
abs coeff, µ (mm-1)
0.994
0.632
a
2
GOF on F
final R indices [I > 2σ(I)] R1
1.028
1.017
b
0.0313
0.0428
c
wR2 0.0781
0.0903
b
R indices all data
R1
0.0369
0.0663
c
wR2 0.0818
0.0995
a
2
2
2
1/2
GOF ) [∑w(F
o
- F ) /(M - N)] (M ) number of reflections, N )
c
(
suitable for diffraction measurements) was obtained via slow
evaporation of the solution of this red solid in a CHCl /toluene
mixture (yield: 0.034 g, 21%). Anal. Calcd for C29 Cl
1·2CHCl ): C, 41.69; H, 2.78; N, 8.39. Found: C, 41.58; H, 2.71;
N, 8.12. Selected IR frequencies (KBr disk, cm ): 1833 (νNO, vs),
630 (vs), 1594 (vs), 1484 (m), 1355 (m), 1319 (s), 756 (w), 723
w), 682 (m). Electronic absorption spectrum in MeCN, λmax (in
b
c
2
number of parameters refined). R1 ) ∑|F
F
o c o
| - |F |/∑|F |. wR2 ) [∑w(F
o
3
2
2
2 1/2
-
c
) /∑w(F
o
) ]
.
H
23RuN
5
O
5
6
(
3
(
cluster of thick needles) were obtained after 5 days (0.035 g, 28%
yield). Anal. Calcd for C28 BF : C, 49.45; H, 3.27; N,
0.30. Found: C, 49.23; H, 3.19; N, 10.27. Selected IR frequencies
-1
H
22RuN
5
O
4
4
1
(
1
(
1
-1
KBr disk, cm ): 1872 (νNO, vs), 1680 (vs), 1650 (vs), 1386 (m),
325 (s), 1174 (m), 1082 (s), 875 (w), 764 (m). Electronic
-1
-1
1
nm) (ꢀ in M cm ): 300 (13 000), 390 (6700). H NMR (500
MHz. CDCl , δ from TMS): 2.35 (s, 6H), 7.09 (t, 2H), 7.23 (t,
H), 7.49 (d, 2H), 7.68 (t, 2H), 8.14 (t, 2H), 8.29 (d, 2H), 8.54 (s,
3
-1
-1
absorption spectrum in MeCN, λmax (in nm) (ꢀ in M cm ): 240
(
1
2
67 000), 320 (16 000), 600 (br, 2000). 1H NMR (500 MHz.
H), 9.13 (d, 2H).
CD CN, δ from TMS): 2.25 (s, 3H), 2.35 (s, 3H), 6.65 (d, 1H),
3
2
The acetate analogue of 1, namely, [(Me bpb)Ru(NO)(OAc)],
7
.04 (s, 1), 7.10 (t, 1H), 7.58 (t, 1H), 8.14 (m, 3H), 8.44 (m, 3H),
was also synthesized in the present study by following the same
procedure (except for the use of sodium acetate in place of
benzoate). The reddish-orange microcrystalline product was isolated
in 70% yield. Anal. Calcd for C22
N, 13.10. Found: C, 49.33; H, 3.49; N, 13.37. Selected IR
8
.53 (s, 1H), 8.65 (d, 1H), 8.88 (d, 1H), 8.94 (d, 1H).
Physical Measurements. Absorption spectra were recorded on
a Cary 50 Varian spectrophotometer. IR spectra were obtained with
a Perkin-Elmer 1600 FTIR spectrophotometer. Electron paramag-
netic resonance (EPR) spectra of the photoproducts were monitored
on a Bruker ELEXSYS 500 spectrometer. A Varian 500 MHz
5 5
H19RuN O : C, 49.43; H, 3.59;
-1
frequencies (KBr disk, cm ): 1841 (νNO, vs), 1639 (vs), 1598 (vs),
484 (m), 1358 (s), 1292 (s), 1097 (vw), 760 (w), 684 (m). This
nitrosyl is sparingly soluble in most solvents [MeCN, N,N-
1
1
spectrometer was employed to record the H NMR spectra at 298
K.
dimethylformamide (DMF), and CHCl
any solution study.
