obtained as a white crystals (56 mg, 50 %). Mp 75–77 °C. IR
obtained as white crystals (190 mg, 71 %). Mp 100–104 °C. IR
-1 1
(neat) 2940, 1315, 1189, 1154, 1098 cm . H NMR (300 MHz,
-
1
1
(neat) 3076, 2940, 1187, 1120, 1093 cm . H NMR (300 MHz,
CDCl ) 8.52 (d, 1H, J 8.1 Hz, H ), 8.41 (dd, 2H, J 15.7, 8.1
CDCl ) 8.47 (d, 1H, J 8.1 Hz, H ), 8.39 (d, 1H, J 8.1 Hz, H ),
3
Ar
3
Ar
Ar
Hz, H ), 7.89 (d, 1H, J 8.1 Hz, H ), 7.82 (t, 2H, J 7.7 Hz, H ),
8.18 (d, 1H, J 7.7 Hz, H ), 7.88–7.79 (m, 1H, H ), 7.73–7.64
Ar
Ar
Ar
Ar Ar
7
6
1
2
.71–7.59 (m, 2H, H ), 7.54–7.45 (m, 1H, H ), 4.52 (d, 2H, J
(m, 1H, H ), 7.45–7.31 (m, 2H, H ), 4.56 (d, 2H, J 6.2 Hz, H-
Ar Ar
Ar
Ar
.2 Hz, H-1), 3.14–2.93 (m, 4H, H-5 + H-4eq), 2.43 (t, 2H, J
1.0 Hz, H-4ax), 2.08–1.86 (m, 3H, H-3eq + H-2), 1.71–1.50 (m,
1), 3.12–2.94 (m, 4H, H-5 + H-4eq), 2.50–2.36 (m, 2H, H-4ax),
13
2.07–1.86 (m, 3H, H-3eq + H-2), 1.70–1.52 (m, 2H, H-3ax).
C
1
3
H, H-3ax). C NMR (100 MHz, CDCl ) 158.8 (CNO), 143.3
NMR (100 MHz, CDCl ) 158.9 (CNO), 157.5 (d, J 251.7 Hz,
3
3
(
(
(
(
C ), 134.8 (C ), 130.8 (C ), 128.7 (C ), 127.8 (C ), 127.2
C ), 124.9 (C ), 124.3 (C ), 122.4 (C ), 122.1 (C ), 121.9
C ), 120.1 (C ), 119.1 (qt, J 286.7, 36.0 Hz, CF CF ), 114.9
tq, J 254.6, 36.0 Hz, CF CF ), 70.2 (CH O), 57.0 (t, J 22.4 Hz,
C F), 134.2 (d, J 2.9 Hz, C ), 132.7 (d, J 10.7 Hz, C ), 131.1
Ar
Ar
Ar
Ar
Ar
Ar
Ar
Ar
(C ), 127.7 (C ), 125.1 (C ), 124.5 (d, J 1.9 Hz, C ), 123.9 (d,
Ar
Ar
Ar
Ar
Ar
Ar Ar Ar Ar
J 7.8 Hz, C ), 122.2 (C ), 120.2 (C ), 119.1 (qt, J 286.7, 35.0
Ar Ar Ar
Ar
Ar
2
3
Hz, CF CF ), 117.5 (d, J 3.9 Hz, C ), 114.9 (tq, J 254.6, 36.0
2
3
2
2 3 Ar
19
CH CF ), 54.6 (CH N), 35.2 (CHCH O), 29.3 (CH CH N).
F
Hz, CF CF ), 113.9 (d, J 19.4 Hz, C ), 70.4 (CH O), 57.0 (t, J
2 3 Ar 2
2
2
2
2
2
2
NMR (282 MHz, CDCl ) 84.1 (s, 3F, CF ), 119.3 (t, 2F, J
22.4 Hz, CH CF ), 54.6 (CH N), 35.2 (CHCH O), 29.3
2 2 2 2
19
3
3
+
1
7.2 Hz, CF ). LC-MS (ESI): t 9.65 min; [M+H] : 425.30.
(CH CH N). F NMR (282 MHz, CDCl ) 84.2 (s, 3F, CF ),
2
R
2
2
3
3
+
HRMS/ESI calcd. for C H F N O [M+H] 425.1647, found
119.3 (t, 2F, J 17.2 Hz, CF ), 126.8 - 126.6 (m, 1F, C F).
2
2
22
5
2
2
Ar
+
4
25.1646.
LC-MS (ESI): t 9.55 min; [M+H] : 443.31. HRMS/ESI calcd.
for C H F N O [M+H] 443.1553, found 443.1555.
