164 Zunszain et al.
Arch. Pharm. Pharm. Med. Chem. 2002, 335, 159–166
a stirrer, thermometer, and dropping funnel. 2-Chlorobenzo-
trichloride (4.19 mL, 27.2 mmol) dissolved in benzene (7.8 mL)
was added dropwise to this suspension over a period of 3 h,
with stirring, and the mixture was refluxed for 14 h.The reaction
mixture was then diluted with CH2Cl2 (60 mL) and poured into
ice (60 g) and concentrated HCl (29 mL) with stirring. The or-
ganic phase was separated, washed with HCl (4 × 50 mL),
dried (Na2SO4), and evaporated. CH3CN (100 mL) was added
to the residue and stirred for 30 min at 0°C, and the solid prod-
uct was filtered off. This procedure was repeated twice to give
2-chlorophenyl-diphenylmethyl chloride (Ia, 3.1 g, 36%). 3-
Aminopyridine (0.45 g, 4.8 mmol) was placed in a round bottom
flask equipped with stirrer and dropping funnel. Under nitrogen
atmosphere, compound Ia (1.5 g, 4.8 mmol) dissolved in dry
CH3CN (10 mL) was added dropwise while slowly warming the
mixture to 50°C. At the same time, triethylamine (6 mL) was
added dropwise, and the mixture was heated under reflux for
4 h. The solvents were removed in vacuo to give an oil, which
was dissolved in CHCl3 and washed with water.The organic ex-
tract was dried (Na2SO4), filtered and evaporated to an oil,
which was further purified by column chromatography using
CHCl3 as eluent, to give 3a (0.31 g, 18%) as a solid; mp 100–
102°C, decomp. 1H-NMR (400 MHz, CDCl3) δ 7.96 (d, 1H, J =
2.9 Hz), 7.84 (dd, 1H, J = 4.7 Hz, 1.3 Hz), 7.57 (m, 1H), 7.22 (m,
H), 6.79 (m, 1H), 6.62 (m, 1H), 5.45 (bs, 1H). IR 3397 cm–1.
FAB-MS m/z 371 (M + H)+. HPLC system: 80:20, Rt 13.6 min,
purity 99.4%.Anal.(C24H19N2Cl, 0.2 C3H7OH, 0.2 H2O) C, H, N.
Experimental
Chemistry
Materials and methods
Melting points were determined using an Electrothermal melt-
ing point apparatus and are uncorrected.1H NMR spectra were
recorded on a Bruker AMX-300 at 300 MHz, a Bruker AMX-400
at 400 MHz, or a Bruker AVANCE-500 NMR Spectrometer at
500 MHz. Analytical HPLC was carried out on a Shimadzu
HPLC apparatus with a Kromasil C18 5 µm reversed phase col-
umn (250 × 4.6 mm) at a flow rate of 1 mL/min and detected at
254 nm. The mobile phases were mixtures of A = methanol +
0.1% trifluoroacetic acid, and B = water + 0.1%, and are indi-
cated as the ratio A:B. Mass spectra were recorded on a VG
7070H Double Focusing Mass Spectrometer or a VG ZAB-SE
Double Focusing Mass Spectrometer.Infrared spectra were re-
corded on a Perkin-Elmer 1605 FT-IR spectrometer using KBr
discs. Elemental analyses were determined on a Perkin Elmer
2400 CHN elemental analyser and were carried out at UCL
Departmental Microanalysis Service.Column chromatography
was conduced using Merck silica gel 60 (70–230 mesh). TLC
was carried out using appropriate proportions of hexane and
ethyl acetate, on Merck Kieselgel 60 F254 aluminium sheets,
visualised at 254 nm and stained with potassium iodo platinate
or 5% H2SO4 in ethanol. CH3CN was dried by refluxing over
P2O5 and distilled prior to use.Triphenylmethanol (trityl alcohol,
2c) was purchased from Aldrich.Tris-(p-chlorophenyl)-metha-
nol (2d) was purchased from Lancaster, purified by column
chromatography using petroleum spirit-chloroform 7:3 as elu-
ent, and recrystallised from petroleum spirit. Both 2c and 2d
were fully characterized prior to use (HPLC, IR, NMR, FAB,
Anal.).
