7278 J. Am. Chem. Soc., Vol. 118, No. 31, 1996
Rajca et al.
102.478(7)°, Z ) 2, and Fcalc ) 1.159 Mg m- . The crystal of 9B was
3
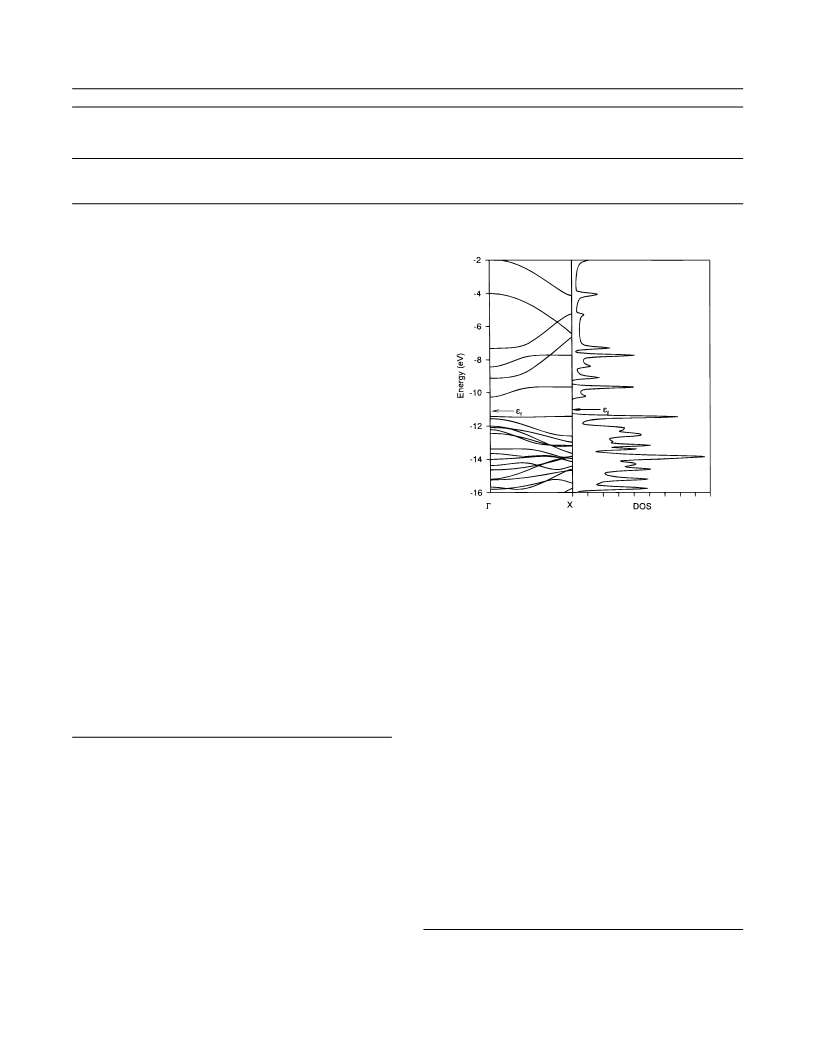
a large DOS at the Fermi level in solids (superconductor, metal,
ferromagnet, etc.) are difficult to predict. Although for a class
of oligomers with topological multi near degeneracies, such as
organic high-spin molecules, strong electron localization and
ferromagnetic spin coupling are predictable from the molecular
3
a yellow-orange plate, 0.38 × 0.15 × 0.1 mm ; its space group was
determined to be P2 /c with a ) 14.423(1) Å, b ) 6.1657(6) Å, c )
1
-
3
1
7.994(1) Å, â ) 104.125(5)°, Z ) 2, and Fcalc ) 1.123 Mg m . The
3
crystal of 9C was an orange plate, 0.3 × 0.3 × 0.06 mm ; its space
group was determined to be P2 /n with a ) 14.856(4) Å, b )
1
1
3b,23
topology;
i.e., in this case, straightforward models can
1
4
9
2.727(5) Å, c ) 17.198(5) Å, â ) 103.379(7)°, and presumably Z )
sufficiently account for the effects of electron-electron repul-
. The crystals were selected from the following two samples: (1)
4
6
sion.