R
+
2
2
21
6
2
4
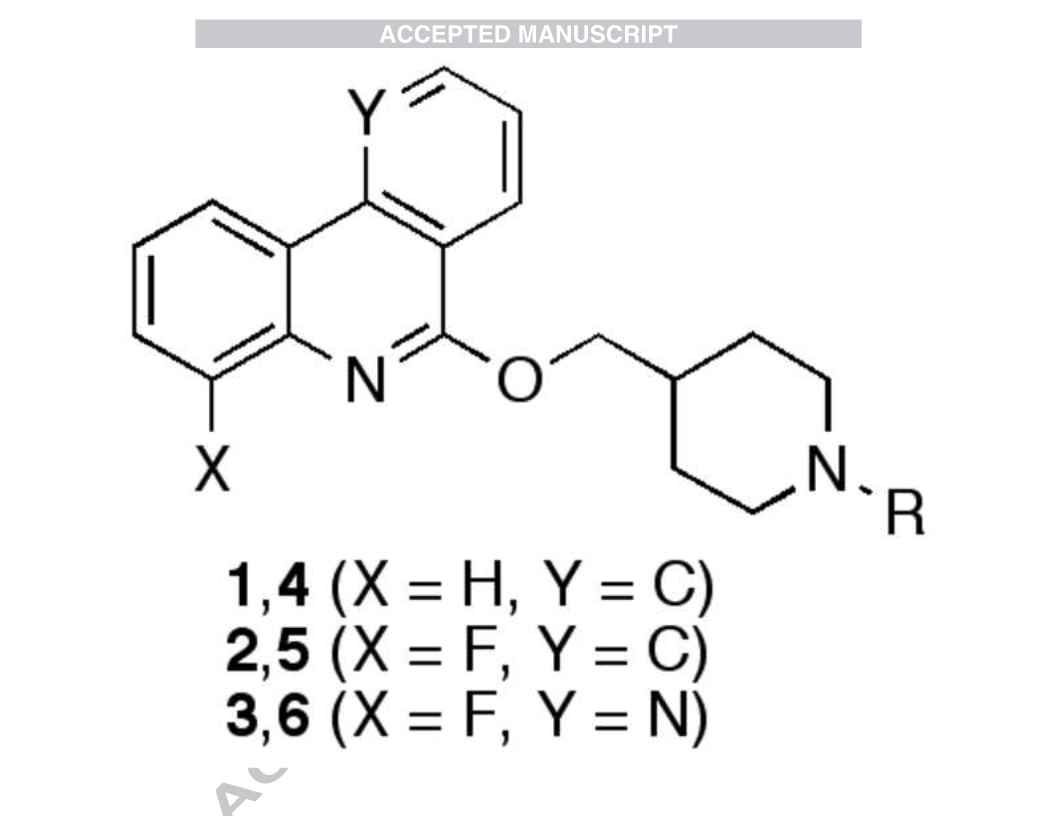
-Fluoro-6-[1-(2,2-difluoroethyl)piperidin-4-
yl]methyloxyphenanthridine 5a. Starting from 17 (57 mg, 0.27
mmol), following general procedure C and using n-hexane/ethyl
acetate 9/1 as the eluent for the column chromatography, 5a was
obtained as yellow crystals (79 mg, 79 %). Mp 128-130 °C. IR
4-Fluoro-6-[1-(3,3,3-trifluoropropyl)piperidin-4-
yl]methyloxyphenanthridine 5d. Starting from 17 (66 mg, 0.31
mmol), following general procedure
C
and using n-
hexane/acetone 85/15 as the eluent for the column
chromatography, 5d was obtained as yellow crystals (68 mg, 54
%). Mp 178–181 °C. IR (neat) 2937, 2784, 2745, 1662, 1591,
-
1 1
(neat) 2940, 1590, 1314, 1128, 1049 cm . H NMR (300 MHz,
CDCl ) 8.47 (d, 1H, J 8.0 Hz, H ), 8.39 (d, 1H, J 8.1 Hz, H ),
3
Ar
Ar
-
1 1
8
7
4
4
4
3
2
.18 (dd, 1H, J 7.0, 1.5 Hz, H ), 7.87–7.78 (m, 1H, H ), 7.72–
1317, 1223, 1153 cm . H NMR (300 MHz, CDCl ) 8.44 (d,
Ar
Ar
3
.63 (m, 1H, H ), 7.45–7.30 (m, 2H, H ), 5.93 (tt, 1H, J 56.0,
1H, J 3.0 Hz, H ), 8.40-8.36 (m, 1H, H ), 8.20-8.10 (m, 1H,
Ar
Ar
Ar
Ar
.4 Hz, H-6), 4.56 (d, 2H, J 5.9 Hz, H-1), 3.09–2.98 (m, 2H, H-
eq), 2.78 (td, 2H, J 15.0, 4.4 Hz, H-5), 2.36–2.22 (m, 2H,H-
ax), 2.08–1.87 (m, 3H, H-3eq + H-2), 1.69–1.51 (m, 2H, H-
H ), 7.81 (ddd, 1H, J 8.3, 7.1, 1.4 Hz, H ), 7.66 (ddd, 1H, J 8.1,
A
r
A
r
7.1, 1.1 Hz, H ), 7.41-7.30 (m, 2H, H ), 4.55 (d, 2H, J 6.1 Hz,
Ar
Ar
H1), 3.04-2.86 (m, 2H, H-4eq), 2.70-2.60 (m, 2H, H5), 2.48-2.27
1
3
1
ax). C NMR (75 MHz, CDCl ) 158.9 (CNO), 157.4 (d, J
(m, 2H, H6), 2.09 (td, 2H, J 11.7, 2.2 Hz, H4-ax), 2.04-1.88 (m,
3
1
3
52.1 Hz, C F), 134.3 (d, J 3.3 Hz, C ), 132.7 (d, J 10.0 Hz,
3H, H2 + H-3eq), 1.70-1.50 (m ,2H, H-3ax). C NMR (100
Ar
Ar
C ), 131.2 (C ), 127.7 (C ), 125.1 (C ), 124.5 (d, J 2.2 Hz,
MHz, CDCl ) δ 158.9 (CNO), 157.5 (d, J 246 Hz, C F), 134.3
Ar
Ar
Ar
Ar
3
Ar
C ), 123.9 (d, J 7.7 Hz, C ), 122.2 (C ), 120.2 (C ), 117.6 (d, J
(d, J 3 Hz, C ), 132.7 (d, J 10 Hz, C ), 131.2 (C ), 127.7 (C ),
126.7 (q, J 275 Hz, CF ), 125.2 (C ), 124.5 (C ), 123.9 (d, J 7
