G. Temel et al. / Journal of Photochemistry and Photobiology A: Chemistry 219 (2011) 26–31
27
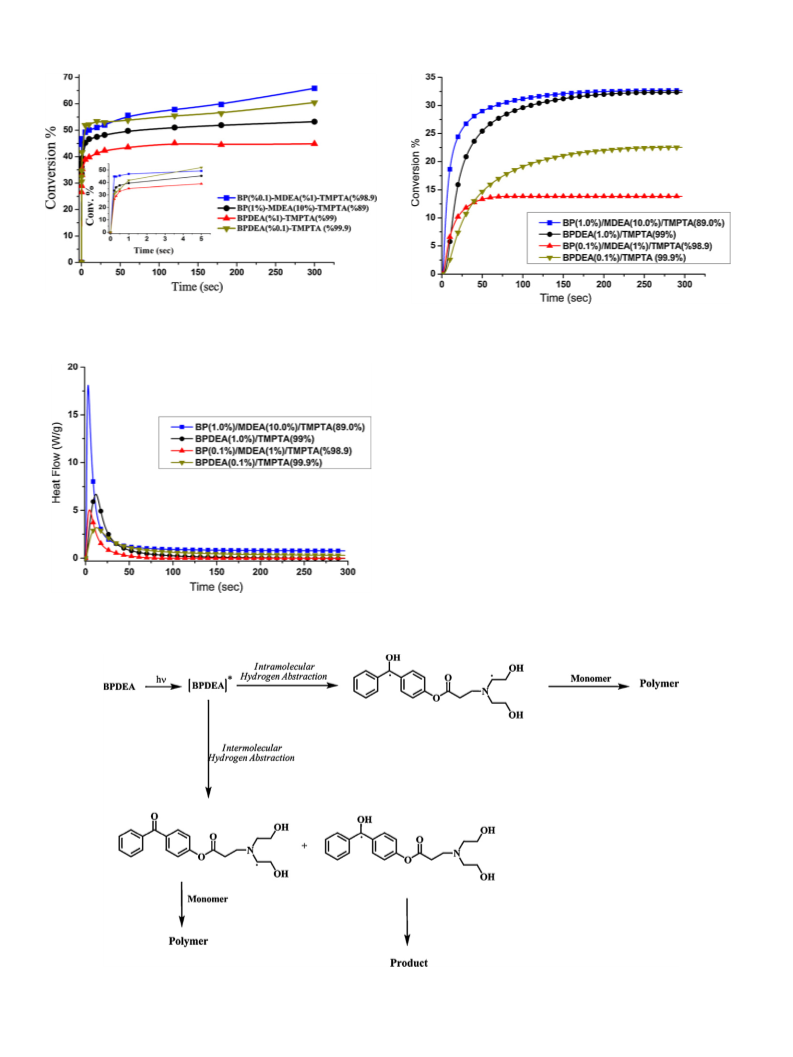
Scheme 1. Photoinitiated free radical polymerization by using aromatic carbonyl compound in the presence of N-methyldiethanolamine as a coinitiator.
NaOH solution and dried over Na2SO4 just before use. Trimethylol-
propane triacrylate (TMPTA) was purchased from Cognis and used
as received.
2.5. General photopolymerization procedure of MMA
Appropriate solutions of the MMA, photoinitiators and coini-
tiator were irradiated in a photoreactor consisting of 12 lamps at
ꢀ = 350 nm for 60 min in either air or N2 atmosphere. The pho-
toreactor consists of 12 Philips black lights the light intensity was
measured as I0(UV-A) = 13 W m−2. Polymers were obtained after pre-
cipitation in methanol and drying under vacuum. Conversions were
calculated gravimetrically.
2.2. Instruments
UV–vis spectra were taken on a Varian UV-Visible Carry 50
Spectrophotometer. Phosphorescence spectra were recorded on a
Jobin Yvon–Horiba Fluoromax-P in cold finger at 77 K. A Nicolet
6700 FT-IR spectrophotometer was used for recording IR spectra.
Gas chromatography–mass spectroscopy (GC–MS) was performed
using an Agilent 6890/5973 inert gas chromatograph/mass selec-
tive detector system in electrospray ionization mode (70 eV),
through an HP-5MS capillary column using helium as the carrier
gas at a flow rate of 1.6 mL min−1. Gel permeation chromatogra-
phy (GPC) analyses of the polymers were performed with a set-up
consisting of a pump (Waters) and four ultrastyragel columns of
different porosities. Tetrahydrofuran (THF) was used as the eluent
(flow rate 0.3 mL min−1), and detection was carried out with the
aid of a differential refractometer. The number-average molecular
weights were determined using polystyrene standards.
