
Journal of Organic Chemistry p. 9810 - 9819 (2016)
Update date:2022-08-25
Topics:
 Sharif, Salaheddin A.I.
Sharif, Salaheddin A.I.
 Calder, Ewen D. D.
Calder, Ewen D. D.
 Harkiss, Alexander H.
Harkiss, Alexander H.
 Maduro, Marie
Maduro, Marie
 Sutherland, Andrew
Sutherland, Andrew
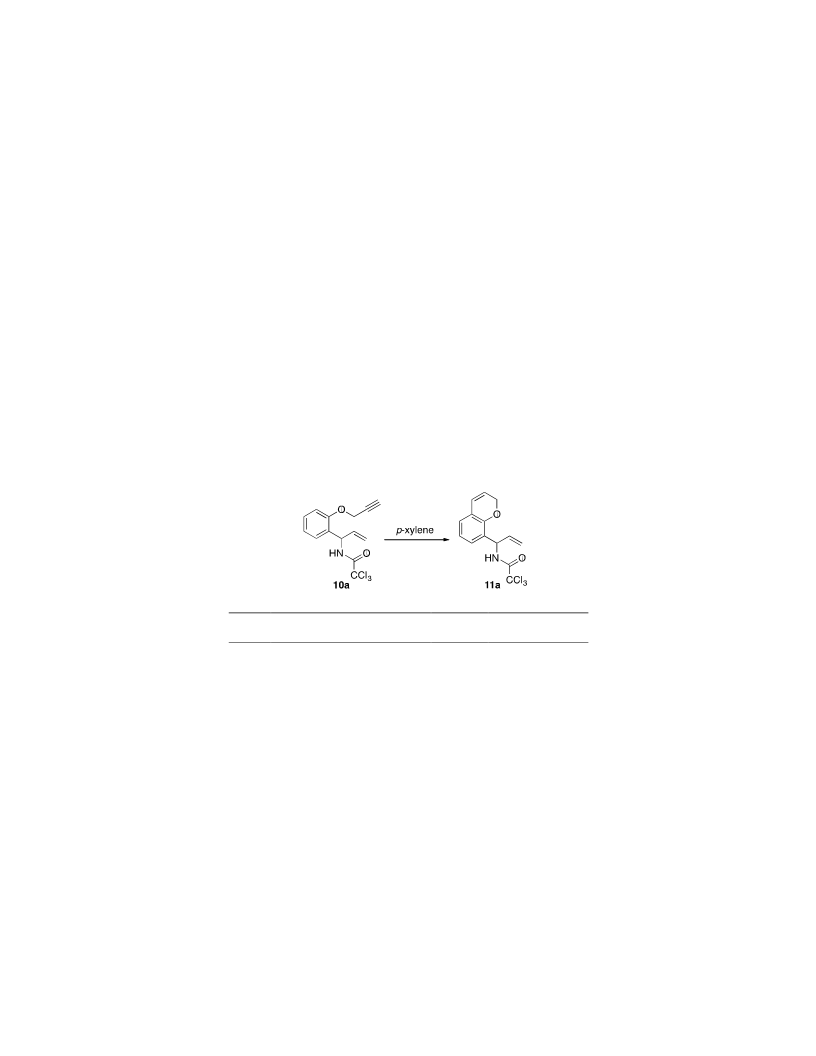
A four-step synthesis of allylic trichloroacetimidates bearing a 2-proparyloxyaryl group has been developed from readily available 2-hydroxybenzaldehydes, and these have been used for the preparation of allylic amide derived 2H-chromenes using an Overman rearrangement and a 6-endo-dig hydroarylation. High yields of the 2H-chromenes were achieved using a stepwise approach involving an Overman rearrangement under thermal conditions followed by a hydroarylation reaction with a gold(I) triflimide catalyst. An alternative method where both reactions were performed as a one-pot process was also developed and instead used a gold(I) chloride catalyst activated by silver(I) hexafluoroantimonate for the cycloisomerization step. The allylic amide derived 2H-chromenes were converted to the corresponding coumarin analogues by a pyridinium dichromate (PDC)-mediated chemoselective allylic oxidation.
View More










Beijing Hope Pharmaceutical Co., Ltd.
Contact:86-10-67886402
Address:309,Block 1 District B,No.12 Hongda North Road , Beijing Economic-Technological Development Area, Beijing, China
Jiangxi Hito Chemical Co., Ltd.
Contact:+86-792-3170318
Address:No. 6, Tianhong Ave., Xinghuo Industry Park, Yongxiu, Jiujiang, Jiangxi, China
Contact:86-512-69362780,69362785
Address:No.69 Weixin Road,Weiting Town,Suzhou Industrial Park
Chongqing Acme Tech. Co., Ltd.
Contact:+86-23-89118971
Address:2501 Huachuang Bldg., No.10 Red-yellow Road, Jiangbei
Tianjin Emulsion Science&Technology Development Co.,Ltd
Contact:13901380442
Address:Vake Garden New Town New Yi Bai Road Beichen District Tianjin,China
Doi:10.1021/jo01267a018
(1968)Doi:10.1016/j.apcata.2010.08.056
(2010)Doi:10.1039/c39790000285
(1979)Doi:10.1021/acs.orglett.8b01457
(2018)Doi:10.1007/s11837-001-0103-y
(1914)Doi:10.1002/oms.1210140509
(1979)