802
Chemistry Letters Vol.36, No.6 (2007)
Highly Efficient Electron-transporting Phenanthroline Derivatives
for Electroluminescent Devices
Lixin Xiao,ꢀ1;2 Hsinglin Lan,3 and Junji Kidoꢀ1;3
1Optoelectronic Industry and Technology Development Association, 1-20-10 Sekiguchi, Bunkyo-ku, Tokyo 112-0014
2State Key Laboratory for Artificial Microstructures and Mesoscopic Physics, Department of Physics,
Peking University, Beijing 100871, P. R. China
3Department of Polymer Science and Engineering, Yamagata University, 4-3-16 Jonan, Yonezawa 992-8510
(Received February 26, 2007; CL-070210; E-mail: kid@yz.yamagata-u.ac.jp, xiao66@pku.edu.cn)
A series of phenanthroline derivatives containing quinoxa-
thermal evaporation on quartz. According to the absorption
edge, the energy gap (Eg) of the phenanthroline derivatives
can be calculated and listed in Table 1. The lowest unoccupied
molecular orbitol (LUMO) energy level of the synthesized phen-
anthroline derivatives are equal to or lower than that of Alq3, i.e.,
the electron affinity (Ea) energy of phenanthroline derivatives
might be equal to or higher than that of Alq3 (3.0 eV). Therefore,
it is likely that an equal or higher ET property for the phenan-
throline derivative can be obtained than that of the conventional
ET material Alq3.
line moieties were synthesized, and their electron-transporting
properties were investigated by using N,N0-di(1-naphthyl)-N,N0-
diphenyl-(1,10-biphenyl)-4,40-diamine (ꢀ-NPD) and tris(8-hy-
droxyquinolato)aluminum (Alq3) as the hole-transporting
material and the emitting layer, respectively. Higher external
quantum efficiency for the phenanthroline derivative than Alq3
in the electroluminescent devices at a luminance of 100 cd/m2
was obtained.
To study the ET properties of phenanthroline derivatives,
OLEDs were fabricated using a 50-nm layer of ꢀ-NPD as the
HT material, a 40-nm layer of Alq3 as the emitter. A 30-nm layer
of phenanthroline derivatives 1 and 2 as well as Alq3 (reference
device) was then deposited onto the emitter surface as the ET
material. Finally, 0.5 nm of LiF together with 100 nm of Al were
deposited as the cathode. The device architecture is shown in
Figure 1.
A high efficiency for organic light-emitting device (OLED)
is a crucial factor to promote the application of OLED in the
next generation of flat panel display. The efficiency of OLED
is composed of the recombination efficiency of holes and elec-
trons injected from the electrodes to the organic emitting layer
and photoluminescence quantum yield of the emitting layer.