3 Ar Ar
Ar
Ar
Ar
Ar
Ar Ar Ar Ar
1
4
.4 Hz, C ), 115.8 (t, J 241.0 Hz, CHF ), 114.0 (d, J 18.8 Hz,
Ar
2
2
C ), 70.5 (CH O), 60.6 (t, J 25.4 Hz, CH CHF ), 54.3 (CH N),
3
Hz, C ), 122.2 (C ), 120.2 (C ), 117.6 (d, J 3 Hz, C ), 114.0
Ar
2
2
2
2
Ar Ar Ar Ar
1
9
5.3 (CHCH O), 29.2 (CH CH N). F NMR (282 MHz, CDCl3)
(d, J 19 Hz, C ), 70.5 (C1), 53.5 (C4), 51.1 (d, J 3 Hz, C5), 35.7
Ar
19
2
2
2
118.3 (dt, 2F, J 55.9, 17.2 Hz, CHF ), 126.7 (br. s, 1F,
(C2), 31.9 (q, J 28 Hz, C6), 29.2 (C3). F NMR (282 MHz,
CDCl ) δ -65.6 (t, J 8.5 Hz, CF ), -126.7 (m, C F). LC-MS
2
+
C F). LC-MS (ESI): t 5.41 min; [M+H] : 375.46. HRMS/ESI
Ar
R
3
3
Ar
+
+
calcd. for C H F N ONa [M+Na] 397.1498, found 397.1493.
(ESI): tR 5.54 min; [M+H] : 407.34. HRMS/ESI calcd. for
2
1
21
3
2
+
C H F N O [M+H] 407.1747, found 407.1741.
2
2
23
4
2
4
-Fluoro-6-[1-(2,2,2-trifluoroethyl)piperidin-4-
yl]methyloxyphenanthridine 5b. Starting from 17 (60 mg, 0.28
mmol), following general procedure C and using n-hexane/ethyl
acetate 9/1 as the eluent for the column chromatography, 5b was
obtained as white crystals (77 mg, 69 %). Mp 144–146 °C. IR
4-Fluoro-6-[1-(4,4,4-trifluorobutyl)piperidin-4-
yl]methyloxyphenanthridine 5e. Starting from 17 (55 mg, 0.26
mmol), following general procedure and using n-
C
hexane/acetone 3/1 as the eluent for the column chromatography,
-
1 1
(
neat) 3078, 2940, 1125, 1094 cm . H NMR (300 MHz, CDCl )
5e was obtained as a yellow oil (45 mg, 41 %). IR (neat) 2939,
3
-
1 1
1
7
3
2
8.47 (d, 1H, J 8.1 Hz, H ), 8.39 (d, 1H, J 8.0 Hz, H ), 8.18 (d,
2820, 2771, 2359, 1591, 1318, 1247, 1108 cm . H NMR (300
Ar
Ar
H, J 7.0 Hz, H ), 7.83 (td, 1H, J 8.1, 0.7 Hz, H ), 7.68 (t, 1H, J
MHz, CDCl ) 8.46 (d, 1H, J 8.3 Hz, H ), 8.40 (dd, 1H, J 8.1,
Ar
Ar
3
Ar
.7 Hz, H ), 7.45–7.31 (m, 2H, H ), 4.56 (d, 2H, J 6.2 Hz, H-1)
1.0 Hz, H ), 8.17 (d, 1H, J 7.3 Hz, H ), 7.82 (ddd, 1H, J 8.3,
Ar
Ar
Ar Ar
.12–2.96 (m, 4H, H-4eq + H-5), 2.51–2.39 (m, 2H, H-4ax),
7.1, 1.3 Hz, H ), 7.72-7.65 (m, 1H, H ), 7.46-7.32 (m, 2H, H ),
Ar Ar Ar
1
3
.08–1.88 (m, 3H, H-3eq + H-2), 1.69–1.52 (m, 2H, H-3ax).
