thracene 2-sulfonate (bromaminic acid sodium salt, 1) with
an arylamine in the presence of a copper catalyst (Scheme
Table 1. Comparison of Yields of Classical Methods (A and
1
c-f
1).
The reaction typically requires harsh conditions, e.g.,
B) and Method C without or with Application of Microwaves
MW
MW
MW
(A
, B , C ) for Selected Compounds
a
method (catalyst)
Scheme 1. Syntheses of Anilinoanthraquinone Derivatives
(
CuCl)
AMW
(CuSO4)
(Cu0)
products
Ab
B
BMW
C
CMW
3
3
3
3
3
3
3
3
3
3
a
b
c
d
e
f
g
h
i
0%
nd
nd
nd
nd
nd
nd
nd
nd
nd
0%
<5%
0%
0%
0%
<5%
0%
<5%
nd
20%
30%
0%
0%
0%
10%
5%
20%
0%
nd
60%
60%
<5%
15%
70%
76%
70%
58%
70%
43%
64%
83%
58%
72%
0%
0%
0%
0%
0%
<10%
nd
5%
<10%
<5%
20%
40%
50%
80%
0%
j
40%
0%
a
For reaction conditions A, B, and C, see Scheme 1. Reaction conditions
AMW, B , and C : same reagents as for A, B, and C, but application of
microwaves, 40-100 W, 80-120 °C, 1-20 min. General procedure A:
rt; heating did not affect the yields significantly.
MW
MW
b
tory yield of 40% (Table 1). Subsequent rise of the
temperature from room temperature to 40, 60, 80, 100, up
to 120 °C did not alter the yields. Results with the classical
method B applying copper(II) sulfate and sodium carbonate
at temperatures up to 120 °C were similarly disappointing
high temperatures and long reaction times, and it suffers from
mostly poor yields.1
c-f
(
Table 1). A thorough search of the patent literature yielded
Our group has been interested in the preparation of a series
of anilinoanthraquinone derivatives derived from the dye
Reactive Blue 25 (3l) (Figure 1) for pharmacological
6
hints that the use of Cu(0) might be advantageous. We
therefore performed the reactions in the presence of Cu(0)
in sodium phosphate buffer, pH 6-7, at 120 °C for 2-15 h
4
evaluation as antagonists of purine P2 receptors and as
(method C).
5
potential ectonucleotidase inhibitors, to study their structure-
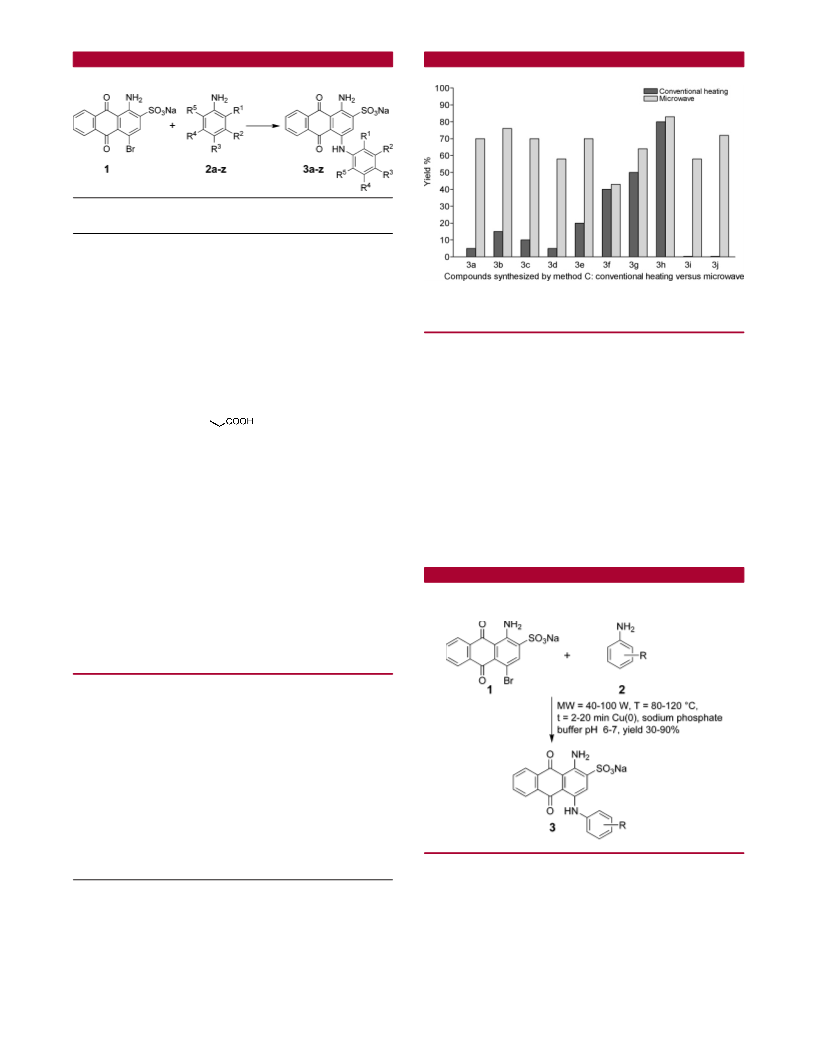
In comparison to the classical methods A and B, method
activity relationships.
