Evaluation Only. Created with Aspose.PDF. Copyright 2002-2021 Aspose Pty Ltd.
Full Paper
doi.org/10.1002/chem.202101508
Chemistry—A European Journal
Versatile Reaction Pathways of 1,1,3,3,3-
Pentafluoropropene at Rh(I) Complexes [Rh(E)(PEt3)3] (E=H,
GePh3, Si(OEt)3, F, Cl): C-F versus C-H Bond Activation Steps
Abstract: The reaction of the rhodium(I) complexes [Rh-
(E)(PEt3)3] (E=GePh3 (1), H (6), F (7)) with 1,1,3,3,3-pentafluor-
opropene afforded the defluorinative germylation products Z/
E-2-(triphenylgermyl)-1,3,3,3-tetrafluoropropene and the fluo-
rido complex [Rh(F)(CF3CHCF2)(PEt3)2] (2) together with the
alents of complex 2 reacted further by CÀ F bond oxidative
addition to yield [Rh(CF=CHCF3)(PEt3)2(μ-F)3Rh-
(CF3CHCF2)(PEt3)] (9). The role of the fluorido ligand on the
reactivity of complex 2 was assessed by comparison with the
analogous chlorido complex. The use of complexes 1, 4 and 6
as catalysts for the derivatization of 1,1,3,3,3-pentafluoropro-
pene provided products, which were generated by hydro-
defluorination, hydrometallation and germylation reactions.
fluorophosphorane
E-(CF3)CH=CF(PFEt3).
For
[Rh(Si-
(OEt)3)(PEt3)3] (4) the coordination of the fluoroolefin was
found to give [Rh{Si(OEt)3}(CF3CHCF2)(PEt3)2] (5). Two equiv-
Introduction
the formation of the strong element-fluorine bonds, such as
HÀ F, BÀ F, GeÀ F or SiÀ F bonds, in CÀ F bond activation reactions,
but also the control of regio- and chemoselectivities, which is
related to the activation pathways.[5o] However, the reactivity of
pentafluoropropenes have been barely studied, neither at
transition metal nor at main group element compounds.
Notably, the Lentz group has previously described the catalytic
hydrodefluorination of 1,1,3,3,3-pentafluoropropene using gal-
lium hydrides[8] as well as at titanium complexes in the presence
of silanes.[6a] No stoichiometric studies have been performed,
yet.
Herein, the reactivity of 1,1,3,3,3-pentafluoropropene to-
wards several rhodium (I) complexes is reported. The reaction
routes impart coordination of the olefin as well as C(sp2)-F and
C(sp2)-H bond activation pathways. In addition, the formation of
a dirhodium(III) fluorido complex bearing bridging fluorides is
described. Catalytic derivatization reactions of the olefin using
both HSi(OEt)3 and HGePh3 were achieved.
The interest in hydrofluoroolefins (HFOs) is partly due to their
lower ozone depletion and global warming potentials when
compared to the ones for chlorofluorocarbons and hydro-
fluorocarbons, which resulted in the replacement of the latter
as refrigerants by HFOs in automobile air conditioning
systems.[1] In addition, HFOs can be used as monomers in
