
Monatshefte fur Chemie p. 285 - 299 (2006)
Update date:2022-08-25
Topics:
 Ghoneim, Mohammed M.
Ghoneim, Mohammed M.
 El-Hallag, Ibrahim S.
El-Hallag, Ibrahim S.
 El-Baradie, Kamal Y.
El-Baradie, Kamal Y.
 El-Desoky, Hanaa S.
El-Desoky, Hanaa S.
 El-Attar, Mona A.
El-Attar, Mona A.
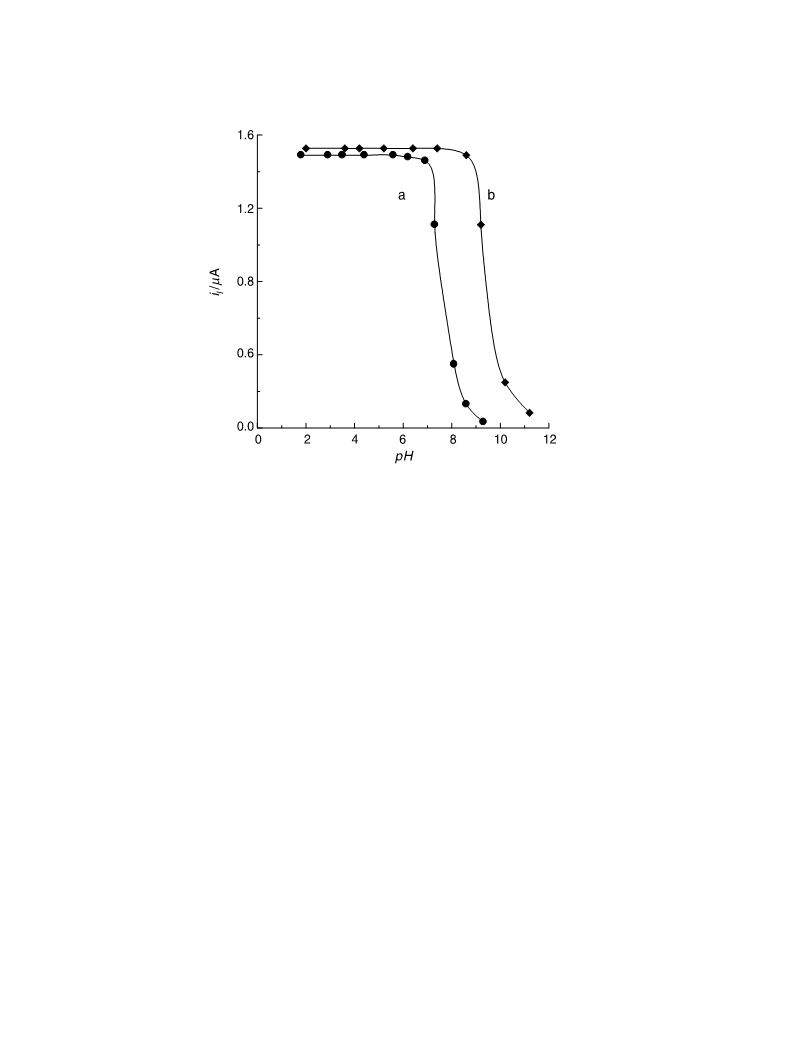
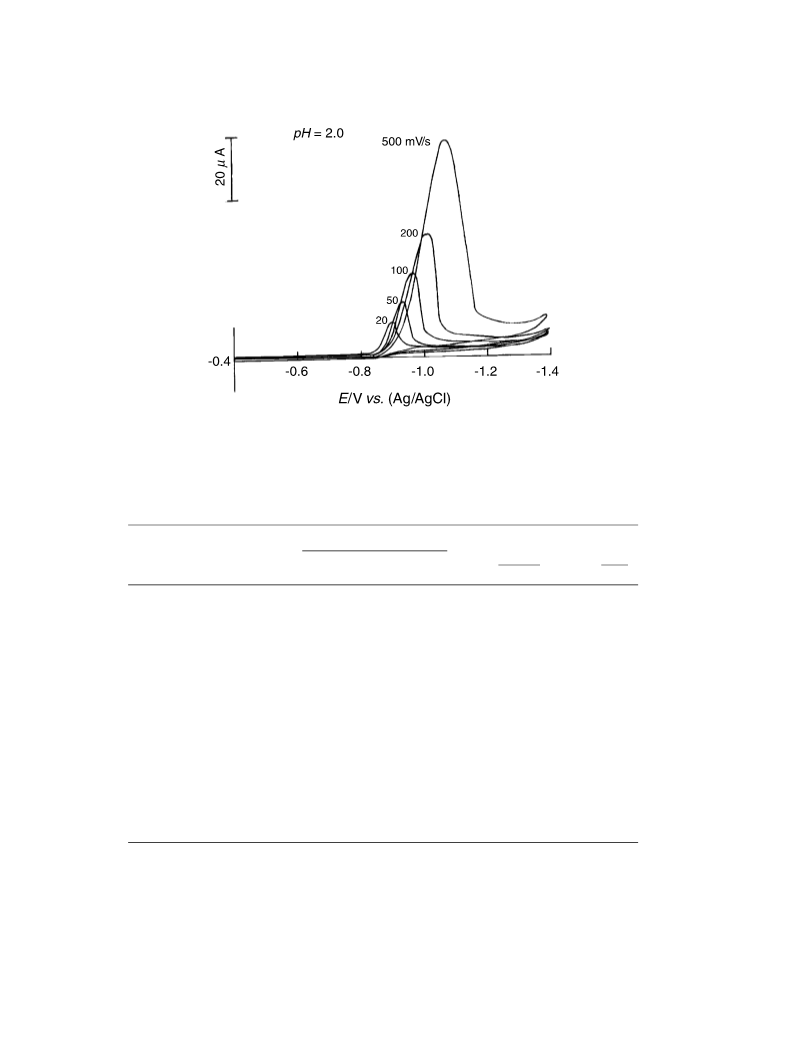
The electrochemical behavior of some hydrazones derived from 6-chloro-2-hydrazinopyridine in the Britton-Robinson universal buffer of pH 2-11 containing 35% ethanol was investigated at the mercury electrode using dc-polarography, controlled-potential coulometry, and cyclic voltammetry techniques. The examined hydrazones were reduced in solutions of pH < 9 in a single 4-electron diffusion-controlled irreversible step corresponding to both the saturation of -N=C< double bond and cleavage of the -HN-NH- single bond of the hydrazone molecule via the consumption of two electrons for each center. Whereas the starting compound, 6-chloro-2-hydrazinopyridine, was reduced in a single 2-electron diffusion-controlled irreversible step corresponding to cleavage of its -NH-NH2 single bond. The mechanistic pathway of the electrode reaction of