A, obtained by slow evaporation of organic solvents (methylene
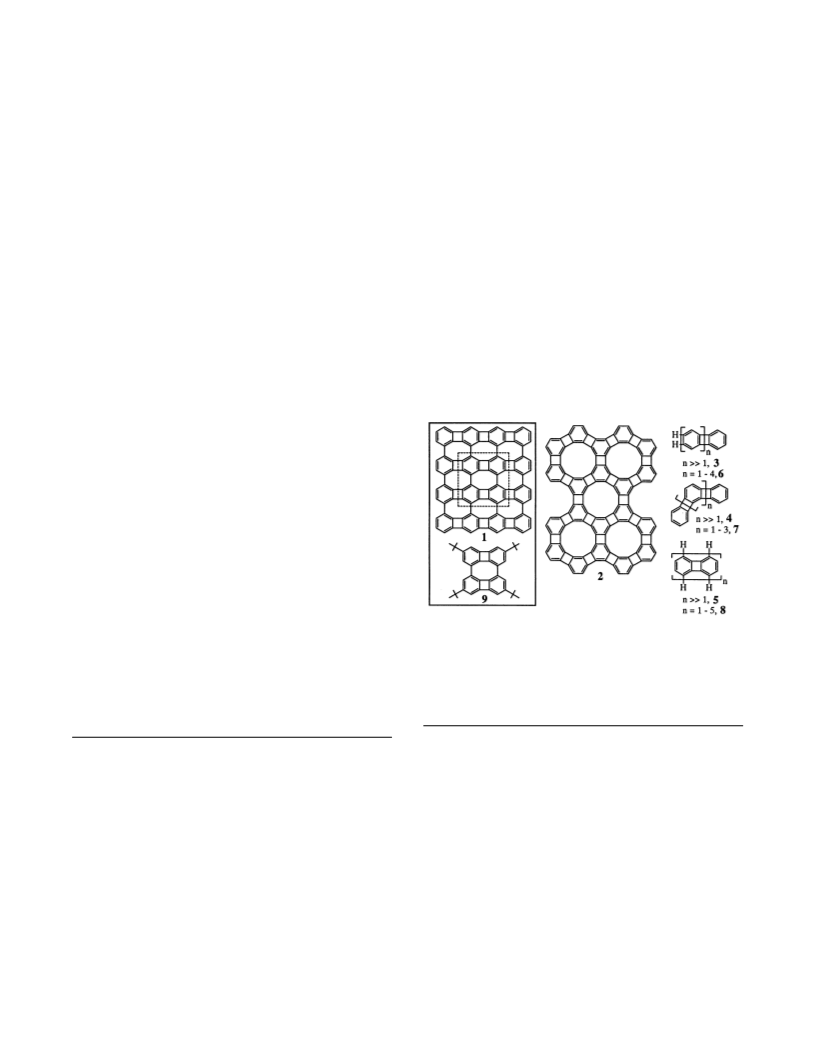
Dissections of net 1 into polymers 3 and 5, and then into a
molecular fragment, biphenylene dimer 8 (n ) 2), can be
compared to those of the graphite net, poly(peri-naphthalene),
and, then, perylene (naphthalene dimer).47 At the molecular
level, the biphenylene dimer and naphthalene dimer have quite
chloride and ether), (2) 9B and 9C, obtained by heating under Ar at
380 °C.
The initial structure models were determined by direct methods,
developed by Fourier methods, and refined by full-matrix least-squares
techniques. Hydrogen atom positions were calculated from known
geometry (dC-H ) 0.96 Å); H atoms were refined using a riding-atom
model and two isotropic U parameters (one for the methyl hydrogens,
the second for the aromatic hydrogens). The carbon atoms were refined
with anisotropic atomic displacement parameters.
different electronic structures; the differences for the polymers
and nets remain to be determined.48
Experimental Section
-
1
2
2
For 9A, final cycles were weighted {w ) σ (F) + 0.0006F }.
Refinement of 183 variables converged to R ) 0.0529, wR ) 0.0563,
General Procedures. Ether and tetrahydrofuran (THF) for use on
a vacuum line were distilled from sodium/benzophenone in a nitrogen
atmosphere. Major chemicals were obtained from Aldrich. 1-Iodo-
-1
2
and S ) 1.21. For 9B, final cycles were weighted {w ) σ (F) +
0.0007F }. Refinement of 183 variables converged to R ) 0.0539,
2
2,6-dibromobenzene and 1-iodo-2,6-dibromo-4-tert-butylbenzene were
wR ) 0.0611, and S ) 1.24. For 9C, only the unit cell and space
group are known at this time.
prepared using standard procedures of functional group transformations;
starting materials were sulfanilamide (or 2,6-dibromoaniline) and tert-
Data collection and analysis are described in detail in deposited
material. Fractional atomic coordinates, atomic displacement param-
eters, and parameters describing the bonding geometry and conformation
are available as supporting information.