fluorinated polymers.[2]
In order to safely use these compounds in industry,
extended studies into their properties and reactivity is
advisable.[3] Furthermore, they might be considered as useful
building blocks to generate molecules of higher value.[4] One
approach imparts the development of routes for main group or
metal-mediated functionalization including both CÀ F and CÀ H
bond activation reactions.[5] In this regard, studies on the
chemistry of HFOs such as 2,3,3,3-tetrafluoropropene, E-1,3,3,3-
tetrafluoropropene, 3,3,3-trifluoropropene or hexafluoropro-
pene have been in the focus of interest of several research
groups.[6] At rhodium, the high reactivity of rhodium(I) com-
plexes [Rh(E)(PEt3)3] (E=H, F, GePh3, B(O2C2Me4) or Si(OEt)3) Results and Discussion
towards fluoroaromatics and fluoroolefins has already been
demonstrated.[5o,7] The nature of the anionic ligand is crucial for
the activation step for a number of reasons. Key factors include
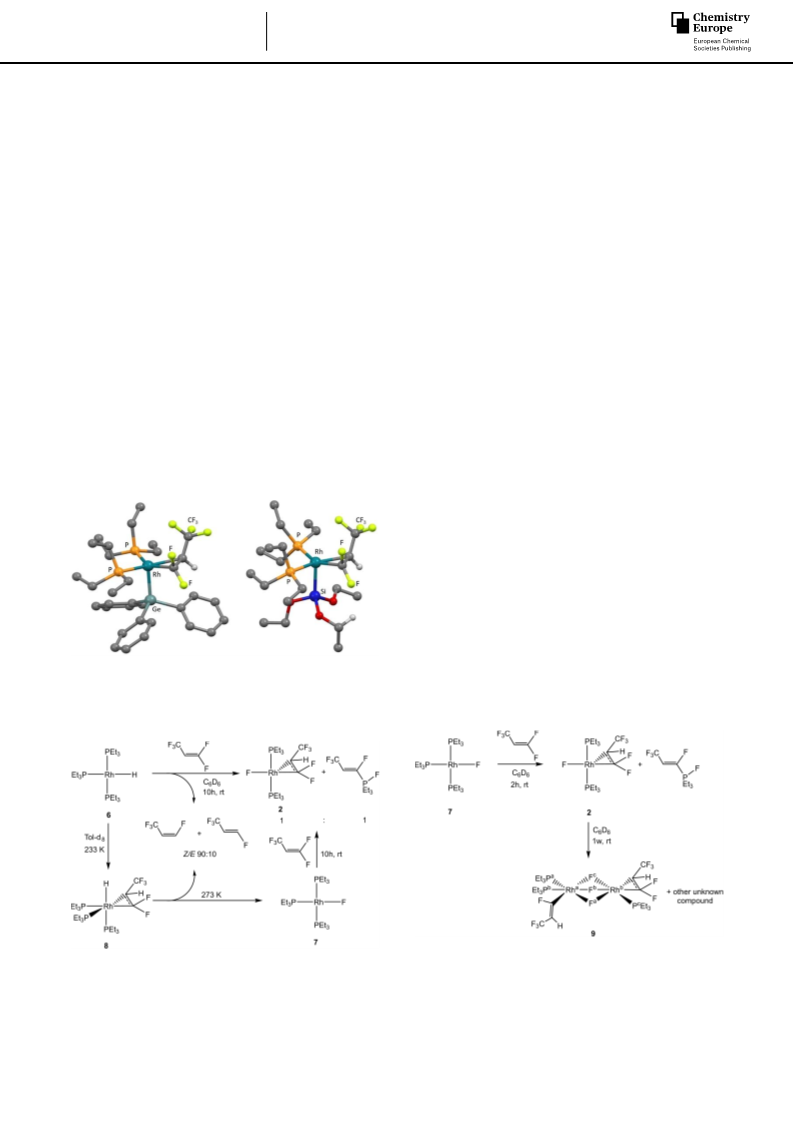
Treatment of the rhodium(I) germyl complex [Rh(GePh3)(PEt3)3]
(1) with an excess of 1,1,3,3,3-pentafluoropropene in the
presence of NEt3/Cs2CO3 yielded, after 30 minutes, the rhodium
fluorido complex [Rh(F)(CF3CHCF2)(PEt3)2] (2). In addition, the
formation of one equivalent of the fluorophosphorane E-(CF3)
CH=CF(PFEt3) as well as one equivalent of the defluorinative
germylation products Z/E-2-(triphenylgermyl)-1,3,3,3-tetrafluor-
opropene (Z/E ratio 35:65) was observed (Scheme 1). NMR
spectroscopic studies showed that the reaction does not occur
at low temperature and, therefore, no intermediates were
detected. However, after 5 minutes at room temperature (31%
conversion of complex 1), complex [Rh(GePh3)(CF3CHCF2)(PEt3)2]
(3) and complex 2 were identified with 19% and 12%
conversion, respectively. Compound 3 then reacted further to
[a] Dr. M. Talavera, Prof. Dr. T. Braun
Department of Chemistry
Universität zu Berlin
Brook-Taylor Str. 2 12489 Berlin (Germany)
E-mail: thomas.braun@cms.hu-berlin.de
Supporting information for this article is available on the WWW under
© 2021 The Authors. Chemistry - A European Journal published by Wiley-
VCH GmbH. This is an open access article under the terms of the Creative
Commons Attribution Non-Commercial NoDerivs License, which permits use
and distribution in any medium, provided the original work is properly cited,
the use is non-commercial and no modifications or adaptations are made.
Chem. Eur. J. 2021, 27, 1–10
1
© 2021 The Authors. Chemistry - A European Journal published by Wiley-VCH GmbH
��
These are not the final page numbers!

 Braun, Thomas
Braun, Thomas
 Talavera, Maria
Talavera, Maria