the studied compounds was elucidated and discussed. The pK a values of the examined hydrazones and the stoichiometry of their complexes in solution with some transition metal ions were determined spectrophotometrically. The dissociation constants and the thermodynamic parameters of the investigated hydrazones, and the stability constants of their metal complexes in solution were determined potentiometrically. Springer-Verlag 2006.
View More









Anhui Gusheng Import&Export CO.,LTD
Contact:86-551-63662296
Address:Jinzhai Road NO.162 ,hefei, china
Weifang Arylchem Chemical Co., LTD
Contact:86-536-5217866
Address:Development Zone, Shouguang, Shandong Province
Contact:86-371-63655023
Address:No.85,jinshui road,zhengzhou,China
Contact:(1) 206-3550089
Address:5115 NE 8TH PL, Renton, WA 98059 USA
Tianjin Crest Pharmaceutical R&D Co., Ltd. (Tianjin Yao Technology Development Co., Ltd.)
Contact:+86-22-66211386
Address:Building B5-405, No, 80 4th Avenue, TEDA, Tianjin, China P.R. 300457
Doi:10.1016/0039-128X(80)90102-6
(1980)Doi:10.1016/j.molstruc.2012.06.016
(2012)Doi:10.1007/s10600-013-0532-1
(2013)Doi:10.1039/c8nj04385j
(2019)Doi:10.1016/j.ica.2006.05.011
(2006)Doi:10.1002/jccs.201700359
(2018)