4
9-51
butylbenzene, respectively.
described elsewhere.
The vacuum lines and glovebox are
2
3
1
NMR spectra were obtained using Omega spectrometers ( H, 500
and 300 MHz) in either CDCl
3
or THF-d
8
; the chemical shift references
); THF-d , 3.58 ppm (THF-
, 67.45 ppm.
ESR spectra were acquired with a Bruker 200D X-band instrument
2,2′,6,6′-Tetrabromo-4,4′-di-tert-butylbiphenyl. n-BuLi (5.74 mL
of a 2.5 M solution in hexane, 14.4 mmol) was added to a solution of
1-iodo-2,6-dibromo-4-tert-butylbenzene (5.00 g, 12.0 mmol) in ether
(180 mL) at -78 °C. After the solution was stirred for 2 h at -78 °C,
1
were as follows: ( H) TMS, 0.0 ppm (CDCl
3
7
1
3
8 3 8
d ); ( C) CDCl , 77.0 ppm; THF-d
at ambient temperature, using 4-mm CFQ tubes equipped with high-
vacuum PTFE stopcocks (Kontes).
CuBr (16.2 g, 72.0 mmol) was added, and then the reaction mixture
2
was allowed to attain ambient temperature over a 12-h period. Cold
water was added to the reaction mixture, and usual aqueous workup
gave 4.47 g of a yellow oil. Treatment of the crude product with cold
hexane (30 mL) afforded 1.04 g (30%) of a white solid. Mp: 269-
271 °C. Anal. Calcd for C H Br : C, 41.27; H, 3.81. Found: C,
UV-vis absorption spectra were recorded at ambient temperature
in a 2-mm-path-length quartz cell using a Perkin-Elmer Lambda 6
spectrophotometer. The quartz cell was equipped with high-vacuum
PTFE stopcocks (Kontes). The spectrophotometer sample chamber was
accessible from a Vacuum Atmospheres glovebox.
Cyclic voltammograms were recorded using a PARC 270 electro-
chemistry system at a 100-mm-diameter. Pt-disk working electrode
in THF/TBAP (TBAP ) tetrabutylammonium perchlorate) at ambient
temperature; ferrocene (0.510 V vs SCE) was used as a reference.
HPLC was carried out using a Waters 600 instrument equipped with
20
22
4
41.31; H, 3.99. LR/HR EIMS: m/z (ion type, % RA for m/z ) 50-
+
600, deviation for the formula) 581.840 40 ((M + 4) , 45%, -2.2 ppm
deviation for 1 C201H22 Br2 Br ), 579.842 47 ((M + 2) , 31%, -1.9
2
79
81
+
2
deviation for C201H22 Br381Br ), 577.844 07 (M , 8%, -2.5 ppm
12
79
+
1
deviation for 12C 01H Br ), 566.822 16 ((M + 4 - CH ) , 100%,
79
+
2
22
4
3
12
1
79
81
+
3
7.0 ppm deviation for
C
19
H
19 Br
2
Br
2
79
), 562.824 78 ((M - CH
) ,
): δ
): δ 154.6, 139.2,
17%, 4.9 ppm deviation for 2C191H19 Br
1
). H NMR (CDCl
1
8
× 10 Radial Pak Cartridges (NovaPak silica and NovaPak C18 with
4
3
7.63 (s, 4 H), 1.35 (s, 18 H). 13C{ H} NMR (CDCl
1
4
-µm particle sizes). For analysis of dimer 9, the eluents were hexane
3
(5.5 min at 0.8 mL/min) and acetonitrile/THF/water (65/30/5; 8.2 min
129.0, 124.1, 35.0, 31.1.
at 2.0 mL/min).
2,2′,6,6′-Tetrabromobiphenyl. The crude product was obtained
using the procedure analogous to that for the tert-butyl-substituted
derivative; either CuBr or CuCl was used as the oxidant. From six
Elemental analyses were carried out by Dr. G. M. Dabkowski,
Director-Microlytics, P.O. Box 199, S. Deerfield, MA 01373, and
M-H-W Laboratories, P.O. Box 15149, Phoenix, AZ 85060.