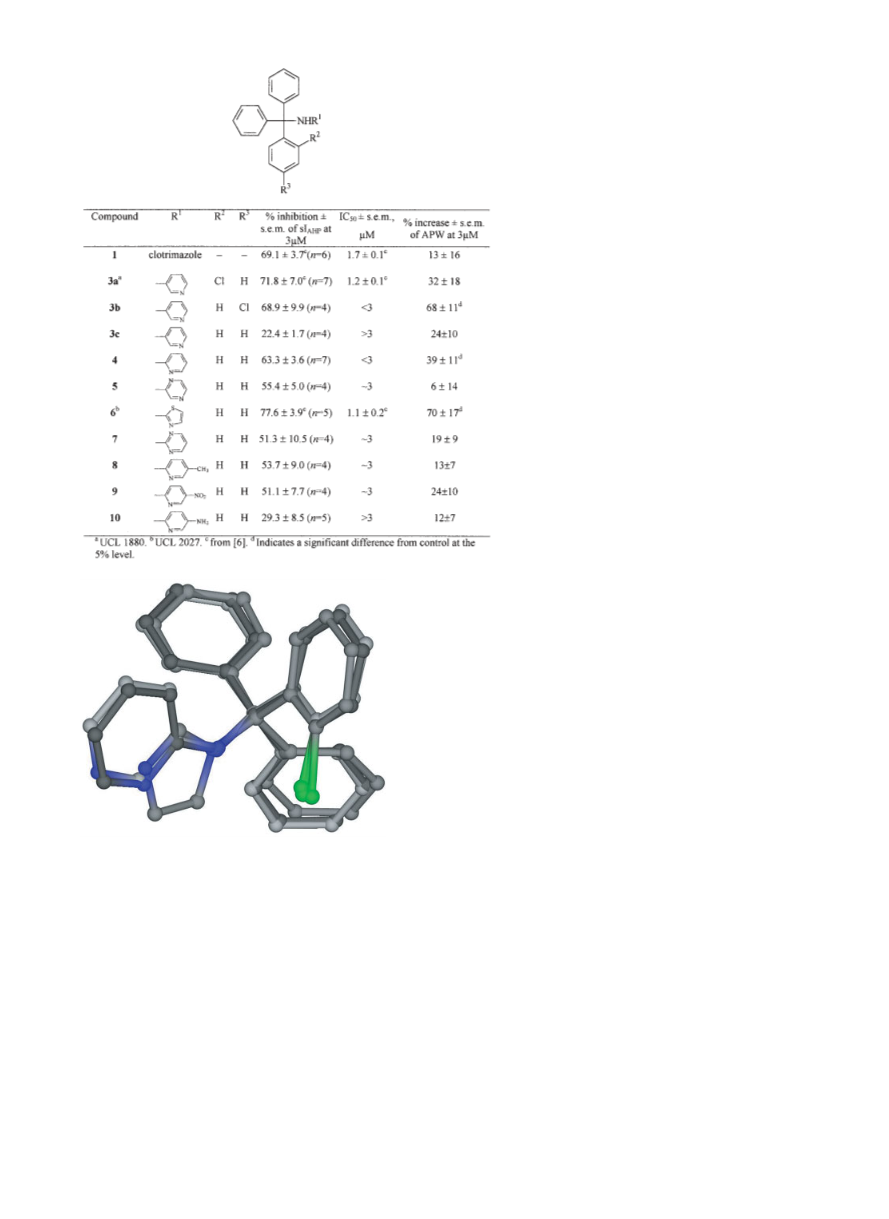
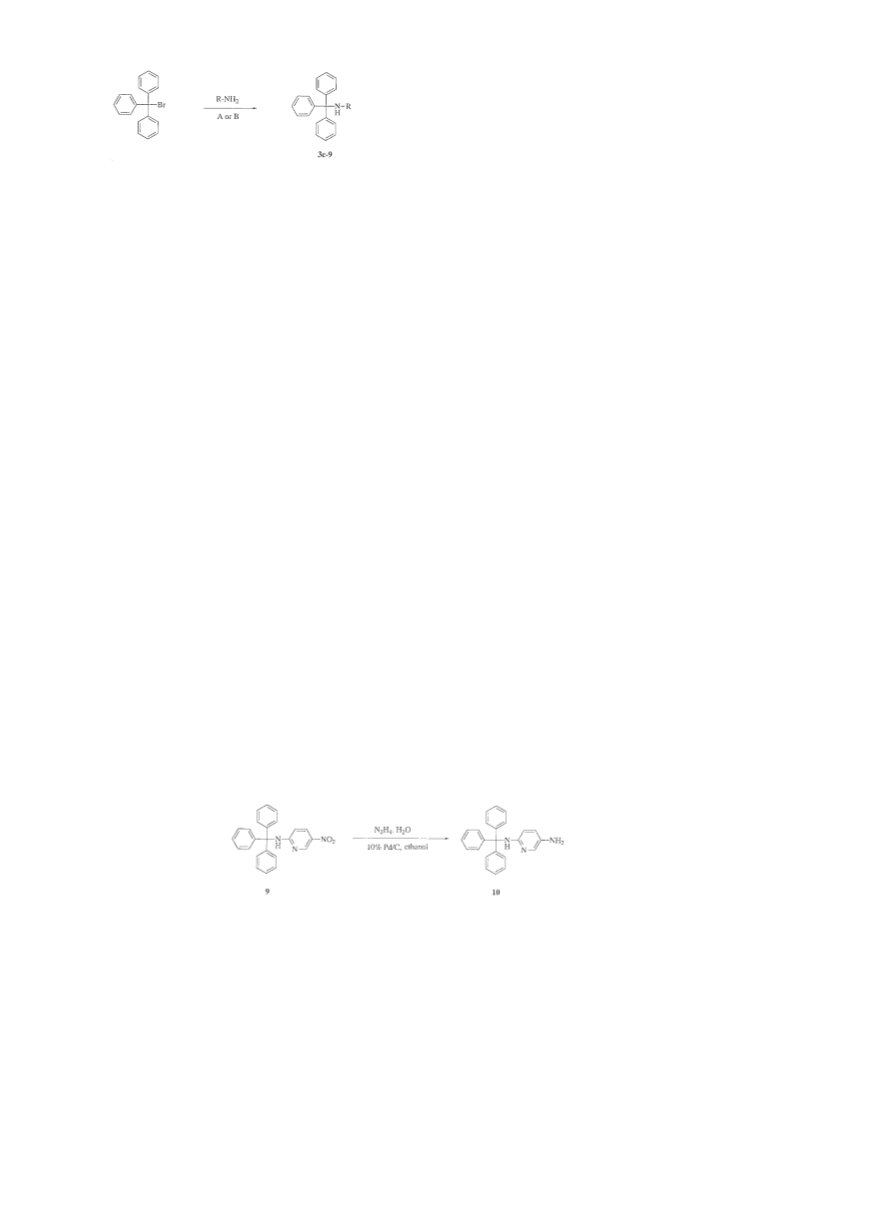
3-{N-1-(4-Chlorophenyl)-1,1-diphenyl]methyl}aminopyridine
(3b)
Prepared as described for 3a, from 3-aminopyridine (0.15 g,
1.59 mmol) and 4-chlorophenyl-diphenylmethyl chloride (Ib,
0.5 g, 1.59 mmol, synthesised from benzene, AlCl3 and 4-chlo-
robenzotrichloride as stated above) to give compound 3b as a
solid (0.5 g, 48%); mp 159–161°C. 1H-NMR (400 MHz, CDCl3)
δ 7.90 (m, 1H), 7.82 (m, 1H), 7.25 (m, 14H), 6.75 (m, 1H), 6.45
(m, 1H), 5.04 (bs, 1H). IR 3409 cm–1. FAB-MNOBA matrix m/z
371 (M)+. HPLC system: 80:20, Rt 14.1 min, purity 98.8%.
Anal. (C24H19ClN2 0.1 H2O) C, H, N.
