4762 J. Agric. Food Chem., Vol. 50, No. 17, 2002
Freisleben et al.
separated by filtration. The acetic anydride was evaporated, and the
residual 15 (0.847 g, 3.62 mmol, 69%) was resuspended in water and
lyophilized.
1H NMR: ([2H6]DMSO) δ ) 2.21 (3H, s, COCH3), 5.87 (1H, t,
OH), 9.04 (1H, s, NH), 9.27 (1H, s, CH), 10.08 (1H, s, CHO).
RESULTS AND DISCUSSION
Consideration of the Most Suitable Labelings and Selec-
tion of Vitamers to be Synthesized. A literature survey
indicated that different forms of folic acid labeled by stable
isotopes have been prepared in the past. In a prior attempt Plante
et al. (16) reported the synthesis of [carbonyl-13C1]folic acid
before Gregory and Toth prepared [3′,5′-2H2]folic acid (17) as
well as pteroyl[2H4]glutamic acid (18), and Dueker et al. (19)
generated [2′,3′,5′,6′-2H4]folic acid. More recently, Rogers and
co-workers (20) synthesized [13C5]folic acid by coupling
[13C5]glutamic acid with trifluoroacetyl pteroic acid.
Positive ESI-MS: m/z (%) ) 252 (100), 485 (62), 234 (55), 467
(15).
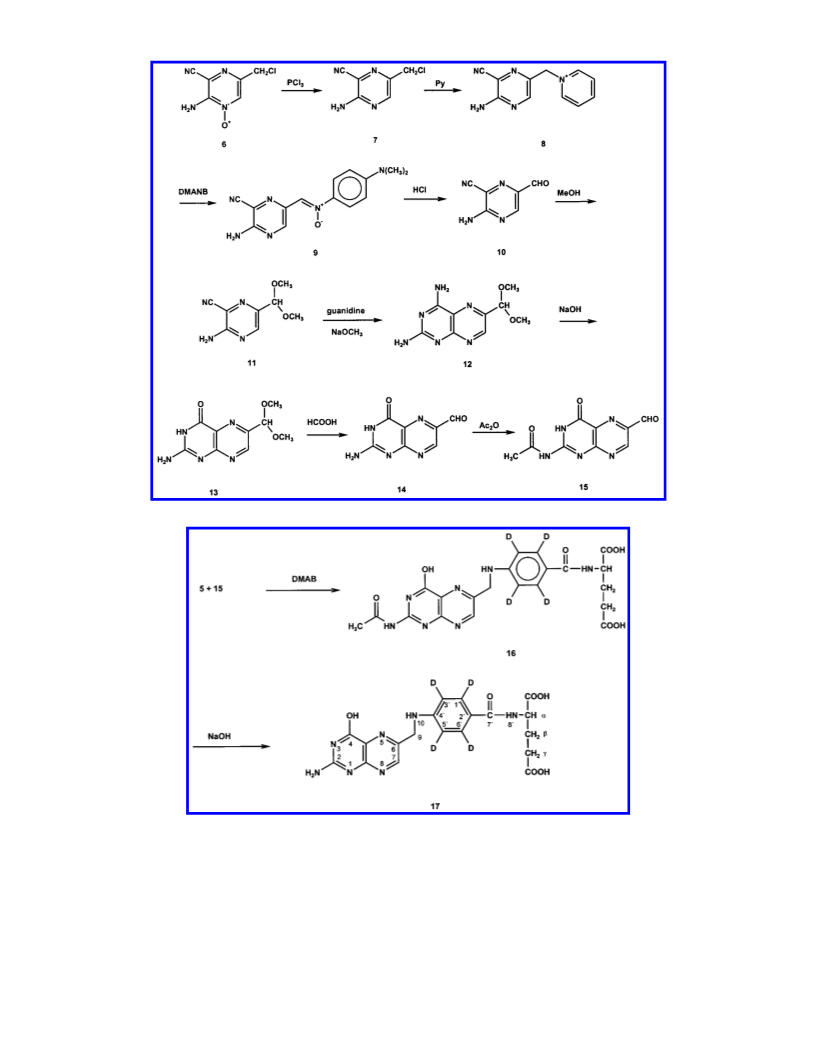
[2H4]Folic Acid. N2-Acetyl[2H4]folic Acid (16). To a flask containing
glacial acetic acid (30 mL) N2-acetyl-6-formylpterin (559 mg, 2.4 mmol)
and [2H4]4-aminobenzoylglutamate (700 mg, 2.6 mmol) were added.
After maintaining the solution for 10 min, dimethylaminoborane (153.4
mg, 2.6 mmol) in glacial acetic acid (10 mL) was added, and the
solution was heated for 20 min at 60 °C in an oil bath. The resulting
solution was evaporated and the residue was redissolved in aqueous
ammonium formate (0.15 mol/L) at pH 7. The solution was purified
by anion exchange chromatography as described above. The fractions
absorbing at 280 nm were verified to contain 16 using HPLC-MS,
then pooled and lyophilized to yield 415 mg (0.85 mmol, 35%) of 16.