2.6. Real-time infrared spectroscopy photopolymerization studies
Uniform samples of photocurable formulations consisting of a
photoinitiator, co-initiator and multifunctional monomer (TMPTA)
were prepared by casting on a KBr pellet. The samples were placed
in the compartment of a Fourier transform infrared spectrometer
and were simultaneously exposed to a UV photolyzing light and an
IR analyzing light beam. The photolyzing light was generated by
a medium-pressure mercury lamp (Flexicure UV system) and was
directed through a flexible fiber optic to the IR compartment. The
spectrometer was operated in the absorbance mode and the detec-
tion wavelength was set at 810 cm−1 (C C–H twist) to monitor the
disappearance of the double bonds. The degree of conversion, ˛,
can be expressed by the following relation:
2.3. Synthesis of acrylic acid 4-benzoyl-phenyl ester (BPAc)
Acryloyl chloride (0.6 mL, 7.5 mmol) in DCM (3 mL) was added
dropwise at 0 ◦C to a solution of 4-hydroxybenzophenone (0.99 g,
5 mmol) and triethylamine (0.70 mL, 5 mmol) in dichloromethane
(DCM) (5 mL), and the resulting mixture was stirred at room tem-
perature overnight. Then the mixture was washed two times with
water (100 mL): a saturated aqueous solution of NaHCO3 (200 mL)
and a saturated aqueous solution of NaCl (100 mL). The organic
layers were dried over anhydrous Na2SO4, filtered, and then the
solvent was evaporated. The resulting residue was purified by
recrystallization from n-hexane. Yield: 80%, mp: 48.5 ◦C.
(A0 − At)
˛ =
A0
−1
where A0 is the initial absorbance at 810 cm
absorbance value at irradiation time t.
and At is the
2.7. Photo differential scanning calorimetri (Photo-DSC)
The photoinitiated polymerization of TMPTA was carried out
by TA-DSCQ100 equipped with a medium pressure mercury arc
lamp. This unit emits radiation predominantly in the 220–400 nm
range and provides light intensity of 40 mW cm−2 as measured by
a UV radiometer capable of broad UV range coverage. The mass of
the samples was approximately 2 0.1 mg and the measurements
were carried out in isothermal mode at room temperature under
a nitrogen atmosphere (nitrogen flow: 50 mL min−1). The samples
were irradiated for 300 s at room temperature. The heat flow as a
isothermal conditions, and both the rate of polymerization and
conversion were then calculated as a function of time. The heat of
reaction value ꢁHptheor = 86 kJ/mol was used as the theoretical heat
evolved for acrylate double bonds [17]. Rates of polymerization
were calculated according to the following equation; Rp = (Q/s)M/n
ꢁHpm where Q/s is heat flow per second, M is the molar mass of the
monomer, n is the number of double bonds per monomer molecule
and m is the mass of monomer in the sample.
1H NMR (250 MHz, CDCl3), 7.87–6.04 ppm (m, 9H, aromatic),
6.68–6.61 ppm (d, 1H, CH CH2), 6.38–6.34 ppm (m, 2H).
FT-IR (ATR): 3060, 2363, 1730, 1646, 1594 cm−1
GC–MS (EI, 70 eV); 252 (M+)
.
2.4. Synthesis of 3-[bis-(2-hydroxy-ethyl)-amino]-propionic acid
4-benzoyl-phenyl ester (BPDEA)
BPAc (0.5 g, 1.98 mmol) and then diethanolamine (0.2 g,
1.98 mmol) were added to 10 mL of ethanol. The mixture was
stirred at room temperature for 30 min. Then the temperature of
the solution was raised to 35 ◦C and kept at that temperature for
4 h. After the TLC control, ethanol was removed under high vacuum
at room temperature.
FT-IR (ATR): 3362, 2949, 1731, 1638, 1599 cm−1
.
UV (CHCl3): ꢀmax(ε) = 294 nm (10,400 mol−1 L cm−1).
GC–MS (EI, 70 eV): 198, 121, 105, 77.

 Temel, Gokhan
Temel, Gokhan
 Enginol, Burak
Enginol, Burak
 Aydin, Meral
Aydin, Meral
 Balta, Demet Karaca
Balta, Demet Karaca
 Arsu, Nergis
Arsu, Nergis