To improve the efficiency of OLED, the development of highly
efficient materials is the major issue, especially the development
of highly efficient electron-transporting (ET) materials for
OLED. In generally, the mobility of holes in hole-transporting
(HT) materials is much higher than that of electrons in ET
materials for organic semiconducting materials. Moreover, few
materials, mainly bathocuproine1 and bathophenanthroline
(Bphen),2 oxadiazoles,3,4 triazoles,5 1,3,5-tris(N-phenylbenzimi-
dazol-2-yl)benzene (TPBI),6 have been used as ET materials
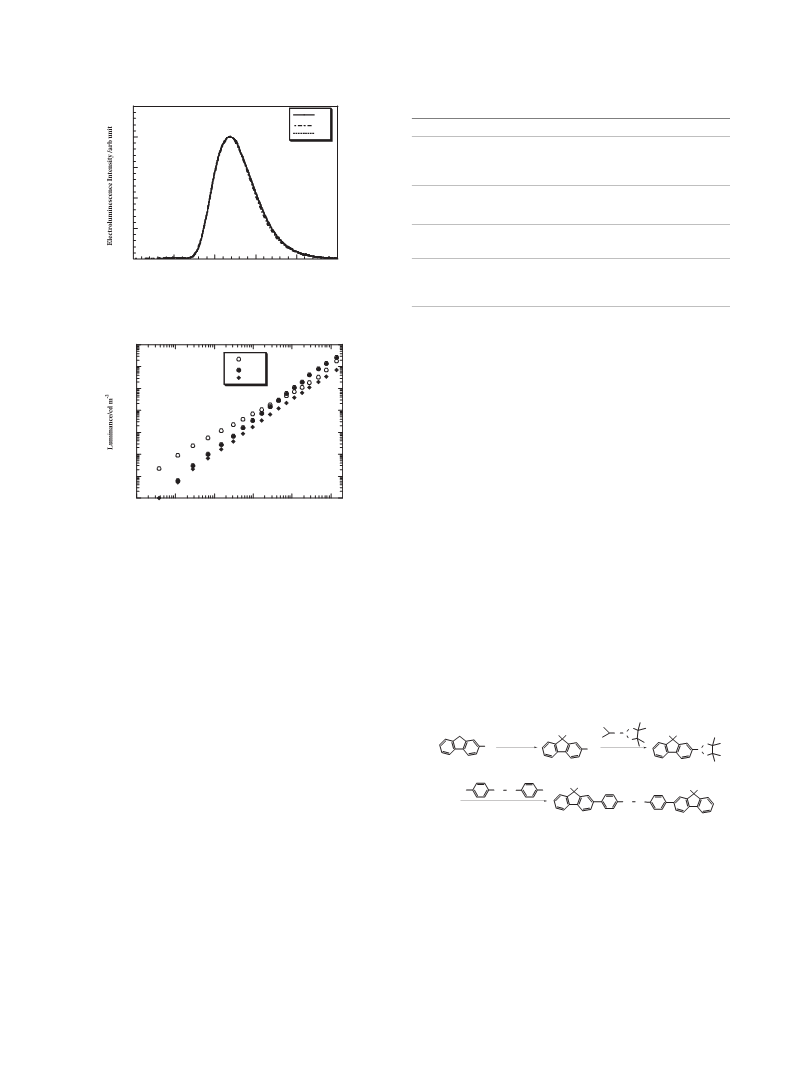
The electroluminescence (EL) spectrum of the OLED is
shown in Figure 2. An emission peak located at 538 nm is found
which is consistent with that of the reference device by using
Alq3 as the ET and the emitter. These indicate that only Alq3
gives an emission in these devices.
At higher current density than 5 mA/cm2, higher luminance
CH3
N
N
N
N
1-BuOH
H3C
CO CO
CH3
7
for OLEDs. Alq3 is still the most efficient ET materials since
CH3
the first thin film OLED has been used, although the electron
mobility of phenanthroline8 is faster than that of Alq3, the
prompt crystallization prevents it from a practice. Here, we
report a series of highly efficient ET phenanthroline derivatives
containing quinoxaline moieties, in which phenanthroline and
quinoxaline moieties are coupled to form planar compounds
resulting in high electron mobility; moreover, larger groups of
fluorene are applied to reduce the crystallization of the phenan-
throline derivative.
N
N
NH2
NH2
1
CH3
+
H3
C
H3
C
CH3
H3C
CH3
N
N
N
N
1-BuOH
CO CO
2
H3
C
CH3
Scheme 1. Syntheses of the phenanthroline derivatives 1 and 2.
The phenanthroline derivatives were synthesized as shown
in Scheme 1 following the reference.9 2,3-Di-p-tolylpyrazino-
[2,3-f][1,10]phenanthroline (1) and 2,3-bis(4-(9,9-dimethyl-
9H-fluoren-2-yl)phenyl)pyrazino[2,3-f][1,10]phenanthroline (2)
were synthesized from 5,6-diamino-1,10-phenanthroline react-
ing with 1,2-di-p-tolylethane-1,2-dione and 1,2-bis(4-(9,9-di-
methyl-9H-fluoren-2-yl)phenyl)ethane-1,2-dione,10 respective-
ly. The purified resultant phenanthroline derivatives by sublima-
tion were identified by 1H NMR and mass spectrometric and
elemental analyses.11
The absorption spectra of 1 and 2 were measured on films
(about 100 nm) that were deposited by high-vacuum (10ꢁ6 Torr)
Figure 1. The structure of the OLEDs.
Copyright Ó 2007 The Chemical Society of Japan

 Xiao, Lixin
Xiao, Lixin
 Lan, Hsinglin
Lan, Hsinglin
 Kido, Junji
Kido, Junji