C appeared to be superior. Only the phenolic products 3i
and 3j were obtained in higher yields by methods A and B.
This may be explained by the basic conditions applied in
methods A and B (pH ∼ 9) but not in method C (pH 6-7).
The phenolate anion formed at pH 9 increases the electron
density in the ring and as a consequence increases the
nucleophilicity of the amino group. Nevertheless, not only
the classical methods A and B but also method C, despite
its superiority, were still unsatisfactory. Recently, microwave
Initially, we investigated two different classical procedures
for reacting bromaminic acid sodium salt (1) with aromatic
1
c,d
amines (2a-j) as outlined in Scheme 1: method A,
reaction in the presence of CuCl, Na CO , and Na SO
2
3
2
3
in
H O at room temperature for 8-24 h, or under reflux at 120
2
1f
°
C for 8-10 h; and method B, reaction in the presence of
CuSO and Na CO in H O at 120 °C for 12-48 h. In many
4
2
3
2
cases, no products or only poor yields could be obtained.
The main undesirable product identified in all reactions was
(
MW) irradiation has emerged as an efficient tool in organic
1-amino-4-hydroxy-9,10-dioxo-9,10-dihydroanthracene 2-sul-
7-9
synthesis, and its benefits have been well documented.
fonate (4, Scheme 1) formed by attack of the competing
nucleophile hydroxide. A comparison of the two standard
methods A and B for the preparation of a set of 10 selected
compounds 3a-j is shown in Table 1. Method A using
copper(I) chloride as a catalyst performed in the presence
of sodium carbonate and sodium sulfite did not lead to
detectable conversion in most cases (monitoring by RP-TLC).
Only the p-phenol derivative (3j) was obtained in a satisfac-
In a number of studies, it has been shown that microwave
irradiation can circumvent the need for prolonged heating
and it generally accelerates the rate of chemical reactions,
often with increased yields. The use of MW irradiation for
the formation of carbon-carbon as well as carbon-hetero-
(6) Eltz, A. Reactive dyes useful for dyeing and printing e.g. cellulose,
polyamide or polyester. DE 4417719A1, 1995.
(
7) (a) Kappe, C. O.; Dallinger, D. Nat. ReV. Drug DiscoVery 2006, 5,
(
4) (a) M u¨ ller, C. E. Curr. Pharm. Des. 2002, 8, 2353. (b) Brunschweiger,
A.; M u¨ ller, C. E. Curr. Med. Chem. 2006, 13, 289.
5) (a) Iqbal, J.; Vollmayer, P.; Braun, N.; Zimmermann, H.; M u¨ ller, C.
51. (b) Lidstr o¨ m, P.; Tierney, J.; Wathey, B.; Westman, J. Tetrahedron
2001, 57, 9225.
(8) Xu, G.; Wang, Y. G. Org. Lett. 2004, 6, 985.
(9) Hayes, B. L. MicrowaVe Synthesis, Chemistry at the Speed of
Light; CEM Publishing: Matthews, North Carolina, 2002; Chapter 1,
p 16.
(
E. Purinergic Signalling 2005, 1, 349. (b) M u¨ ller, C. E.; Iqbal, J.; Baqi,
Y.; Zimmermann, H.; R o¨ llich, A.; Stephan, H. Bioorg. Med. Chem. Lett.
006, 16, 5943.
2
1272
Org. Lett., Vol. 9, No. 7, 2007

 Baqi, Younis
Baqi, Younis
 Muller, Christa E.
Muller, Christa E.