C
4.55 (d, 2H, J 6.1 Hz, H1), 3.03-2.92 (m ,2H, H-4eq), 2.45-2.35
(m, 2H, H5), 2.25-2.08 (m, 2H, H7), 2.06-1.90 (m, 5H, H4-ax +
H2 + H-3eq), 1.85-1.72 (m, 2H, H6), 1.65-1.50 (m ,2H, H-3ax).
NMR (75 MHz, CDCl ) 158.9 (s, CNO), 157.4 (d, J 251.0 Hz,
3
C F), 134.3 (d, J 3.3 Hz, C ), 132.7 (d, J 11.1 Hz, C ), 131.2
Ar
Ar
Ar
1
3
(
C ), 127.8 (C ), 125.6 (q, J 280.9 Hz, CF ), 125.1 (C ), 124.5
C NMR (100 MHz, CDCl ) δ 159.0 (CNO), 157.5 (d, J 251 Hz,
Ar
Ar
3
Ar
3
(
d, J 2.2 Hz, C ), 123.9 (d, J 7.7 Hz, C ), 122.2 (C ), 120.2
C F), 134.3 (d, J 3 Hz, C ), 132.7 (d, J 10 Hz, C ), 131.2 (C ),
Ar Ar Ar Ar
Ar
Ar
Ar
(
C ), 117.6 (d, J 4.4 Hz, C ), 114.0 (d, J 18.8 Hz, C ), 70.4
127.8 (C ), 127.4 (q, J 275 Hz, CF ), 125.2 (C ), 124.5 (C ),
Ar
Ar
Ar
Ar 3 Ar Ar
(
CH O), 58.8 (q, J 31.0 Hz, CH CF ), 54.0 (CH N), 35.2
123.9 (d, J 8 Hz, C ), 122.3 (C ), 120.3 (C ), 117.6 (d, J 5 Hz,
Ar Ar Ar
2
2
3
2
1
9
(CHCH O), 29.3 (CH CH N). F NMR (282 MHz, CDCl )
C ), 114.0 (d, J 19 Hz, C ), 70.7 (C1), 57.4 (C5), 53.5 (C4),
Ar Ar
19
2
2
2
3
69.0 (t, 3F, J 8.6 Hz, CF ), 126.7 (br. s, 1F, C F). LC-MS
35.9 (C2), 31.8 (q, J 29 Hz, C7), 29.3 (C3), 19.7 (C6). F NMR
(282 MHz, CDCl ) δ -66.5 (t, J 11.3 Hz, CF ), -126.7 (m, C F).
3
Ar
+
(
ESI): tR 8.41 min; [M+H] : 393.29. HRMS/ESI calcd. for
3
3
Ar
+
+
C H F N O [M+H] 393.1585, found 393.1591.
LC-MS (ESI): t 5.65 min; [M+H] : 421.42. HRMS/ESI calcd.
for C H F N O [M+H] 421.1903, found 421.1898.
2
1
21
4
2
R
+
2
3
25
4
2
4
-Fluoro-6-[1-(2,2,3,3,3-pentafluoropropyl)piperidin-4-
yl]methyloxyphenanthridine 5c. Starting from 17 (128 mg, 0.6
mmol), following general procedure C and using n-hexane/ethyl
acetate 9/1 as the eluent for the column chromatography, 5c was
7-Fluoro-5-[1-(2,2-difluoroethyl)piperidin-4-
yl]methyloxybenzo[h]-1,6-naphthyridine 6a. Starting from 18
(99 mg, 0.46 mmol), following general procedure C and using n-

 Fontenelle, Clement Q.
Fontenelle, Clement Q.
 Wang, Zhong
Wang, Zhong
 Fossey, Christine
Fossey, Christine
 Cailly, Thomas
Cailly, Thomas
 Linclau, Bruno
Linclau, Bruno
 Fabis, Frederic
Fabis, Frederic