X-ray Crystallography. X-ray diffraction data were collected with
a Siemens P4 automatic diffractometer using graphite-monochromatized
Mo KR radiation (λ ) 0.710 73 Å). The crystal of 9A was an orange
2
2
reactions, using CuCl as the oxidant, a white precipitate (9.03 g, 33%)
2
from hexane was obtained. Mp: 214-216 °C (lit.52 mp: 215 °C). H
1
NMR (CDCl ): δ 7.66 (d, J ) 8 Hz, 4 H), 7.16 (t, J ) 8 Hz, 2 H).
3
GC-MS (EI): 470.
3
prism, 0.6 × 0.2 × 0.1 mm ; its space group was determined to be
Column chromatography (flash silica, hexane) of the mother liquid
allows for isolation of additional amounts of 2,2′,6,6′-tetrabromobi-
phenyl (overall yield 40+%) and the major side products.
1
P2 /n with a ) 15.420(2) Å, b ) 5.967(1) Å, c ) 16.735(2) Å, â )
(46) Borden, W. T.; Davidson, E. R. J. Am. Chem. Soc. 1977, 99, 4587.
2
-Chloro-1,3-dibromobenzene (CuCl
MHz, CDCl ): δ 7.58 (d, J ) 8 Hz, 2 H), 6.98 (t, J ) 8 Hz, 1 H).
GC-MS (EI): m/z 271.
as Oxidant). 1H NMR (500
2
Ovchinnikov, A. A. Theor. Chim. Acta 1978, 47, 297. Klein, D. J.; Nelin,
C. J.; Alexander, S.; Matsen, F. A. J. Chem. Phys. 1982, 77, 3101. Borden,
W. T.; Iwamura, H.; Berson, J. A. Acc. Chem. Res. 1994, 27, 109.
3
(47) Poly(peri-naphthalene) and oligomers: Viruela-Martin, R.; Viruela-
as Oxidant, 25%). 1H NMR
): δ 7.58 (d, J ) 8 Hz, 2 H), 7.03 (t, J ) 8 Hz, 1 H). GC-MS
1
,2,3-Tribromobenzene (CuBr
2
Martin, P. M.; Orti, E. J. Chem. Phys. 1992, 97, 8470. Baumgarten, M.;
Koch, K. -H.; M u¨ llen, K. J. Am. Chem. Soc. 1994, 116, 7341.
(
(
CDCl
3
EI): m/z 314, 316.
(48) Both net 1 and graphite are metals (Eg ) 0) at the EHT level; for
calculations on graphite, see: Whangbo, M.-H.; Hoffmann, R.; Woodward,
R. B. Proc. R. Soc. London, A 1978, 366, 23. However, the electronic
structure of net 1, composed of polymer 3, may not be adequately described
at this level of theory.
1,8-Dibromo-3,6-di-tert-butylbiphenylene. n-BuLi (3.44 mL of a
2.5 M solution in hexane, 8.59 mmol) was added to a solution of
,2′,6,6′-tetrabromo-4,4′-di-tert-butylbiphenyl (2.00 g, 3.44 mmol) in
THF (200 mL) at -78 °C. After the mixture was stirred for 2 h at
78 °C, CuCN (0.77 g, 8.6 mmol) was added and the mixture was
2
(49) Seikel, M. K. Organic Syntheses; Wiley: New York, 1955; Collect.
-
Vol. III, p 262.
allowed to warm to room temperature. After the CuCN had completely
(
50) Shoesmith, J. B.; Mackie, A. J. Chem. Soc. 1928, 2334.
(51) Drake, N. L.; Eaker, C. M.; Garman, J. A.; Hamlin, K. E., Jr.; Hayes,
dissolved (red color solution), the reaction was recooled to -78 °C
R. A.; Haywood, S. T.; Peck, R. M.; Preston, R. K.; Sterling, J., Jr.; van
Hook, J. O.; Walton, E. J. Am. Chem. Soc. 1946, 68, 1602.
(52) Meyer, R; Meyer, W. Chem. Ber. 1920, 53, 2034-2052.

 Rajca, Andrzej
Rajca, Andrzej
 Safronov, Andrej
Safronov, Andrej
 Rajca, Suchada
Rajca, Suchada
 Ross II, Charles R.
Ross II, Charles R.
 Stezowski, John J.
Stezowski, John J.