[2H4]Folic Acid (17). Compound 16 (415 mg, 0.85 mmol) was
dissolved in aqueous sodium hydroxide (50 mL, 0.1 mol/L) and stirred
for 6 h at room temperature. The reaction was quenched by adjusting
the pH to 3.5 by addition of hydrochloric acid (1 mol/L). [2H4]Folic
acid (303 mg, 0.68 mmol, 80%) precipitated quantitatively after standing
overnight at 4 °C and was lyophilized after the solution was centrifuged
and the supernatant was discarded.
1H NMR: ([2H6]DMSO): 1.91 (2 H, m, â-C-2H); 2.32 (2 H, m,
γ-C-2H); 4.30 (1 H, t, R-C-1H); 4.47 (2 H, d, C9-2 H); 6.89 (1 H, t,
C4-OH); 7.99 (1 H, d, N8′-H); 8.64 (1 H, s, C7-H).
Positive ESI-MS: m/z (%) ) 446 (100), 447 (23), 468 (17), 445
(7).
Deuterated Vitamers of Folic Acid. [2H4]Tetrahydrofolic Acid. A
reaction vessel with a three-way gas inlet and a flowmeter counter was
filled with glacial acetic acid (25 mL) and platinum oxide (100 mg).
The vessel was flushed for 1 min with nitrogen. Upon addition of
[2H4]folic acid (250 mg, 0.562 mmol), the mixture was flushed
repeatedly with hydrogen gas and finally stirred under a slight positive
pressure of hydrogen for 4 h. At the end of the reaction, the catalyst
was filtered off, and the solution was lyophilized to give [2H4]tetra-
hydrofolic acid diacetate (197 mg, 0.348 mmol, 62%).
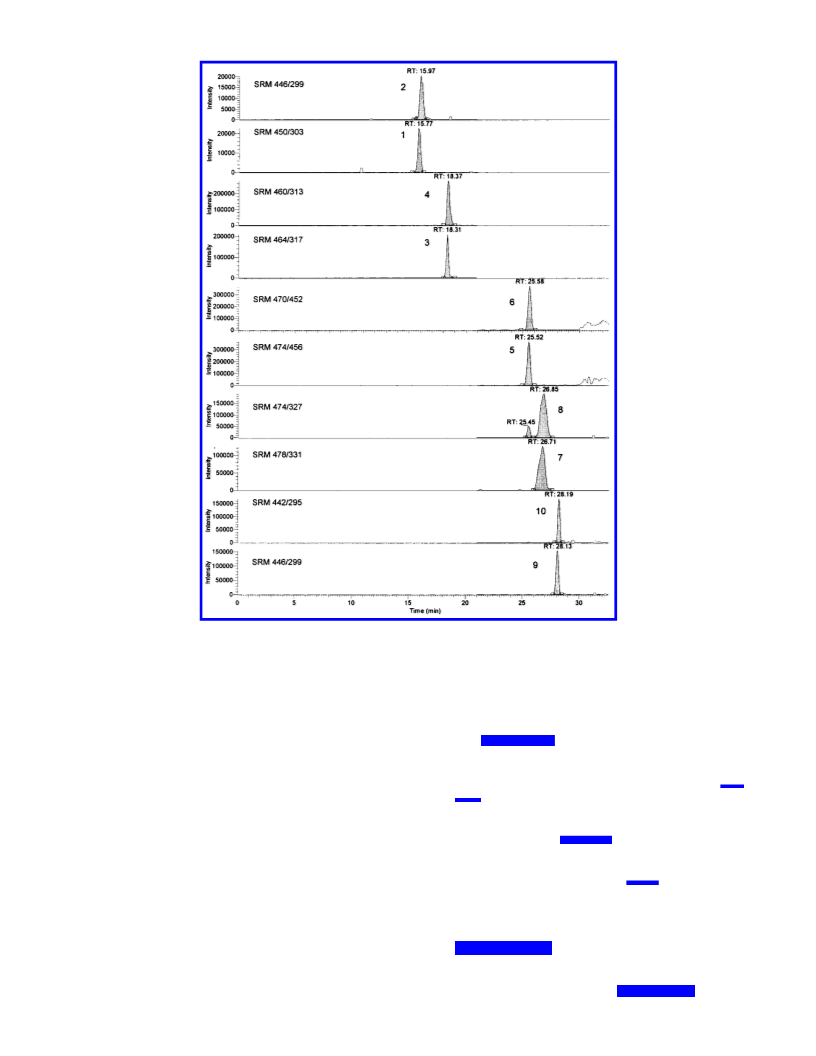
The anticipated determination by LC-MS, however, renders
only a few labelings promising. As quantification would be
based on measuring the quasimolecular ions (M + 1)+ of folic
acid vitamers which all contain at least 19 carbon, 7 nitrogen,
and 6 oxygen atoms, interferences from naturally occurring
isotopes in the analytes have to be taken into consideration.