2-Chlorophenyl-diphenylmethanol (2a)
A solution of clotrimazole (3.9 g, 11.5 mmol) in isopropanol
(280 mL), concentrated HCl (4 mL) and water (120 mL) was
heated at reflux for 17 h. The solution was then evaporated in
vacuo, producing a white solid that was purified by column
chromatography using petroleum spirit-chloroform 9:1 as elu-
ent. The product obtained was recrystallised from petroleum
spirit, to give 1.52 g of 2a (36%); mp 92–94°C (ref. [22] 91–
3-Tritylaminopyridine (3c)
Method A: A solution of trityl bromide (0.12 g, 0.38 mmol) and
3-aminopyridine (0.07 g, 0.77 mmol) in toluene (3 mL) was
heated at reflux for 24 h. After cooling, the solid formed was fil-
tered off, and the solution evaporated to dryness.The syrup ob-
tained crystallised upon addition of EtOH, to give 0.05 g of 3c
(38%). Recrystallised from the same solvent, it gave mp 179–
180°C. 1H-NMR (300 MHz, CDCl3) δ 7.94 (d, 1H, J = 2.6), 7.84
(d, 1H, J = 4.6), 7.31 (m, 15H), 6.82 (dd, 1H, J = 8.4, 4.6), 6.56
(dd, 1H, J = 8.4, 1.8).FAB-MS m/z 337 (M + H)+.HPLC system:
70:30, Rt 7.49 min, purity 99.7%. Anal. (C24H20N2) C, H, N.
1
93°C). H-NMR (400 MHz, CDCl3) δ 7.41 (m, 1H), 7.31 (m,
11H), 7.12 (m, 1H), 6.72 (m, 1H), 4.45 (bs, 1H). IR 3568,
3543 cm–1. FAB-MS m/z 294 (M+). HPLC system: 70:30, Rt
14.2 min, purity 100%. Anal. (C19H15ClO) C, H, Cl.
4-Chlorophenyl-diphenylmethanol (2b)
4-Chlorophenyl-diphenylmethyl chloride (1.0 g, 3.19 mmol)
was dissolved in CH3CN (100 mL). To this solution, NaOH
(1.4 g, 35.3 mmol) dissolved in water (10 mL) was added.The
mixture was heated under reflux for 14 h. The solvent was re-
moved in vacuo to give an oil.Water was added and the result-
ing mixture was extracted with CHCl3 (4 × 50 mL). The com-
bined organic extracts were dried over Na2SO4, filtered and
evaporated in vacuo to give a brown solid, which was recrystal-
lised from petroleum spirit to give 2b (0.32 g, 36%); mp 77–
2-Tritylaminopyridine (4)
Prepared from trityl bromide (1 g, 3.09 mmol) and 2-aminopyri-
dine (0.58 g, 6.18 mmol) according to method A, to give 0.41 g
1
(41%); mp 150–152°C (ref. [24] 150–152°C). H-NMR (300
MHz, CDCl3) δ 8.05 (d, 1H), 7.30 (m, 15H), 7.05 (t, 1H), 6.15
(bs, 1H), 6.05 (t, 1H), 5.80 (d, 1H).FAB-MS m/z 337.3 (M + H)+.
HPLC system: 70:30, Rt 5.8 min, purity 97.6%. Anal.
(C24H20N2) C, H, N.
1
80°C (ref. [23] 84.8–85.5°C). H-NMR (400 MHz, CDCl3) δ
7.36–7.24 (m, 14H), 2.81 (bs, 1H). IR 3444 cm–1 (broad, OH)
FAB MNOBA matrix MS m/z 294 (M – H)+. HPLC system:
80:20, Rt 15.8 min, purity 98.7%. Anal. (C19H15ClO) C, H, Cl.
2-Tritylaminopyrazine (5)
3-{N-1-(2-Chlorophenyl)-1,1-diphenyl]methyl}aminopyridine
(3a, UCL1880)
Method B: to a solution of trityl bromide (1 g, 3.09 mmol) in
CH3CN (10 mL), 2-aminopyrazine (0.29 g, 3.09 mmol) and
Na2CO3 (0.33 g, 3.09 mmol) were added. The solution was
heated at reflux overnight, when TLC showed total transforma-
Anhydrous benzene (17 mL) and aluminium trichloride (4 g,
29.9 mmol) were placed in a round bottomed flask equiped with

 Zunszain, Patricia A.
Zunszain, Patricia A.
 Shah, Mala M.
Shah, Mala M.
 Miscony, Zena
Miscony, Zena
 Javadzadeh-Tabatabaie, Mazyar
Javadzadeh-Tabatabaie, Mazyar
 Haylett, Dennis G.
Haylett, Dennis G.
 Ganellin, C. Robin
Ganellin, C. Robin