3
] and has not been used for
Photolysis Experiments. The apparent rates of NO release (kNO
)
from 1 and 2 were determined by electronic absorption spectroscopy
[
(Me
mmol) of AgBF
of 0.10 g (0.164 mmol) of [Ru(Me
2
Qb)Ru(NO)(qca)]BF
4
(2). A solution of 0.041 g (0.211
18
using a Varian Cary 50 spectrophotometer. Solutions of the
4
in 5 mL of DMF was added to a stirred solution
complexes (100-500 µM) in MeCN were used. The cuvette was
held at a fixed distance of 1 cm from the light source (5 mW UV
lamp). Absorption spectra were taken after certain time intervals
1
8
2
bQb)(NO)(Cl)] in 25 mL of
DMF, and the reaction mixture was heated to 60 °C for 16 h. The
precipitate of AgCl that separated during this period was then
removed by filtration through a sintered-glass frit with a Celite pad
on top. DMF was removed by short-path distillation, and the solid
was triturated four times with 10 mL of MeCN. The residue was
finally dissolved in 30 mL of MeCN, and diethyl ether was allowed
to diffuse into it slowly at room temperature. X-ray-quality crystals
(
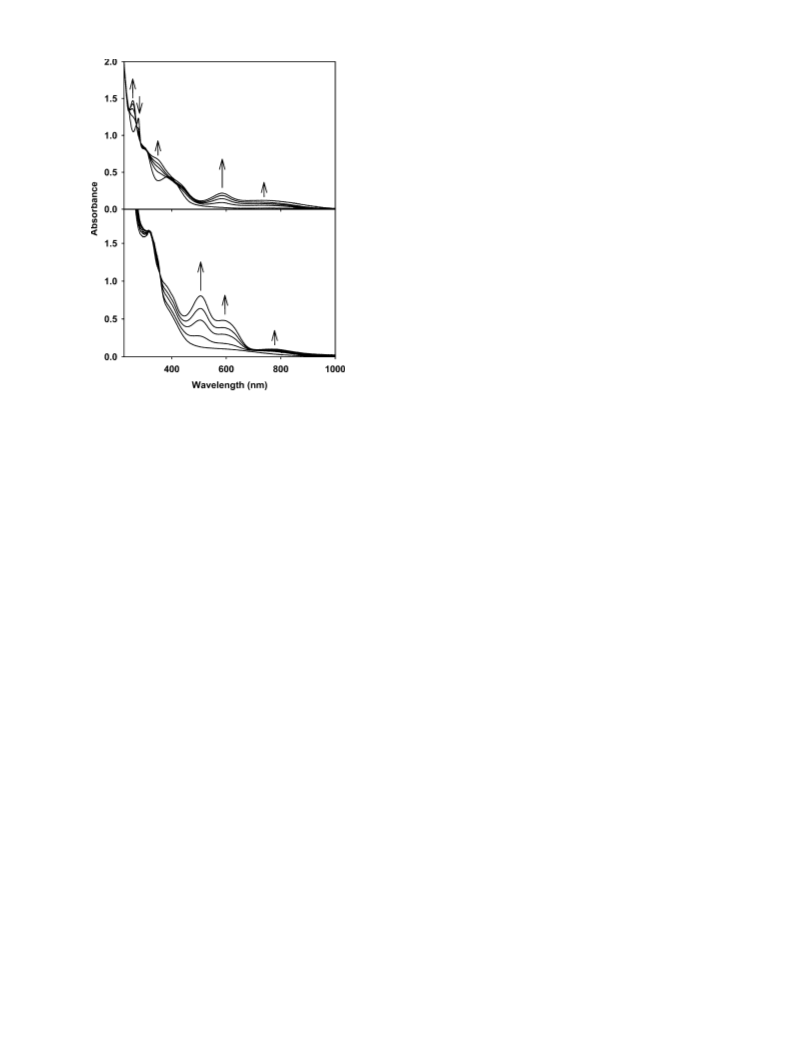
usually 15-20 s), and the absorbance values at specific wave-
lengths (585 nm for 1 and 590 nm for 2) were noted. The apparent
rates of NO photorelease (kNO) were calculated from plots of the
concentrations of the photoproducts (absorbing at the 550-650 nm
range) versus the total time of exposure using the equation c ) c
a exp(-kNOt). The quantum yields at 300 nm were determined
0
+
2
2
by standard ferrioxalate actinometry. An Oriel Apex illuminator
(
(
30) Greene, S. N.; Richards, N. G. J. Inorg. Chem. 2004, 43, 7030–7041.
31) Sizova, O. V.; Ivanova, N. V.; Lyubimova, O. O. Russ. J. Gen. Chem.
1
(
8
150 W Xe lamp) with an Oriel / m Cornerstone monochromator
2
004, 74, 155–163.
was used as the light source. The NO amperogram was recorded
with an amiNO-2000 electrode (part of an inNO Nitric Oxide
Measuring System, Innovative Instruments, Inc.).
X-ray Crystallography. Diffraction data were collected on a
Bruker Apex II system at 150 K. Mo KR (0.710 73 Å) radiation
was used, and the data were corrected for absorption (Table 1).
The structure was solved by direct methods (standard SHELXS-97
package).
(
(
(
(
32) Works, C. F.; Jocher, C. J.; Bart, G. D.; Bu, X.; Ford, P. C. Inorg.
Chem. 2002, 41, 3728–3739.
33) Bordini, J.; Hughes, D. L.; Da Motta Neto, J. D.; da Cunha, J. C.
Inorg. Chem. 2002, 41, 5410–5416.
34) Bottomley, F.; Hahn, E.; Pickardt, J.; Schumann, H.; Mukaida, M.;
Kakihana, H. J. Chem. Soc., Dalton Trans. 1985, 2427–2431.
35) Toma, H. E.; Alexiou, A. D. P.; Formiga, A. L. B.; Nakamura, M.;
Dovidauskas, S.; Eberlin, M. N.; Tomazela, D. M. Inorg. Chim. Acta
2
005, 358, 2891–2899.
1492 Inorganic Chemistry, Vol. 48, No. 4, 2009

 Halpenny, Genevieve M.
Halpenny, Genevieve M.
 Mascharak, Pradip K.
Mascharak, Pradip K.