Due to natural abundance of the isotopes C-13, N-15, and O-18,
an intensity of nearly 4% can be calculated for the quasi-
molecular ions of doubly labeled folates. Therefore, using a
labeling of only two mass units would result in mass spectro-
metric overlap between the unlabeled analytes and the labeled
internal standards. For this reason we considered a label
producing at least three mass increments to overcome this
hindrance. Regarding the syntheses mentioned above, only
[2′,3′,5′,6′-2H4]folic acid, pteroyl[2H4]glutamic acid or pteroyl-
[13C5]glutamic acid would be suitable. The latter preparations,
however, both require the use of labeled glutamic acid, which
is too expensive to give sufficient folic acid to be used as starting
component for the syntheses of further vitamers. Therefore, we
decided to synthesize [2′,3′,5′,6′-2H4]folic acid by introducing
a 4-fold deuterium label in the benzene ring of p-aminobenzoic
acid, which enabled us to obtain higher yields of labeled folic
acid.
Recent analyses of folates by high-performance liquid chro-
matography (HPLC) revealed tetrahydrofolate, 5-methyltetra-
hydrofolate, 5-formyltetrahydrofolate, 10-formylfolate, and folic
acid to predominate in different foods (7, 21) and 5-methyl-
tetrahydrofolate to be the most important vitamer in blood (22).
Therefore we chose these five vitamers (Figure 1) to be
synthesized starting from [2H4]folic acid.
Positive ESI-MS: m/z (%) ) 450 (100), 451 (22), 449 (4), 452 (2).
[2H4]5-Formyltetrahydrofolic Acid. [2H4]Tetrahydrofolic acid di-
acetate (80 mg, 0.142 mmol) was dissolved in formic acid (960 µL,
98-100%) and aqueous dipotassium hydrogenphosphate (10 mL, 50
mM containing 0.5% mercaptoethanol), and the solution was adjusted
to pH 3.5 with aqueous ammonium hydroxide (0.1 mol/L). Then,
1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (32 mg, 0.168 mmol)
was added, and the mixture was stirred for 15 min under an argon
atmosphere. Lyophilization gave the crude product, which was purified
by anion exchange chromatography yielding [2H4]5-formyltetrahydro-
folate (14 mg, 0.029 mmol, 18%) with a purity exceeding 98% (HPLC).
Synthesis of Labeled Folic Acid. The intended synthetic
approach to labeled folic acid required the coupling of labeled
p-aminobenzoylglutamic acid and 6-formylpterin.
The first component can be generated either by labeling
p-aminobenzoic acid in a catalytic protium-deuterium exchange
followed by coupling to glutamic acid, or by reacting deuterated
toluene to nitrobenzoylglutamate and subsequent reduction of
the nitro group. In a first series of experiments the latter reaction
sequence, according to Dueker et al. (19), was pursued as it
proposed higher yields. The synthesis involved nitration of
[2H8]toluene, followed by oxidation of the methyl group and
formation of the chloride. The subsequent coupling with
glutamyl diethylester and reduction to p-aminobenzoylglutamic
acid gave a reaction mixture in which purification of the target
compound from a host of byproducts could not be accomplished.
Therefore, we chose the former alternative starting with deu-
teration of p-aminobenzoic acid as depicted in Figure 2.
Comparing the mass spectra of unlabeled and labeled p-
aminobenzoic acid (Figure 3) the incorporation of the four
deuterium atoms can be clearly seen from the mass shift of the
quasimolecular ion from m/z 138 to m/z 142. The isotopic peak
areas of p-aminobenzoic acid indicate a 95% incorporation of
Positive ESI-MS: m/z (%) ) 478 (100), 479 (20), 477 (4), 480 (3).
[2H4]5-Methyltetrahydrofolic Acid. [2H4]Tetrahydrofolic acid (80 mg,
0.142 mmol) was dissolved in an aqueous solution of TRIS at pH 8
(25 mL) and stirred for 15 min at room temperature upon the addition
of aqueous formaldehyde (150 µL, 37%). Sodium borohydride (800
mg, 21 mmol) was added and the solution was heated at 50 °C for 1
h. The excess of reducing agent was destroyed by adding acetic acid
(5 mol/L) dropwise. Purification by anion exchange chromatography
and lyophilization of the pooled eluates provided the title compound
(22 mg, 0.048 mmol, 34%).
Positive ESI-MS: m/z (%) ) 464 (100), 465 (19), 317 (7), 466 (3).
[2H4]10-Formylfolic Acid. [2H4]Folic acid (30 mg, 0.067 mmol) was
dissolved in 50 mL of concentrated formic acid and heated for 3 h at
60 °C. Formic acid was evaporated, and the residue was redissolved
in 1 mL of aqueous formic acid (50%). Purification by high-
performance liquid chromatography provided [2H4]10-formylfolic acid
with a purity exceeding 97% (5.1 mg, 0.0107 mmol, 16%).
Positive ESI-MS: m/z (%) ) 474 (100), 475 (27), 473 (10), 476
(9).

 Freisleben, Achim
Freisleben, Achim
 Schieberle, Peter
Schieberle, Peter
 Rychlik, Michael
Rychlik, Michael